
VETERINARY PRACTICE GUIDELINES
2020 AAHA/AAFP Feline Vaccination Guidelines*
Amy E. S. Stone, DVM, PhD
y
, Gary O. Brummet, DVM, Ellen M. Carozza, LVT, Philip H. Kass, DVM, MPVM, MS, PhD,
DACVPM (Specialty in Epidemiology), Ernest P. Petersen, DVM, PhD, DABVP (Feline), Jane Sykes, BVSc (Hons), P hD,
DACVIM, MBA, Mark E. Westman, BVSc (Hons), PhD, MANZCVS (Animal Welfare), GradCert Ed Stud (Higher Ed)
ABSTRACT
The guidelines are a consensus report on current recommendations for vaccination of cats of any origin, authored by a
Task Force of experts. The guidelines are published simultaneously in the Journal of Feline Medicine and Surgery (volume
22, issue 9, pages 813–830, DOI: 10.1177/1098612X20941784) and the Journal of the American Animal Hospital Association
(volume 56, issue 4, pages 249–265, DOI: 10.5326/JAAHA-MS-7123). The guidelines assign approved feline vaccines to
core (recommended for all cats) and non-core (recommended based on an individualized risk-benefit assessment) cate-
gories. Practitioners can develop individualized vaccination protocols consisting of core vaccines and non-core vaccines
based on exposure and susceptibility risk as defined by the patient’s life stage, lifestyle, and place of origin and by
environmental and epidemiologic factors. An update on feline injection-site sarcomas indicates that occurrence of this
sequela remains infrequent and idiosyncratic. Staff education initiatives should enable the veterinary practice team to be
proficient in advising clients on proper vaccination practices and compliance. Vaccination is a component of a preventive
healthcare plan. The vaccination visit should always include a thorough physical exam and client education dialog that
gives the pet owner an understanding of how clinical staff assess disease risk and propose recommendations that help
ensure an enduring owner-pet relationship. (J Am Anim Hosp Assoc 2020; 56:249–265. DOI 10.5326/JAAHA-MS-7123)
AFFILIATIONS
From the Department of Small Animal Clinical Sciences, University of Florida,
Gainesville, Florida, USA (A.E.S.S.); Veterinary Teaching Hospital, College of
Veterinary Medicine, University of Illinois at Urbana-Champaign, Urbana,
Illinois, USA (G.O.B.); Nova Cat Clinic, Arlington, Virginia, USA (E.M.C.);
Department of Population Health and Reproduction, School of Veterinary
Medicine, University of California, Davis, Davis, California, USA (P.H.K.);
Animal Hospital of Parkland, Tacoma, Washington (E.P.P.); University of Cal-
ifornia, Davis, Davis, California, USA (J.S.); and Sydney School of Veterinary
Science, University of Sydney, Sydney, New South Wales, Australia (M.E.W.).
KEYWORDS
Vaccination pr inciples; vaccines; lifestyle; risk assessment; veterinarian;
injection site; rabies; leuk emi a; guidelines; maternally derived antibodies
* Boehringer Ingelheim Animal Health USA Inc., Elanco Animal Health,
Merck Animal Health, and Zoetis Petcare supported the development of
the 2020 AAHA/AAFP Feline Vaccination Guidelines and resources
through an educational grant to AAHA.
†
A. E. S. Stone was chair of the 2020 AAHA/AAFP Feline Vaccination
Guidelines Task Force.
These guidelines were prepared by a Task Force of experts convened by
the American Animal Hospital Association (AAHA) and the American
Association of Feline Practitioners (AAFP) and were subjected to a formal
peer-review process. This document is intended as a guideline only, not an
AAHA or AAFP standard of care. These guidelines and recommendations
should not be construed as dictating an exclusive protocol, course of
treatment, or procedure. Variations in practice may be warranted based on
the needs of the individual patient, resources, and limitations unique to
each individual practice setting. Evidence-based support for specific rec-
ommendations has been cited whenever possible and appropriate.
Other recommendations are based on practical clinical experience and
a consensus of expert opinion. Further research is needed to document
some of these recommendations. Because each case is different, vet-
erinarians must bas e their decisions on the best a vailable scientific
evidence in conjunction with their own knowledge and experience.
DNA (deoxyribonucleic acid); FCV (feline calicivirus); FeLV (feline leu-
kemia virus); FHV-1 (feline herpesvirus type 1); FIP (feline infectious
peritonitis); FISS (feline injection-site sarcoma); FPV (feline panleuko-
penia virus); Ig (immunoglobulin); IM (intramuscular); MDA (maternally
derived antibodies); SC (subcutaneous); WSAVA (World Small Animal
Veterinary Association)
ª 2020 by American Animal Hospital Association, American Association of Feline Practitioners, and International Society of Feline Medicine JAAHA.ORG 249
Introduction
As a medically essential and cost-effective method of infectious
disease control, vaccination continues to be a mainstay of feline
practice and a critical component of an individualized preventive
healthcare plan. These guidelines provide the most current infor-
mation and recommendations for feline vaccination as determined
by a Task Force of experts in feline practice. The recommendations
are evidence-guided, based on current peer-reviewed literature and
data, and complemented by clinical insights collectively derived from
decades of experience. The guidelines update the 2013 AAFP Feline
Vaccination Advisory Panel Report and utilize similar recommenda-
tions from the 2016 WSAVA [World Small Animal Veterinary As-
sociation] Guidelines for the Vaccination of Dogs and Cats.
1,2
Both of
these previously published resources should still be considered rel-
evant and actionable complements to the 2020 guidelines.
The guidelines continue the established approach of considering
inclusion of core (recommended for all cats) and non-core (rec-
ommended based on an individualized risk-benefit assessment)
vaccines in an individualized protocol. As explained in the guidelines,
a patient-specific vaccination plan should consider environmental
risk factors and life stage and lifestyle factors that determine the
likelihood of infectious disease exposure and susceptibility. For
example, not all feline patients originate from a home environment,
and conversely, most cats described as “indoor only” might find
themselves periodically exposed to other cats. The guidelines discuss
other presentation scenarios that can potentially affect a risk-benefit
assessment and include updates on feline injection-site sarcomas
(FISSs) and other vaccination-related reactions.
A key component of the guidelines are comprehensive, easy-to-
reference tables listing approved core and non-core feline vaccines and
the relevant considerations for their use. The guidelines are com-
plemented by an online resource center at aaha.org/felinevaccination
and supplemental materials at catvets.com/vaccination. The online
resources include frequently asked questions about vaccination that
clinicians and pet owners raise as well as a vaccine protocol calculator
that uses a cat’s life stage and lifestyle information to suggest an
appropriate, indivi dualized vaccination protocol.
The guidelines discuss in some detail the importance of staff and
client education in implementing vaccination protocols and rec-
ommendations for feline patients. This emphasis is noteworthy in
view of the fact that many pet owners, especially cat owners, associate
professional veterinary care primarily with two events, vaccination
and treatment of acute conditions.
3
Thus, a healthcare visit for the
purposes of vaccination becomes an opportunity to more broadly
discuss an overall preventive healthcare strateg y with the pet owner.
Implicit in this approach is an explanation of how the clinician
considers life stage, lifestyle, patient health status, environmental,
and epidemiologic factors in making vaccination recommendations.
The vaccination event then occurs in the context of a practitioner-
client discussion on how preventive healthcare forms the basis for
the pet owner to maintain a long, rewarding relationship with the
animal in his or her care.
Vaccination Principles
Active immunization, achieved through proper vaccination, plays a
critical role in the control of infectious diseases, both for individual
cats and for the cat population as a whole. Some vaccines also reduce
the potential for spread of zoonotic infections to humans (e.g., ra-
bies). The benefits of routine, widespread vaccination are clear: the
incidence of serious disease caused by pathogenic organisms, such as
feline panleukopenia virus (FPV), can be reduced dramatically when
widespread vaccination is practiced. However, the quality of vaccine-
induced immunity is influenced by the patient’s environment, the
characteristics of the vaccine, the pathogen, and the patient’s
immune competence. Accurate prediction of the outcome of vacci-
nation or the likelihood of exposure to a pathogen is impossible.
Therefore, it is important that veterinarians inform cat owners that
vaccination is not a guarantee of protection.
In general, kittens are more susceptible to infection and disease
than adults. Thus, they represent a primary target population for
immunization. As part of a routine wellness program, the vaccination
needs of all cats should be assessed annually, in conjunction with a
comprehensive physical examination, modifying vaccination and
other control recommendations as necessary based on the current
risk (see “Vaccination Risk-Benefit Assessment”).
Kittens born to immune queens lack significant transplacentally
acquired antibodies
4
and instead absorb specific maternally derived
antibodies (MDA) through colostrum, which provides important
protection during early life. Most absorption occurs within 24 hours
of birth. However, this MDA also interferes with active immuniza-
tion. Serum MDA inhibits immunoglobulin (Ig)G production
within the neonate through negative feedback mechanisms. It also
neutralizes vaccine antigens and prevents them from stimulating an
immune response. MDA then declines at a variable rate. Maternally
derived IgG in kittens in one study was lowest at around 3–4 weeks
of age, and serum IgG and IgA increased dramatically at 5–7 weeks
of age.
4
These results suggested that kittens may be susceptible to
infectious diseases at about 1 month of age, perhaps as much as
2 weeks earlier than puppies.
Nevertheless, it is critical to recognize that there is considerable
individual variation in the rate of decline of MDA, and some kittens
maintain high concentrations for months.
5
The persistence of MDA
is one of the most common reasons for vaccine failure. The amount
250 JAAHA | 56:5 Sep/Oct 2020
of MDA in a kitten at any one time point cannot be predicted be-
cause it varies depending on the titer of the dam and the amount of
colostrum ingested after birth. As a result, a series of vaccinations is
administered to kittens every 2–4 weeks through 16–18 weeks of age in
order to increase the chance that successful immunization will occur
soon after the decline of MDA to sufficiently low titers. The series is
started no sooner than 4 weeks of age, because neonates are more
likely to develop vaccine organism–associated disease and may not
respond well to vaccination. During administration of the series, a
window exists when MDA concentrations are high enough to in-
terfere with immunization but are not sufficient to prevent natural
infection. This window of susceptibility can be minimized by de-
creasing the interval between vaccinations in the series, althoug h use
of intervals less than 2 weeks can interfere with successful immu-
nization, especially with attenuated live vaccines.
Once vaccination has been successfully achieved after the de-
cline of MDA, it is generally recommended that a booster vaccine be
given 3–4 weeks later (this is especially important for inactivated
vaccines, although a boostering effect will also occur following
revaccination with attenuated live vaccines). This means that the
series must be extended 3–4 weeks beyond the period in which the
decline in MDA occurs, with the final vaccination dose being a booster.
In the past, it was recommended that revaccination be performed
1 year after the initial kitten series, and then for most vaccines every
3 years thereafter. However, owing to studies that suggest up to one-
third of kittens may fail to respond to a final core vaccine at
16 weeks and may have blocking MDA at 20 weeks, the WSAVA
recommends that the 1 year vaccine (feline viral rhinotracheitis-
calicivirus-panleukopenia only) be replaced with revaccination at
6 months of age.
2,6,7
In this update, this Task Force has adopted the same recom-
mendation of revaccination against FPV, feline herpesvirus type 1
(FHV-1), and feline calicivirus (FCV) at 6 months of age to potentially
reduce the window of susceptibility in kittens with MDA toward the
end of the kitten series (16–18 weeks). The Task Force recognizes that
this means an additional visit will still be necessary for adminis-
tration of the annual feline leukemia virus (FeLV) and rabies vac-
cinations in young cats.
The risk of infection and disease varies with factors such as the age
and health of the cat, magnitude of exposure to the infectious agent,
the pathogenicity of the agent, and the vaccination history of the cat.
Some of the factors that impact an individual animal’s ability to re-
spond to vaccination include interference from MDA, congenital or
acquired immunodeficiency, concurrent disease, inadequate nutri-
tion, chronic stress, and very young or old age. Some vaccines (e.g.,
those for FPV) induce a stronger protective response than others (e.g.,
those for FHV-1). Because vaccine-induced protection is variable and
not absolute, vaccination should not be used as the only form of
protection, and other control measures, such as those that reduce
exposure to infectious agents, should also be employed.
Types of Feline Vaccines
Vaccines, including those from different manufacturers that are li-
censed to protect against the same pathogen, should not be assumed
as equivalent. Differences in processes and technology used to
produce vaccines, as well as additives such as adjuvants, and vaccine
route of administration influence effi cacy, safety, and duration of
immunity. Vaccines may be inactivated, attenuated live, or recom-
binant (Table 1). All veterinary vaccines, before licensing, are
assessed for efficacy, safety, potency, and purity. Vaccine efficacy is
often expressed as preventable fraction,defined as the proportion of
vaccinated animals that do not develop a disease after challenge (so-
called sterilizing immunity, e.g., FPV, FeLV, and rabies vaccines),
compared with unvaccinated animals that do develop the disease. It
can also be expressed as mitigatable fraction (proportion with re-
duction in severity of clinical signs, e.g., FHV-1 and FCV vaccines).
Other claims include reduction of pathogen shedding, prevention of
a speci fic clinical sign, or prevention of mortality. The level or de-
gree of protection claim can therefore be limited.
Inactivated vaccines are vaccines in which the target pathogen is
“killed” and therefore unable to replicate in the host. Although these
vaccines are unable to revert to virulence, they often contain adju-
vants and other excipient proteins to promote an adequate immune
response, which have been implicated in acute and delayed adverse
reactions in cats. Inactivated vaccines produce weaker immune re-
sponses of shorter duration when compared with attenuated live
vaccines, and more frequent booster immunizations may be required
(generally annually). W ith the ex c eption of rabies, two initial doses of
vaccine 3–4 weeks apart in the absence of MDA are absolutely essential
to produc e an effective immune r esponse, and if more than 6 weeks
elapses between these doses, it is recommended in other guidelines
reports that the series be repeated.
2,8
Full protection may not develop
until 2–3 weeks after the last dose. Inactivated vaccines ar e generally
considered safer than attenuated live vaccines for use during pregnancy
and in immunosuppressed animals, although systemic allergic reac-
tions could still jeopardize pregnancy.
Attenuated live vaccines (modified live vaccines) contain mi-
croorganisms that are artificially manipulated so as to reduce their
virulence or are field strains of low virulence. Repeated passage
through cell culture is the most common means of attenuation.
Because organisms in attenuated live vaccines replicate in the host,
they stimulate an immune response that more closely mimics
protection from natural infection. There is generally a more rapid
onset of immunity than with inactivated vaccines, and, in the
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 251
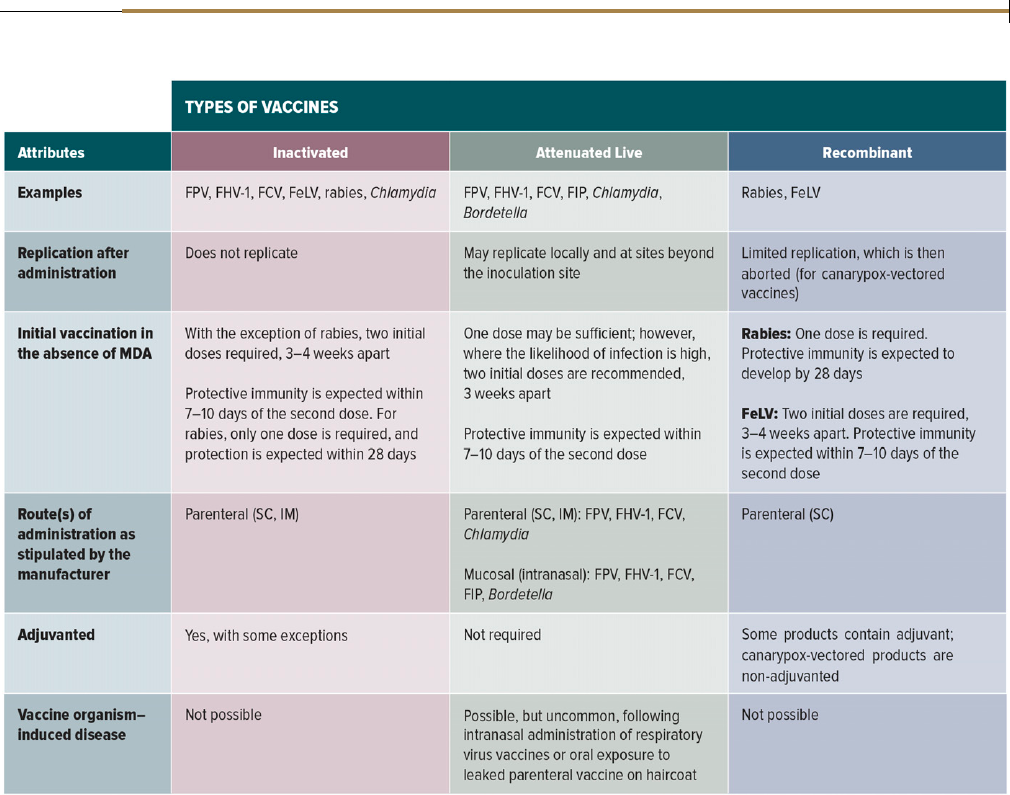
absence of MDA, only one dose of vaccine may be sufficient to
provide protection. Partial immunity after vaccination with a single
dose of attenuated live FPV vaccines can occur within hours.
9–11
In
addition, live vaccine organisms that are shed can immunize other
animals in a population. However, the potential for vaccine or-
ganism–induced disease exists. This is most likely to occur in
immunosuppressed animals, su ch as n eonate s that are younger
than 4 weeks old. In addition, use of attenuated live vaccines is
more likely to result in the generation of false-positive results as
indicated by diag nostic tests that are designed t o dete ct the
target pathogen (antigen or nucleic acid). With prolonged
shedding of live vaccine o rganisms, this can be a problem for
weeks after vaccination. All bacter ial and viral vaccines licensed
for intranasal administration in cats are attenuated live, as are a
number of parenter al vaccines.
Recombinant vaccines are created through manipulation of the
deoxyribonucleic acid (DNA) of a pathogen in the laboratory, with
reduction in pathogen virulence. Types of recombinant vaccines
include subunit, deletion mutant, vectored, and DNA vaccines.
Currently, the only available recombinant vaccines for cats in North
America are vectored vaccines, which use a recombinant canarypox
virus as a vector. In these vaccines, DNA of the pathogen that en-
codes for an immunogenic antigen is incorporated into the canar-
ypox genome, which then undergoes aborted (limited) replication
in the host with expression of the immunogen, in turn inciting a
protective immune response. Compared with inactivated vaccines,
canarypox vectors offer a more rapid onset of immunity and may be
more effective in the face of persistent MDA. Canarypox-vectored
vaccines also do not requir e adjuvant and have been associated with a
reduced risk of injection-site sarcomas in cats.
12
However , one study
suggested that the degree of protection induced by the recombinant
canarypoxFeLVvaccinemaynotbeasrobustasthatinducedby
whole inactivated FeLV vaccines,
13
which might produce sterilizing
immunity.
14
However, moderate to severe immunosuppression
TABLE 1
Types of Feline Vaccines and Their Attributes
252 JAAHA | 56:5 Sep/Oct 2020

may have impacted the results, so further studies are required to
determine whether a clinically impor tant difference e xists.
To facilitate vaccine selection, vaccines for dogs and cats have been
divided into core vaccines, non-core vaccines, and those generally not
recommended. Core vaccines are for all cats with an unknown vacci-
nation history. The targeted diseases cause significant morbidity and
mortality and are widely distributed. In general, vaccination for core
diseases results in good protection. The Task Force recommends
vaccines for FHV-1, FCV, FPV, rabies, and FeLV (cats younger than 1
year old) as core vaccines (Table 2, pet cats; Table 3, shelter-housed
cats). N on-core vaccines are optional vaccines that should be consid-
ered in the light of exposure risk; that is, based on geographic dis-
tribution and the lifestyle of the cat (Table 4). Optional or non-core
vaccines for cats include FeLV (for cats older than1 year), Chlamydia
felis, and Bordetella bronchiseptica vaccines.
The not generally recommended category of vaccines pertains to
diseases of low clinical significance or that respond readily to treatment;
vaccines for which evidence of efficacy in the field is minimal; or
vaccines that may produce a relatively higher incidence of adverse
events with limited benefit. The Task Forc e lists the feline infectious
peritonitis (FIP) vaccine as not general ly rec om mended (Table 5). This
vaccine is labeled for administration from 16 weeks of age, whereas
many kittens become infected with coronaviruses well before this age. It
also contains a serotype II strain of FIP virus. Serotype I FIP virus
strains predominate in the field and do not have cross-reactiv e neu-
tralizing epitopes with serotype II strains. Therefor e, as noted in the
previous iteration o f these guidelines,
1,33
there remains insufficient e v-
idence that this vaccine induces clinically relevant protection in the
field.
The decision to vaccinate, even with core vaccines, should be
based on a risk-benefit assessment for each cat and for each vaccine
antigen. Benefits of vaccination should be balanced against the risk
of adverse events, likelihood of exposure, and disease severity. Every
effort should be made to ensure that cats are healthy before vacci-
nation. However, concurrent illness (including retroviral infections)
does not necessarily preclude vaccination.
34
The 2020 AAFP Feline
Retrovirus Testing and Management Guidelines state that vaccines
should not be avoided in cats with retroviral infection because they
can develop more severe clinical disease related to FPV and upper
respiratory tract infections after natural exposure compared with
uninfected cats.
34
Potential Therapeutic Benefits of Vaccination
Active immunization can enhance non-specific immunity, leading to
reduction in disease caused by non-target pathogens. One study
showed that vaccination of cats with an intranasal FHV-1-FCV
vaccine was associated with reduction in clinical signs following
challenge with B bronchiseptica.
24
More studies are needed to assess
the non-target effects of different vaccine types. There is also interest
in whether vaccines might provide therapeutic benefits in cats al-
ready infected with target pathogens. Improvement in chronic up-
per respiratory tract signs that were previously refractory to other
treatments was documented in 13 cats vaccinated with an intranasal
FHV-1-FCV vaccine.
35
Most vaccines, however, provide no thera-
peutic benefit, as clearly documented for FeLV vaccines.
36
Vaccination Risk-Benefit Assessment
The Task Force supports the WSAVA’s recommendation that veterinarians
should vaccinate every animal with core vaccines and give non-core
vaccines no more frequently than is deemed necessary .
2
The decision
whether or not to administer a vaccine to a cat, and how frequently , relies
on an individual case-by-case assessment by the veterinarian. This in-
volves consideration of the animal, the animal’s enviro nment, and the
pathogen in question. Additionally , risk-benefit assessments should
consider the safety of the vaccine, other adverse effects of vaccination
(e.g., the effect of f eline immunodeficiency virus vaccination on in-clinic
diagnostic test kits), and the efficacy of the vaccine. The result of this
assessment should be an individualized, evidence-guided rec ommenda-
tion to vaccinate or not to vaccinate.
Patient’s Characteristics
Age is an important factor in assessing an individual’sriskprofile. In
contrast to puppies, kittens born to immune queens appear to lack
transplacentall y acquired antibodies and instead absorb specificMDA
through colostrum,
4
whichprovidesimportantprotectionduringearly
life. Once MD A have waned, however , kittens become susceptible to
infection. Most infectious diseases are more prevalent in kittens than
adults, and therefore, kittens (in particular, those younger than 6
months old) represent a principal primary target population for vac-
cination. Conv ersely, adult cats generally ha ve a more robust adaptive
A Balancing Act
There is always a balance to be struck when considering risks as-
sociated with vaccination and benefits of vaccination for the individual
patient:
·
A decision to vaccinate might involve a young cat resid-
ing in a multi-cat household with outdoor access, living
in an area with a known high prevalence of the pathogen
being vaccinated against.
·
A decision not to vaccinate might in v olve a s enior or geri-
atric cat residing in a single-cat household with no outdoor
access, and a vaccine that has poor efficacy against a path-
ogen with low virulence or limited local prevalenc e.
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 253

TABLE 2
Core Vaccines for Pet Cats
254 JAAHA | 56:5 Sep/Oct 2020

TABLE 2
Table 2 continued
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 255

TABLE 3
Core Vaccines for Shelter-Housed Cats
256 JAAHA | 56:5 Sep/Oct 2020

immune r esponse when challenged (assuming they are healthy and not
immunocompromised), whether due to previous natural exposure or
vaccinatio n, and age-related resistanc e to challenge is particularly a
feature of FeLV infection.
26
Consequently, vaccination of mature cats is
generally considered less critical than vaccination of kittens. The pres-
ence of concurrent disease or stress causing immunosuppression should
also be a consideration prior to vaccination because this may affect an
animal’s susc eptibility to infection and response to vaccination.
Patient’s Environment
Population density and opportunity for exposure to infectious agents
are two critical issues that should form part of the risk-benefi t
assessment. In general, cats and kittens living in larger multi-cat
households and environments (e.g., boarding, breeding, foster, or
shelter facilities) have a higher risk of infection than cats living in one-
or two-cat households. In addition to the possible presence of infected
animals acting as reservoirs for infection in multi-cat households, the
immunosuppressive effects of stress associated with high-density feline
housing may result in reactivation of some infections as well as in-
creased susceptibility to new infections. The introduction of new cats
into multi-cat households also increases the risk of infectious disease
not only to the cat entering the household but also to the whole group
because of possible direct exposure to new infectious agents.
When assessing the opportunity for exposure to a given pathogen
foranindividualcat,thelifestyleofthecatandothercatsinthesame
household needs to be considered. It is critical to determine whether the
cat is indoor -only or has outdoor access (including supervised outdoor
visits on a harness, or boarding) because cats with outdoor access may be
at increased risk of pathogen exposure. Indoor-only cats, however , may
stillbedeterminedtobeatriskofexposuretopathogens,eitherfrom
other cats in the household (i.e., subclinically infected or carrier cats), or
by fomite transmission of pathogens br ought in from out side on the
owner’s body , clothing, or s hoes. Indoor-only cats may also be exposed
to infectious agents when brought to a veterinary clinic for a wellness
examination. In theory , strictly indoor cats may be more susceptible to
developing some infectious diseases (such as FPV and FCV infection)
than cats with outdoor ac ce ss because they may not receive “natural
boosting of immunity” that occurs with natural exposure.
1
The geographic distribution of infectious agents may also result
in different risks of exposure (e.g., rabies), and therefore, questions
regarding future travel should be included in determining the risk of
exposure to specific infectious agents.
TABLE 5
Not Generally Recommended Vaccines for Pet Cats
TABLE 4
Non-Core Vaccines for Pet Cats
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 257

Infectious Agents
The likelihood of infection and disease is influenced by pathogen
factors such as virulence, strain variation, and challenge dose
(i.e., how many infectious units of exposure). The need for vacci-
nation is greatest against pathogens with high virulence, such as FPV,
and pathogens that cause widespread morbidity, such as FHV-1.
Creating an Individualized, Lifestyle-Based
Vaccination Plan
The vaccination needs of each cat should be evaluated individually
and rationally, based on health status, age, and possible, realistic
exposure to disease (Table 6). Owners and veterinarians must work
together to determine the likelihood of the animal coming into
contact with other animals that may spread disease, acquiring par-
asites that may harbor a disease-causing agent, or living in an area
where a disease is known to be endemic or very widespread.
2
Questions must be asked about the lifestyle of that specific c at as
well as any other cats in the household or potentially introduced into the
household. The travel, boarding, housing, and enrichment activities or
excursions outside of t he home should also be considered.
1
This risk
assessment for exposure to disease should be done at least once a year.
Thelifestageofthecatmustalsobeconsideredwithrespectto
possibility of exposure to disease and the health status of the animal.
Infectious diseases are more pr evalent in kittens, and in general, kittens
(younger than 6 months old) are more susceptible to infection.
1
Younger
cats also tend to behave more unpre dictably and require more enrich-
ment activities, which ma y increase their opportunity for exposure.
37
The health status of the individual cat, including any previously
documented adverse events to vaccines, also determines the vaccination
recommendations. The nutritional status, general health (i.e., any
concurrent infections or other disease) and the pregnancy status of
females may change the type and schedule of vac cinatio n for that in -
dividual cat (Table 6). As with lifestyle changes, changes in health status
must be evaluated at least yearly.
1
The population density, along with the opportunity for expo-
sure to other cats, is a major factor in determining the need for
vaccination. Larger multi-cat households are likely to have a greater
risk of infection and disease than households of one or two cats. The
introduction of new cats and the social dynamics of the group may
also cause immunosuppressive stress, leading to increased risk of
disease by new infection or recrudescence. Each cat in a multi-cat
environment must have a vaccination plan that balances the pro-
tection of the individual with that of the household population.
1
Cats entering boarding, breeding, foster , or shelter situations have
increased risk of disease exposure as well as systemic stress. Vaccination
may be warranted prior to entering these environments when possible
(see Tables 2 and 3). Additionally , vaccination intervals may need to be
shortened depending upon t hese possible scenarios.
1
As with multi-cat
households, the vaccination plan for the individual cat must be con-
sideredinrelationtotheentirepopulation.
One vaccination plan or protocol cannot be applied to every cat.
Each animal must be evaluated and an individualized plan created that
will most protect that particular cat. That plan must be reassessed
when changes in health and lifestyle occur, requiring client education
and compliance with at least yearly veterinary visits.
38
Feline Patient Populations
For the purpose of creating specific, individualized vaccination rec-
ommendations based on risk of exposure, the Task Force has identified
and defined the following feline populations based on their environment
and lifestyle. The guidelines begin by discussing pet cats and then discuss
a number of feline populations that are considered to be at relatively high
risk of infectious disease exposure—namely, shelter cats, trap-neuter -
return/t rap-neut er -r elease cats, cattery cats, and foster cats.
Pet Cats
Pet cats include any cat kept by human beings as a source of
companionship and pleasure. Pet cats are further categorized by
TABLE 6
Risk Assessment Variables Determining an Individualized Vaccination Plan
258 JAAHA | 56:5 Sep/Oct 2020
housing status (indoor, outdoor, or indoor-outdoor cats) and
number of cats in the household (single-cat or larger multi-cat).
Although these distinctions are important, the most significant is-
sue to consider regarding vaccination of pet cats is the individual cat’s
exposure risk and exposure frequency to other cats and feline in-
fectious diseases. Even indoor cats from single-cat households will
inevitably be exposed to other feline infectious pathogens
in situations such as a veterinary clinic visit, contact with other cats
entering the premises, or exposure to contaminated fomites intro-
duced by human contact. Client education for owners of these
patients should focus on risk of exposure to other cats rather than
on where the cat eats, sleeps, or spends most of its time.
For high-risk, multi-cat households, the probability of infectious
disease exposure and transmission is proportionate to the number or
density of cats on the premises.
39
It is important to educate clients about
the increased disease risks to this population of cats and to discuss in-
creased owner responsibility t o ensure appropriate preventive healthcare
initiatives associated with housing many cats in a confined space.
Shelter Cats
These are cats living for indeterminate periods in centers for
relinquished or lost animals.
Trap-Neuter-Return/Trap-Neuter-Release Cats
These are community or feral cats of either sex that live entirely separate
from people and cannot safely be handled. Trap-neuter-release/trap-
neuter-return cats may survive completely independently of humans,
but some semiferal colonies receive support from individuals.
Cattery Cats
These cats are maintained in commercial facilities; for example,
breeding or boarding facilities, and pet stores with a showcase model.
Foster Cats
Foster cats are kittens or adult cats temporarily housed for rescue,
rehabilitation, and rehoming purposes. The most important con-
sideration in a foster cat household is ensuring that the permanent
population of the household is appropriately vaccinated to provide
protection from disease exposure originating with foster cats.
Serology and Diagnostics
The inter pretation of an antibody test result can be complex because
antibody testing is used for many reasons. Depending on the anti-
bodies tested for, antibody testing can be used for (1) diagnosis of
infection, (2) identification of previous exposure to pathogens
(particularly in unvaccinated animals), and (3) assessment of im-
munity prior to or following vaccination (Table 7). Clinicians should
understand when and why to perform antibody testing and use this
knowledge to make evidence-based decisions prior to vaccination.
Hemagglutination inhibition (for FPV) and serum neutralization
(for FHV-1, FCV, and rabies) are the reference standards to determine
the presence of effective antibody-mediated immunity. These test
methodologies can only be performed in a laboratory setting using live
cell cultures (i.e., they cannot be performed in a practice using rapid
patient-side test kits). These diagnostic tests are predominantly re-
search tools used in vaccine efficacy and prevalence studies.
It is important when attempting to demonstrate protective im-
munity in a patient using an in-clinic antibody test kit that the per-
formance of the kit be compared against the appropriate reference
standard in order to demonstrate correlation with protective immunity.
The presence of anti-FPV antibodies correlates strongly with
protection (Table 7). Currently, experts recommend antibody testing
for FPV to assess immunity and inform decisions about whether to
vaccinate.
6,40
Rapid in-clinic test kits to detect antibodies to FPV,
FHV-1, and FCV are available to veterinarians in North America and
have been validated in two different studies using the appropriate
reference tests.
47,48
Of concern, however, was the occurrence of some
anti-FPV antibody false-positive results in one study, which in
practice would lead to some unprotected cats not being vaccinated.
48
Adverse Postvaccination Reactions
Although the administration of biological products is never entirely
free of risk, currently available feline vaccines have an excellent safety
record. That said, the true prevalence of adverse reactions is likely to
be underestimated owing to underreporting by both veterinarians
and owners.
49
Therefore, it is important to report any known or
suspected negative events associated with vaccination. In the United
States, veterinarians are requested to contact the manufacturer
(Veterinary Technical Services) of the vaccine(s) considered to be
involved. Veterinarians may also report known or suspected adverse
events directly to the U.S. Department of Agriculture; the Center for
Veterinar y Biologics of the U.S. Department of Agriculture’s Animal
and Plant Health Inspection Serv ice can be contacted by the fol-
lowing means:
·
Website: https://www .aphis.usda.gov/aphis/ourfocus/animalhealth/
veterinary-biologics/adv erse-e vent-r eporting/CT_Vb_adverse_event
·
Mail: Send the report form to the Center for Veterinary Bio-
logics, 1920 Dayton Avenue, PO Box 844, Ames, Iowa 50010
·
Telephone: (800) 752-6255.
At the time of vaccine administration, included in the patient’s
permanent medical record should be the name, ser ial number, ex-
piration date and manufacturer of the vaccine(s) given, date of
administration, name of the person administering the vaccine(s),
and the site and route of the vaccine administration. Adverse events
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 259

should be recorded in a manner that will clearly alert all staff
members during future visits.
Prevalence and Types of Adverse Reactions
Postvaccination adverse events in cats are considered rare.
49
In the
most substantial survey to date, any adverse reactions were recorded
for cats presented to Banfield Pet Hospitals in the United States
between 2002 and 2005.
42
During this period, more than 1.25
million doses of various vaccines were administered to nearly
500,000 cats. Adverse reactions within 30 days of vaccination were
reported at a rate of 0.52% of cats vaccinated. The most commonly
reported vaccine reactions are lethargy, anorexia and fever for a few
days after vaccination, or local inflammation at the site of injec-
tion.
42,50,51
In the Ban field Pet Hospital population, the risk of an
adverse reaction was greatest in cats around 1 year of age and/or
increased as the total volume of vaccine and number of vaccines
administered concurrently increased.
42
Hypersensitivity Reactions
Although anaphylaxis (type I hypersensitivity reaction) is rare
(approximately 1–5 per 10,000 vaccinations),
42,52
it may manifest as
vomiting, diarrhea, respiratory distress, facial or generalized pruri-
tus, facial swelling, and collapse.
51,53,54
Where revaccination is con-
sidered necessary in a cat that has experienced an allergic reaction,
using a different vaccine formulation and premedicating with an
antihistamine and glucocorticoid 20–30 minutes prior to vaccine
administration is recommended, followed by close observation of
the patient for several hours.
42,53
Other forms of hypersensitivity
reactions (types II, III, and IV) almost certainly also occur in cats
after vaccination, but these are rarely documented.
Postvaccination Monitoring
The Task Force recommends that veterinarians and owners monitor
the vaccination site for swelling or lumps using the “3-2-1” rule.
Biopsy of any mass present is warranted if it (1) remains present
3 months after vaccination, (2) is larger than 2 cm in diameter, or
(3) is increasing in size 1 month after vaccination.
1,55
It is recom-
mended to obtain an incisional biopsy on any masses meeting any of
these criteria. Fine-needle aspirates may not provide diagnostic
cellular tissue, whereas excisional biopsies rarely meet appropriate
margins (5 cm in two fascial planes) as required in the case of
injection-site sarcomas, thus increasing the morbidity and mortality
risks associated with sarcoma invasion.
Update on Feline Injection-Site Sarcomas
FISSs, largely caused by vaccines (although other materials have been
implicated), have been recognized since 1991.
56
Three decades later,
much about them remains unknown. Within the United States, FISS
TABLE 7
Possible Uses of In-Clinic Serology Testing
260 JAAHA | 56:5 Sep/Oct 2020

incidence estimates, although low, have varied by at least an order of
magnitude, and worldwide FISS incidence estimates vary by country
depending on the relative use of vaccine types (e.g., FeLV, rabies)
and population susceptibility.
The Task Force makes the following observations regarding
vaccination:
·
Neither vaccinating in the interscapular space nor decreasing
vaccine volume is recommended.
·
Distal limb injection is recommended to facilitate amputation
with 5 cm margins in two fascial planes in the case of injection-
site sarcoma (Figure 1).
·
More recently, ventral abdominal subcutaneous injections have
been used because of the perceived relative ease of tumor removal
without the need for a mputation.
2
Howev er, the need to remove
two fascial planes and 5 cm margins would still necessitate ag-
gressive tissue remo val fro m the abdomen and abdominal c avity.
·
Tail vaccination has also been reported as well tolerated and
elicited acc eptable serological responses to vac cination in the dis-
tal limbs.
57
To facilitate 5 cm margins in the c ase of injection-site
sarcoma, vaccinations must be administered in the distal tail,
something that may not be practical for most clinicians.
·
Follow the 3-2-1 rule for postvaccination swelling.
1,55
Obtain
incisional biopsies for appropriate diagnosis.
The 2013 AAFP Feline Vacc inati on Advisory Panel Report in-
clud ed recommendations for specific vaccine antigens to be ad-
ministered at specific anatomical locations in the distal limbs.
1
This
technique has helped facilitate the identification of the vaccine an-
tigen used if a sarcoma developed subsequently at the injection site.
Since this technique has been widely adopted, these injection-site
recommendations have also led to a shift in the site of tumor for-
mation to the distal limbs, thus facilitating potentially life-saving
surgery for patients suffering from these invasive tumors.
58
The 2020
AAHA/AAFP Feline Vaccination Guidelines Task F or ce rec ognizes and
supports the value of the 2013 recommendations and recognizes that
practitioners may, at times, need to use medically appropriate discre-
tion regarding the anatomical location of vaccine administration.
Practitioners are strongly advised to keep complete, accurate rec or ds
for antigen administration site and route of vaccine administration.
The Task Force offers the following analysis of current research
about vaccine safety:
Experimental studies of vaccine-induced inflammation: These
studies provide weak evidence for detecting differential vaccination
effects on sarcoma incidence yet represent progenitors of the “more
vaccine-induced inflammation leads to increased sarcoma risk”
conjecture. One immediate problem is that it is unclear how to
define inflammation in the context of tumor induction. Macy and
Hendrick (1996) cite an unpublished study that defined inflam-
mation as “the size of the local reaction.”
59
Grosenbaugh et al.
(2004) interpreted it as the presence of “injection-site reaction,”
which could have included “scab, crust, swelling, erosion, ulcera-
tion, or pain at the injection site or development of lameness.”
28
Day
et al. (2007) used histopathological scoring that included quanti-
fying neutrophils, lymphocytes, and macrophages (inflammatory
phase of tissue reaction); quantifying fibroblasts, collagen, and
granulation tissue (repair phase); and assigning a “global severity
score” based on biopsy site reactivity and extension of involvement
of the tissue section.
60
Because the many manifestations of inflam-
mation in cats do not invariably lead to neoplasia, more sensitive
biomarkers such as DNA damage may one day be used to distin-
guish the potential for adjuvanted versus non-adjuvanted vaccines
to induce tumors.
61
But because none of the cats in the above
studies developed sarcomas, such experimental research does not
have logical standing to infer relative vaccine safety.
Associational studies of diagnoses: In the past 10 years, two studies
based on data from pathology registries have provided contradictory
findings. Wilcock et al. (2012) found no decrease in the proportion of
“post-vaccinal sarcomas” in feline skin and subcutaneous mass sub-
missions from 199 2 to 2010 in a Canadian registry despit e the intr o-
duction of a non-adjuvante d rabies vaccine in 2000.
62
In contrast, Graf
et al. (2018) studied the proportion of feline biopsies that were
FIGURE 1
Vaccination sites: Recommended injection sites in the distal limbs and
tail. ª iStock.com/GlobalP.
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 261
fibrosarcomas submitted to Swiss pathology laboratories between 2009
and 2014 and noted “amarkeddropintherelativefrequencyoffi-
brosar c oma diagnoses after t he introduction of a non-adjuvanted FeLV
vaccine into the Swiss market” in 2007 (rabies vaccines ar e rarely used
now in Switzerland).
29
Such studies of diagnostic proportions ar e dif-
ficult to interpret. Moreover , they are influenced not only b y disea se
incidence but also b y factors r elated to differ ential cost and motivation
for histopathologic diagnoses, which are subject to change o ve r time.
Therefor e, there are alwa ys competing explanations for findings.
Longitudinal studies of comparative incidence: A study by Srivastav
et al. (2012) is the only one to perform a comparative (case-contr ol)
analysis of vaccine types in common use in the past 10 years.
12
Unlike
previous epidemiologic studies, it provides tenuous evidence that non-
adjuvanted vaccines ma y be less likely to induce sarcomas than adju-
vanted vaccines. However, the work suffers from sample size limitations
and bias concerns.
30
Although it arguably serves as an epidemiologic-
methods blueprint for futur e inv estigations, it is insufficient to justify a
wholesale r ecommendation for a single vaccine formulation with as yet
unforeseen consequences on population immunity. The Task Force
believes that there is currently insufficient research to justify recom-
mending a single vaccine type. Since injection site sarc omas ar e a risk,
the Task Forc e recommends vaccination in the lower distal limbs to
facilitate clean margins if surgical amputation is required.
Frequently Asked Questions
A summary of frequently asked questions is available online at
aaha.org/felinevaccination and catvets.com/vaccination.
Staff and Client Education
The Veterinarian’s Role and Responsibilities
·
A veterinarian should assess every patient regardless of appoint-
ment type (wellness, acute care or follow-up visit) for current
vaccination status based on age and lifestyle. Informed by this
assessment, an individualized patient vaccination plan should
be developed or modified and then discussed and agreed upon
in collaboration with the cat owner.
·
In addition to overseeing the development of feline vaccination
protocols, the veterinarian should provide staff education on
the following:
o Zoonotic disease prevention.
o Separate administration sites for each vaccination (based on
consistent vaccination site guidelines for that practice).
o Potential life-threatening adverse events (i.e., anaphylaxis)
and minor adverse events (i.e., localized swelling) following
vaccination.
o Vaccine reconstitution and handling (the AAFP recommends
using vaccines within 30 minutes of reconstitution).
53
o Standard sharps safety procedures to prevent accidental nee-
dle sticks.
63
The Centers for Disease Control and Prevention (CDC)
online training module, “You C all the S hots: Vaccine Storag e and
Hand l i n g ,” is a useful resource for staff training on vaccination.
64
Thepracticeshoulddesignateapersontobetheprimaryvaccine
coordinator for the facility. This person will be responsible for
ensuring all vaccines are stored and handled correctly. A second
staff member to ser ve as an alternate in the absence of the pri-
mary coordinator should be appointed (th is is par ticular ly im-
portant in c ase o f after-hours emergencies). Both coordinators
should be fully trained in routine and emergency policies and
procedures.
65
The healthcare team, led by the veterinarian, should emphasize
and educate clients that they are part of a team approach to vaccine
management, requiring the entire staff’s understanding of zoonotic
disease, core and non-core vaccines determined by the pet’s lifestyle,
hospital policy, state law, client compliance, and adverse vaccination
events.
Credentialed Veterinary Technician or Veterinary
Assistant Roles and Responsibilities
A veterinary technician or assistant often assumes the role of des-
ignated vaccine coordinator, assisting in vaccination storage and
inventory management. AAHA guidelines on vaccine storage and
handling, and the CDC Vaccine Storage and Handling Toolkit are
useful resources for this purpose.
64,65
The vaccine coordinator is
often responsible for reconstitution of vaccines and administration
of vaccinations as directed by the attending veterinarian in com-
pliance with state law.
66
This individual is also often given respon-
sibility for implementing feline-friendly handling techniques in the
hospital setting to minimize stress during examinations and vaccine
administration
67
and for maintaining effective client education and
follow-up, including verbal and wr itten instructions on potential
adverse events after vaccine administration and disease prevention.
Roles and Responsibilities of Reception and Other
Client-Service Personnel
The reception staff is typically charged with maintaining patient files
with vaccination information, including date administered, along
with the production lot serial number and expiration date of the
vaccine. Reception personnel are also responsible for contacting
clients and scheduling follow-up appointments for booster series and
yearly vaccinations in advance as directed by the prescribing veter-
inarian. Non-clinical staff should understand the potential life-
threatening and minor adverse events that can occur following
vaccination that require veterinary assistance.
262 JAAHA | 56:5 Sep/Oct 2020

Client Education
Pet owner clients are an essential member of a cat’s healthcare team.
Although clients can be instrumental in helping improve healthcare
for their cats, the Task Force recommends that vaccination be
performed by a veterinarian. Vaccination is a medical procedure.
Vaccines are available through sources other than a veterinarian, but
they may not protect a cat against disease unless properly stored,
handled, and administered. The principles of feline vaccination
outlined in the box below represent a basic client education over-
view for cat owners. To help educate clients about vaccine and
general health issues, both AAHA and the AAFP have handouts
available to members and non-members. Additionally, more ex-
tensive information is available at aaha.org/felinevaccination and
catvets.com/vaccination.
Summary
Vaccination protocols for cats should consist of recommended
core vaccines and discretionar y non-core vaccines as defined and
listed in the guidelines. Vaccines in the latter category are given
based on a risk-benefit assessment. Risk i s det ermined by the
patient’s life stage, lifest yle, clinical history, and health status
and by environmental and epidemiologic risk facto rs. Although
feline vaccination is universally practiced by primar y ca re co m-
panion animal practices, there is no single protocol suitable for
all feline patients. Rather, vaccination of c ats should be patient-
specific and guided by an individual risk-benefit assessment us-
ing the criteria listed in the guidelines. In the case of some
vaccines, practitioners have a choice of different ty pes of anti-
gens, including those that are inact ivated, attenuated, and in
recombinant form. The patient’s clinical and vaccination status,
such as the possible presence of maternally derived immunity or
a histor y of adverse postvaccination reactions, are factors that
may influence t he choice of vaccine type.
Although most feline patients are household pets, practitioners
should anticipate situations in which higher-risk cats are presented
for vaccination, including those from shelter, cattery, feral, or foster
care origins. Adverse postvaccination reactions unavoidably occur in
a small percentage of cats. Because of their neoplastic etiology, FISSs
continue to be the most serious, if infrequent, vaccine-associated
adverse event. Detection of patterns in FISS incidence remains
elusive, and their occurrence continues to be idiosyncratic. Advising
clients in advance of the possibility of hypersensitivity or other re-
actions will help minimize their concerns. All members of the
practice team, including clinical and non-clinical personnel, should
have a well-informed understanding of the importance of vaccination
of feline patients and be able to advise clients of the practice’s ap-
proach to an individualized vaccination plan. The vaccination visit is
an ideal time for a client education dialog in which the clinical staff
Vaccination Talking Points for Clients
Vaccines help protect against specific infectious diseases. They stimulate the body’s immune system to recognize and fight an infection. Without
vaccination, many cats would become seriously ill or die from preventable diseases. Some infections are more difficult to prevent using vaccination than
others. For example, vaccination is very effective against feline panleukopenia infection but does not entirely protect against respiratory virus infections.
However, cats vaccinated against respiratory tract infections generally have milder illness and are far less likely to die from their disease.
A veterinarian is the best person to evaluate a cat’s individual vaccination needs. Many factors need to be taken into consideration when deciding
how often and for what diseases a feline patient needs to be vaccinated. These considerations include health status, age, and lifestyle of the cat; a
vaccine’s duration of immunity; what diseases are prevalent in the area; and the severity of endemic diseases. Even cats living exclusively indoors
require regular vaccination because they still may be exposed to diseases in many circumstances, such as when traveling or boarding, visiting a
veterinary practice, interacting with other cats, or through viruses carried on the pet owner’s hands or clothing.
Veterinarian-administered vaccination is particularly important with respect to rabies. Rabies is a fatal but preventable disease that can be spread to
humans by contact with saliva from an infected individual. If an unvaccinated cat is scratched or bitten by a wild animal, or if it bites a person, it should be
quarantined or euthanized. In many US states, it is against the law for anyone other than a licensed veterinarian to administer a rabies vaccine. Rabies
vaccination of cats is required by law in many but not all states. Ontario is the only Canadian province that requires rabies vaccination of cats. Even in areas
where it is not required, feline rabies vaccination is still recommended (i.e., it is a core vaccine).
Severe vaccine reactions are rare. Veterinarians should convey the appropriate risk-benefit analysis of any vaccination. Cats may experience mild,
short-lived reactions (malaise) such as poor appetite, lethargy, and fever that will resolve without treatment. Clients should seek immediate veterinary
attention if their cat begins vomiting or scratching, develops bumps (hives) or facial swelling, or has difficulty breathing within a few hours of being
vaccinated. The client and veterinary practice team have the same goal: to provide the best possible care for the pet.
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 263

has an opportunity to discuss the role of vaccination as an essential
component of preventive healthcare tailored to the individual patient.
The Task Force gratefully acknowledges the contribution of Mark Dana
of Scientific Communications Services, LL C, and the Kanara Consul-
ting Group, LLC, in the preparation of the guidelines manuscript.
CONFLICT OF INTEREST
Amy E. S. Stone has received speaking fees from Boehringer
Ingel heim Animal Hea lth USA Inc. Jane Sykes receives hon oraria
and research funding from Boehringer Ingelheim Animal Health
US A Inc., Elanco Animal Health, IDEXX Laboratories Inc., and
Merck Animal Health. Ernest P. Petersen was a stockholder of
Phoenix Central Laboratory prior to its sale to Zoetis Petcare (after
these guidelines wer e written) but was not employed at the lab nor on
the Boa rd of Gov ernor s, and was not in vol ved in the sale. The other
members of the Task Force hav e no conflicts of interest to declare.
FUNDING
Boehringer Ingelheim Animal Health USA Inc., Elanco Animal
Health, Merck Animal Health, and Zoetis Petcare supported the
development of the 2020 AAHA/AAFP Feline Vaccination Guidelines
and resources through an educational grant to AAHA.
ETHICAL APPROVAL
This work did not involve the use of animals and, therefore, ethical
approval was not necessarily required.
INFORMED CONSENT
This w ork did not involv e th e use of animals and, ther efore, informed
consent was not required. For any animals individually identifiable
within this publication, informed c onsent (either verbal or written) for
their use in the publication was obtained from the people inv olved.
REFERENCES
1. Scherk MA, Ford RB, Gaskell RM, et al. 2013 AAFP Feline Vaccination
Advisory Panel Report. J Feline Med Surg 2013;15:785–808.
2. Day MJ, Horzinek MC, Schultz RD, et al. WSAVA guidelines for the
vaccination of dogs and cats. J Small Anim Pract 2016;57:E1–45.
3. Volk JO, Felsted KE, Thomas JG, et al. Executive summary of the Bayer
veterinary care usage study. J Am Vet Med Assoc 211;238:1275–82.
4. Casal ML, Jezyk PF, Giger U. Transfer of colostral antibodies from queens
to their kittens. Am J Vet Res 1996;57:1653–8.
5. DiGangi BA, Levy JK, Griffin B, et al. Effects of maternally-derived
antibodies on serologic responses to vaccination in kittens. J Feline
Med Surg 2012;14:118–23.
6. DiGangi BA, Levy JK, Griffin B, et al. Prevalence of serum antibody titers
against feline panleukopenia virus, feline herpesvirus 1, and feline cal-
icivirus in cats entering a Florida animal shelter. J Am Vet Med Assoc
2012;241:1320–25.
7. Jakel V, Cussler K, Hanschmann KM, et al. Vaccination against feline
panleukopenia: implications from a field study in kittens. BMC Vet Res
2012;8:62.
8. American Animal Hospital Association Canine Vaccination Task Force,
Welborn LV, DeVries JG, et al. 2011 AAHA canine vaccination guidelines.
J Am Anim Hosp Assoc 2011;47:1–42.
9. Brun A, Chappuis G, Precausta P, et al. Immunisation against panleu-
kopenia: early development of immunity. Comp Immunol Microbiol In-
fect Dis 1979;1:335–9.
10. Larson LJ, Newbury S, Schultz RD. Canine and feline vaccinations and
immunology. In: Miller L, Hurley K, eds. Infectious disease management
in animal shelters. Ames (IA): Wiley-Blackwell; 2009:61–82.
11. Larson LJ, Schultz RD. Effect of vaccination with recombinant canine
distemper virus vaccine immediately before exposure under shelter-like
conditions. Vet Ther 2006;7:113–8.
12. Srivastav A, Kass PH, McGill LD, et al. Comparative vaccine-specific and
other injectable-specific risks of injection-site sarcomas in cats. JAmVet
Med Assoc 2012;241:595–602.
13. Patel M, Carritt K, Lane J, et al. Comparative efficacy of feline leukemia
virus (FeLV) inactivated whole-virus vaccine and canarypox virus-
vectored vaccine during virulent FeLV challenge and immunosuppres-
sion. Clin Vaccine Immunol 2015;22:798–805.
14. Torres AN, O’Halloran KP, Larson LJ, et al. Feline leukemia virus im-
munity induced by whole inactivated virus vaccination. Vet Immunol
Immunopathol 2010;134:122–31.
15. Sharp NJ, Davis BJ, Guy JS, et al. Hydranencephaly and cerebellar hy-
poplasia in two kittens attributed to intrauterine parvovirus infection. J
Comp Pathol 1999;121:39–53.
16. Disease Information Fact Sheet: Feline Panleukopenia. J Feline Med Surg
2013;15: Supplementary File. This Disease Information Fact Sheet ac-
companies the 2013 AAFP Feline Vaccination Advisory Panel Report
published in J Feline Med Surg 2013;15:785–808.
17. Buonavoglia C, Marsiolio F, Tempesta M, et al. U s e of a feline panleuko-
penia modified live virus vaccine in cats in the primary-stage of feline
immunodeficiency virus infection. Zentralbl Veterinarmed B 1993;40:343–6.
18. Reubel GH, Dean GA, George JW, et al. Effects of incidental infections
and immune activation on disease progression in experimentally feline
immunodeficiency virus-infected cats. J Acquir Immune Defic Syndr
1994;7:1003–15.
19. Chalmers WS, Truyen U, Greenwood NM, et al. Efficacy of feline pan-
leucopenia vaccine to prevent infection with an isolate of CPV2b ob-
tained from a cat. Vet Microbiol 1999;69:41–5.
20. Nakamura K, Ikeda Y, Miyazawa T, et al. Characterisation of cross-
reactivity of virus neutralising antibodies induced by feline panleuko-
penia virus and canine parvoviruses. Res Vet Sci 2001;71:219–22.
21. Lappin MR. Feline panleukopenia virus, feline herpesvirus-1 and feline
calicivirus antibody responses in seronegative specific pathogen-free
kittens after parenteral administration of an inactivated FVRCP vac-
cine or a modified live FVRCP vaccine. J Feline Med Surg 2012;14:161–4.
22. Truyen U, Add ie D, Belák S, et al. Feline pan leukopeni a. ABCD
guidelines on prevention and management. J Feline Med Surg 2009;11:
538–46.
23. Huang C, Hess J, Gill M, et al. A dual-strain feline calicivirus vaccine
stimulates broader cross-neutralization antibodies than a single-strain
vaccine and lessens clinical signs in vaccinated cats when challenged
with a homologous feline calicivirus strain associated with virulent sys-
temic disease. J Feline Med Surg 2010;12:129–37.
24. Bradley A, Kinyon J, Frana T, et al. Efficacy of intranasal administration
of a modified live feline herpesvirus 1 and feline calicivirus vaccine
264 JAAHA | 56:5 Sep/Oct 2020
against disease caused by Bordetella bronchiseptica after experimental
challenge. J Vet Intern Med 2012;26:1121–5.
25. Schultz RD. A commentary on parvovirus vaccination. J Feline Med Surg
2009;11:163–4.
26. Wilson S, Greenslade J, Saunders G, et al. Difficulties in demonstrating long
term immunity in FeLV vaccinated cats due to incr easing age-related resis-
tance to infection. BMC Vet Res 2012;8:125. DOI: 10.1186/1746-6148-8-125 .
27. Jirjis FF, Davis T, Lane J, et al. Protection against feline leukemia virus
challenge for at least two years after vaccination with an inactivated feline
leukemia virus vaccine. Vet Ther 2010:11:E1–6.
28. Grosenbaugh DA, Leard T, Pardo MC, et al. Comparison of the safety
and efficacy of a recombinant feline leukemia virus (FeLV) vaccine de-
livered transdermally and an inactivated FeLV vaccine delivered subcu-
taneously. Vet Ther 2004;5:258–62.
29. Graf R, Guscetti F, Welle M, et al. Feline injection site sarcomas: data
from Switzerland 2009-2014. J Comp Pathol 2018;163:1–5.
30. Kass PH. Prevention of feline injection-site sarcomas: is there a scientific
foundation for vaccine recommendations at this time? Vet Clin North Am
Small Anim Pract 2018;48:301–6.
31. Klingborg DJ, Hustead DR, Curry-Galvin EA, et al. AVMA Council on
Biologic and Therapeutic Agents’ report on cat and dog vaccines. JAm
Vet Med Assoc 2002;221:1401–7.
32. Starr RM. Reaction rate in cats vaccinated with a new controlled-titer
feline panleukopenia rhinotracheitis-calicivirus-Chlamydia psittaci vac-
cine. Cornell Vet 1993;83:311–23.
33. Disease Information Fact Sheet: Feline Infectious Peritonitis. J Feline Med
Surg 2013;15: Supplementary File. This Disease Information Fact Sheet
accompanies the 2013 AAFP Feline Vaccination Advisory Panel Report
published in J Feline Med Surg 2013;15:785–808.
34. Little S, Levy J, Hartmann K, et al. 2020 AAFP feline retrovirus testing
and management guidelines. J Feline Med Surg 2020;22:5–30.
35. Fenimore A, Carter K, Fankhauser J, et al. Evaluation of intranasal
vaccine administration and high-dose interferon- alpha2b therapy for
treatment of chronic upper respiratory tract infections in shelter cats. J
Feline Med Surg 2016;18:603–11.
36. Helfer-Hungerbuehler AK, Spiri AM, Riond B, et al. No benefitof
therapeutic vaccination in clinically healthy cats persistently infected
with feline leukemia virus. Vaccine 2015;33:1578–85.
37. Vogt AH, Rodan I, Brown M, et al. AAFP-AAHA feline life stage
guidelines. J Am Anim Hosp Assoc 2010;46:70–85.
38. Adams CL, Kurtz S. Skills for communicating in veterinary medici ne.
Parsippany (NJ): Dewpoint Publishing; 2017:112.
39. Kass PH, Dent TH. The epidemiology of feline infectious peritonitis in
catteries. Feline Pract 1995;23:27–32.
40. Lappin MR, Andrews J, Simpson D, et al. Use of serologic tests to predict
resistance to feline herpesvirus 1, feline calicivirus, and feline par vovirus
infection in cats. J Am Vet Med Assoc 2002;220:38–42.
41. Boenzli E, Hadorn M, Hartnack S, et al. Detection of antibodies to the
feline leukemia virus (FeLV) transmembrane protein p15e: an alternative
approach for serological FeLV detection based on antibodies to p15e. J
Clin Microbiol 2014;52:2046–52.
42. Moore GE, DeSantis-Kerr AC, Guptill LF, et al. Adverse events after
vaccine administration in cats: 2560 cases (2002–2005). Vet Clin North
Am Small Anim Pract 2010;40:393–407.
43. Uhl EW, Heaton-Jones TG, Pu R, et al. FIV vaccine development and its
importance to veterinary and human medicine: FIV vaccine 2002 update
and review. Vet Immunol Immunopathol 2002;90:113–32.
44. Yamamoto JK, Sanou MP, Abbott JR, et al. Feline immunodeficiency
virus model for designing HIV/AIDS vaccines. Curr HIV Res 2010;8:14–25.
45. Westman ME, Malik R, Hall E, et al. Determining the feline immuno-
deficiency virus (FIV) status of FIV-vaccinated cats using point-of-care
antibody kits. Comp Immunol Microbiol Infect Dis 2015;42:43–52.
46. Levy JK, Crawford PC, Tucker SJ. Performance of 4 point-of-care
screening tests for feline leukemia virus and feline immunodeficiency
virus. J Vet Intern Med 2017;31:521–6.
47. DiGangi BA, Gray LK, Levy JK, et al. Detection of protective antibody
titers against feline panleukopenia virus, feline herpesvirus-1, and feline
calicivirus in shelter cats using a point-of-care ELISA. J Feline Med Surg
2011;13:912–8.
48. Mende K, Stuetzer B, Truyen U, et al. Evaluation of an in-house dot
enzyme-linked immunosorbent assay to detect antibodies against feline
panleukopenia virus. J Feline Med Surg 2014;16:805–11.
49. Gaskell R, Gettinby G, Graham S, et al. Veterinary Products Committee
working group report on feline and canine vaccination. Vet Rec 2002;150:
126–34.
50. Day MJ. Vaccine side effects: fact and fiction. Vet Micro biol 2006;117:51–8.
51. Moore GE, HogenEsch H. Adverse vaccinal events in dogs and cats.
Vet
Clin North Am Small Anim Pract 2010;40:393–407.
52. Clark N, Kushner NN, Barrett CB, et al. Efficacy and safety field trials of
a recombinant DNA vaccine against feline leukemia virus infection. JAm
Vet Med Assoc 1991;199:1433–43.
53. Richards JR, Elston TH, Ford RB, et al. The 2006 Amer ican Association
of Feline Practitioners Feline Vaccine Advisory Panel Report. JAmVet
Med Assoc 2006;229:1405–441.
54. Davis-Wurzler GM. Current vaccination strategies in puppies and kit-
tens. Vet Clin North Am Small Anim Pract 2006;36:607–40, vii.
55. Vaccine-Associated Feline Sarcoma Task Force. The current under-
standing and management of vaccine-associated sarcomas in cats. JAm
Vet Med Assoc 2005;226:1821–42.
56. Hendrick MJ, Goldschmidt MH. Do injection site reactions induce fi-
brosarcomas in cats? J Am Vet Med Assoc 1991;199:968.
57. Hendricks CG, Levy JK, Tucker SJ, et al. Tail vaccination in cats: a pilot
study. J Feline Med Surg 2014;16:275–80.
58. Shaw SC, Kent MS, Gordon IK, et al. Temporal changes in characteristics
of injection-site sarcomas in cats: 392 cases (1990–2006) J Am Vet Med
Assoc 2009;234:376–80.
59. Macy DW, Hendrick MJ. The potential role of inflammation in the de-
velopment of postvaccinal sarcomas in cats. Vet Clin North Am Small
Anim Pract 1996;26:103–7.
60. Day MD, Schoon HA, Magnol JP, et al. A kinetic study of histopatho-
logical changes in the subcutis of cats injected with nonadjuvanted and
adjuvanted multi-component vaccines. Vaccine 2007;25:4073–84.
61. Kang S, Southard T, Hume KR. DNA damage is a feature of feline
injection-site sarcoma. Vet Comp Oncol 2016;15:518–24.
62. Wilcock B, Wilcock A, Bottoms K. Feline postvaccinal sarcoma: 20 years
later. Can Vet J 2012;53:430–4.
63. Weese JS, Jack DC. Needlestick injuries in veterinar y medicine. Can Vet J
2008;49:780–4.
64. Centers for Disease Control and Prevention. CDC Vaccine Storage and
Handling Toolkit. Available at: https://www.cdc.gov/vaccines/hcp/admin/
storage/toolkit/storage-handling-toolkit.pdf. Accessed September 24, 2019.
65. Ford RB, Larson LJ, McClure KD, et al. 2017 AAHA canine vaccination
guidelines. J Am Anim Hosp Assoc 2017;53:243–51.
66. American Veterinary Medical Association. Rabies Vaccination State Law
Summary. Available at: https://www.avma.org/Advocacy/StateAndLocal/
Documents/Rabies-state-law-chart.pdf. Accessed September 24, 2019.
67. Rodan I, Sundahl E, Carney H, et al. AAHA and ISFM feline-friendly
handling guidelines. J Feline Med Surg 2011;13:364–75.
2020 AAHA/AAFP Feline Vaccination Guidelines
JAAHA.ORG 265
