
Pharma Change Control
Strategies for Successful
Company-Wide Implementation
The Executive Brieng Series
From the Editors of
Copyright © 2013 by Washington Business Information Inc. All rights reserved. The Executive Brieng Series from The Food
& Drug Letter (ISSN 0362-6466), is an in-depth analysis of regulations and issues affecting the pharmaceutical and biologics
industries. The series is published monthly, 12 issues per year, for $4,995. Photocopying or reproducing in any form, includ-
ing electronic or facsimile transmission, scanning or electronic storage is a violation of federal copyright law and is strictly
prohibited without the publisher’s express written permission. Subscribers registered with the Copyright Clearance Center
(CCC) may reproduce articles for internal use only. For more information, contact CCC at www.copyright.com or call
(978) 750-8400. For site licenses for multiple users or to purchase multiple copies, contact Nelly Valentin at (703) 538-7656.

Pharma Change Control:
Strategies for Successful Company-Wide Implementation
Table of Contents
About the Authors
Principles of Change Control .................................................................................................................3
Introduction and Operation of Change Control Programs .....................................................................6
Parallel Programs ...............................................................................................................................7
Company-Wide Programs .................................................................................................................7
Grading the Changes .........................................................................................................................8
Trials ................................................................................................................................................10
Deviations ........................................................................................................................................10
Change Control Committee .............................................................................................................10
Documentation .....................................................................................................................................12
Change Requests ..............................................................................................................................12
Appendices ...........................................................................................................................................21
A. CMS’ Change Control Management Plan Version 1.0
B. Change Management Plan Version 1.0
C. EMA Questions and Answers on Post Approval Change Management Protocols
About the Authors
Dr. Michael Hiob has worked for the State of Schleswig-Holstein (Germany) in Drug Control since
1991 as Head of the Laboratory for Drug Analysis and later as GMP Inspector for the Regional Authori-
ties for Health and Occupational Safety of the State of Schleswig Since 2006 he has been working with
the Ministry for Social Affairs, Health, Family, Youth and Senior Citizens as supervising surveillance
member of the expert group on “Qualication/Validation.” He is also active in international committees,
including as expert for the FMEA and the International Conference on Harmonization (ICH).
Thomas L. Peither is a GMP consultant for 18 years and an expert in the European GMPs. He co-
founded the GMP publishing company Maas & Peither (Germany, USA) and the midsize pharma con-
sulting company Halfmann Goetsch Peither (Switzerland, Germany, Singapore), which advises numer-
ous pharmaceutical companies.
Pharma Change Control: Strategies for Successful Company-Wide Implementation
3
Principles of Change Control
As a rule, before a company can manufacture a drug, it must rst gain approval from the appropriate
federal agency to make sure it meets quality, efcacy and safety requirements.
But in order to follow Good Manufacturing Practices (GMP), manufacturers must comply with nu-
merous requirements. Firms must document instructions for manufacture and quality control procedures.
They must specify materials needed and dene the basic conditions required for a reproducible quality,
such as suitable rooms, qualied facilities, trained personnel and type of documentation (See Figure 1).
Before a company can implement these requirements, it needs a regulatory body to review their suit-
ability for the intended purpose. In the theoretical approval model, regulatory authorities carry out the
review as part of an authorization procedure. If approved, applicants receive a notice that the product
is suitable and authorized for use. Pharmaceutical manufacturing companies must prove the suitability
of apparatus/facilities and procedures with qualication/validation. In these cases, someone responsible
must sign the qualication/validation report conrming suitability and authorization for use.
The principle that companies must adhere to suitable requirements is not only valid the rst time
a drug is manufactured or the rst time a facility follows a procedure. They must follow and adhere to
these requirements throughout the whole history of a drug or procedure.
Just as rms must document the entire batch history, they must also document requirements, such as
written specications for materials or directions for procedures. Firms must also document each change
control for the requirements.
As a result of scientic/technical development, changes to the legal basic conditions, or business re-
straints, manufacturers typically have to redene, modify, enhance, or cancel requirements again and again
in practice. In turn, this change to previously approved requirements requires a review and authorization
procedure to keep the system in its original state of proven suitability. This is the task of the change control.
Suitable rooms Qualified facilities Validated procedures Trained personnel
Specifications Written instructions Defined responsibilities Sufficient resources
Based on:
Reproduced with:
Approved
theoretical model
Prerequisites for a
guaranteed medicinal
product quality
Figure 1: Change Control Area of Consideration

Pharma Change Control: Strategies for Successful Company-Wide Implementation
4
Change control programs are considered essential elements of pharmaceutical quality assurance sys-
tems. The glossary to Annex 15 of the EU GMP Guidelines denes “change control” as:
“A formal system by which qualied representatives of appropriate disciplines review
proposed or actual changes that might affect the validated status of facilities, systems,
equipment or processes. The intent is to determine the need for action that would ensure
and document that the system is maintained in a validated state.”
Chapter 5.23 of the EU GMP Guidelines says this about the handling of changes:
“Signicant amendments to the manufacturing process, including any change in
equipment or materials, which may affect product quality and/or the reproducibility of
the process should be validated.”
There are also two brief notes in the Code of Federal Regulations (CFR) on the topic of “change
control” (21 CFR, 211.100 and 21 CFR, 211.160):
§ 211.100 Written procedures; deviations.
(a) “There shall be written procedures for production and process control designed
to assure that the drug products have the identity, strength, quality, and purity they pur-
port or are represented to possess. Such procedures shall include all requirements in this
subpart. These written procedures, including any changes, shall be drafted, reviewed,
and approved by the appropriate organizational units and reviewed and approved by the
quality control unit.”
§ 211.160 General requirements.
(a) “The establishment of any specications, standards, sampling plans, test proce-
dures, or other laboratory control mechanisms required by this subpart, including any
change in such specications, standards, sampling plans, test procedures, or other labo-
ratory control mechanisms, shall be drafted by the appropriate organizational unit and
reviewed and approved by the quality control unit. The requirements in this subpart shall
be followed and shall be documented at the time of performance. Any deviation from the
written specications, standards, sampling plans, test procedures, or other laboratory
control mechanisms shall be recorded and justied.”
In the US the quality control unit is responsible for the verication and authorization of changes.
The responsibility is not assigned in the relevant EU regulations. However, as change control is consid-
ered an essential element of the pharmaceutical quality assurance system, it makes sense to transfer the
responsibility for the function of the change control program to the person responsible for quality assur-
ance (QA representative, QA head).
Change control is not department-specic, rather the task of the whole company. This is due to the
wide area of application of change control, as described in both Annex 15 and in The Pharmaceutical
Inspection Convention and Pharmaceutical Inspection Co-operation Scheme (PIC/S) document PI 006-2.
“Written procedures should be in place to describe the actions to be taken if a change
is proposed to a starting material, product component, process equipment, process en-
vironment (or site), method of production or testing or any other change that may affect

Pharma Change Control: Strategies for Successful Company-Wide Implementation
5
product quality or reproducibility of the process. Change control procedures should en-
sure that sufcient supporting data are generated to demonstrate that the revised process
will result in a product of the desired quality, consistent with the approved specications.
“(Annex 15, no. 43)
“Change control is an important element in any Quality Assurance system. Written
procedures should be in place to describe the actions to be taken if a change is proposed
to a product component, process equipment, process environment (or site), method of
production or testing or any other change that may affect product quality or support sys-
tem operation.” (PIC/S document PI 006, section 6.7.1)
In this way, the change control monitors all types of changes which can inuence the process reli-
ability or product quality, evaluates them in reference to the relevant established requirements, and
determines the measures necessary for implementing the change or decides that a change should not be
implemented. The change control therefore ensures that a system remains in its suitable state.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
6
Introduction and Operation of Change Control
Programs
“Commitment of the company to control change to premises, supporting utilities,
materials, equipment and processes used in the manufacture of medicinal products is
essential to ensure a continued validation status of the systems concerned. This commit-
ment should be stated in the relevant company documentation. For example, the Quality
Manual, Quality Policy Documents or the Validation Master Plan. As part of its Quality
Management System the company should have a dened and formalised Change Control
Procedure.” (PIC/S document PI 006, section 2.6)
In order to successfully introduce a change control program, you must have the support of the com-
pany’s top managers. The program also needs a corresponding statement for quality management (see
Figure 2).
Many types of changes affect several regulation areas simultaneously (e.g. GMP requirements,
authorization requirements, and employment protection requirements).Quality-relevant changes can
affect several areas of a company (e.g. research/development, regulatory affairs, manufacture, quality
control, engineering, and marketing); therefore, the control must be a task for the whole company (see
Figure 3).
Change requests
and other relevant documents
Operating instruction
for change control
QM
statement
Figure 2: Structure of Change Control

Pharma Change Control: Strategies for Successful Company-Wide Implementation
7
Parallel Programs
As a rule, don’t operate different parallel and independently working interdepartment change control
programs. In the past, companies didn’t recognize the need for change control as a quality assurance task
for their entire staff. Instead separate departments handled their own change control. Some companies
are still making this mistake and work with parallel change control programs: a change control program
for the (electronic data processing) (EDP) area, a change control program for the regulatory affairs de-
partment, and a change control program for the engineering department.
The problem with this method is that different departments don’t always collaborate on change con-
trol. For example, control software was changed in the manufacturing area, without the head of produc-
tion being aware of this because he or she was not a member of the change control committee for the
EDP department that implemented this change autonomously.
Independent change control programs cause an unwanted attitude in the department, expressed as
delimitations of responsibility, ineffective inter-department communication, and unfavorable procedure-
and target-oriented attitudes. In addition, decision procedures and documentation formats/contents are
not always consistent and not well suited.
Company-Wide Programs
The central processing of change procedures, coordinated by the quality assurance department, has
several advantages compared to the handling of change control within various departments:
• There is a common understanding of what a change represents;
• The classication schemes used by the staff involved to grade changes are consistent;
• The risks connected with the change can be evaluated in a multi-disciplinary manner; and
• There is a uniform documentation and authorization procedure.
GMP requirements Regulatory requirements
Process security
(Validation)
Product quality
(Specifications, approval)
Effectiveness/safety
(Approval)
Suitable rooms and equipment
(Qualification)
Suitable personnel
(Training, experience)
Figure 3: Areas of Change

Pharma Change Control: Strategies for Successful Company-Wide Implementation
8
The main requirement for the introduction of a change control program is high-quality awareness
and an understanding for the functionality of quality assurance systems among the staff. Change control
can therefore not be assigned “from above,” and instead managers must communicate the signicance
and benets of the program as part of an intensive training. Changes that require control must be rst
recognized as such “on site” so that the company can initiate necessary procedures. Only when staff
are motivated to continuously improve the quality of the product they manufacture will they accept the
change control as a suitable tool and not reject it as bureaucratic formality.
The type and scope of a change control program must comply with the individual company requirements:
• In classical GMP-relevant areas, the change control serves to maintain the validated and speci-
ed status. Validated processes and qualied facilities can be inuenced in their state by
changes so that the drugs manufactured no longer certainly comply with specications. The
same applies for changes to material specications. A complete revalidation/requalication can
then be necessary. In these cases, companies need a formalized procedure to evaluate the risks
associated with the change, determine the measures necessary to maintain the validation status,
and authorize the change after successful revalidation.
• Holders of marketing authorizations must guarantee that for changes requiring reporting or
agreement, the necessary regulatory prerequisites are rst met. Contract manufacturers that do
not have their own authorizations and resulting obligations for reporting must guarantee that
the contract giver is informed of the company-internal changes that could have an inuence on
their application documentation. This requires that the contract giver is included in the change
control program of the contract manufacturer. Conversely, it is frequently the holder of the
authorization that causes the corresponding subsequent changes with the contract manufacturer
because he or she changed authorization. Change control is in this case not only a task that ap-
plies within the company, rather a task that applies across companies. This is valid in particular
for multi-national companies: changes can affect the approval status in different countries or
affect the utilities at different manufacturing sites.
• In areas where drugs are developed, processes are optimized, or clinical research takes place,
it’s not always necessary to comply with the strict reporting obligations linked with the approv-
al. However, persons working in this area expect their work to be comprehensible. Changes in
these areas should also be evaluated and documented in accordance with a described procedure.
During the approval procedure at the latest or during inspections before the approval (pre-ap-
proval-inspection), the development of a drug or process must be consistently proved.
Grading the Changes
According to the area of consideration (e.g. approval conformity or validation status), it may be nec-
essary to use different change procedures as a base. This is the way many companies deal with changes
to printed packaging material (information for use, folding cartons, and labels) in accordance with a
special change control procedure, because these changes occur relatively frequently in practice and the
process sequences can be standardized easily. In these cases, the sequences and the criteria used are not
independent, but are carefully matched to suit and coordinate with each other.
The examples (lists, ow charts) for grading the changes, as used in many companies, can and
should not replace the individual evaluation of a change, in particular the linked consideration of risk.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
9
The example in Figure 4 only shows one way to grade changes. Some companies use different and ex-
tensive grading categories. Others have extensive lists of possible changes and their grades. The display
of change procedures contains large ow charts, which are often complicated and hard to understand.
These companies have previously tried to show all potential types of changes and dene the required
sequences in diagrams. Such a procedure is not incorrect because it shows the scope of consideration of
the change control and example help in the introduction phase for evaluation and development of chang-
es. This can however deceive one into thinking everything is regulated. After the introduction phase,
most companies nd that changes do not always t into a prefabricated chart and instead it’s important
to rely on experience and know-how for particular cases.
Other classications not included in Figure 4 are possible. It is not decisive which and how many
change classes a company has determined, but how it is guaranteed that changes requiring control are
recognized as such and implemented according to a dened procedure.
See the PIC/S document PI 006 for notes on grading changes. In chapter 6.7.4., there is a list of
changes that may make a revalidation necessary, including:
• Physical characteristics of the raw material
Figure 4: Grading of Changes
Changes requiring control
Not requiring
control
Major
(major change)
Minor
(minor change)
Signicance
of change
Inuences product quality
or process reliability.
Inuences a unit
requiring control.
No relevance to GMP or
authorization
Possible
measures
(selection)
• Ofcial license
• New approval
• Revalidation
• Amendment
• Review
• Documentation
• No relevance to GMP or
authorization
Examples
• Change of manufac-
turer: other synthesis
route of a starting ma-
terial (other impurities)
• Removal of processes
to another site
• Change in the product
composition
• Change to the process
parameters
• Replacement of
apparatus part of
the same design
• Change of
cleansing agent
for oors
• Change of laun-
dry for work
clothing (non-
sterile or antibiot-
ics area)
• Introduction of
co-sales right
• Change to working times
• Renovations in adminis-
tration area
• Installation of air condi-
tioner in staff room
• Introduction of electron-
ically-readable plant ID
cards

Pharma Change Control: Strategies for Successful Company-Wide Implementation
10
• Origin of the starting material (change of supplier)
• Packaging material (e.g. replacement of plastic with glass)
• Process changes (e.g. mixing times, drying temperatures)
• Equipment (e.g. introduction of an automatic detection system)
• Production area and supply systems (e.g. new water plant)
• Moving manufacturing into a new building
Trials
The so called “trials” cause a problem area in change control. Trials are preliminary, temporary
changes which can be permanently established or revoked after a trial period. With trials, there is a risk
that these intended temporary changes gradually become permanent changes, without a formal change
control procedure being carried out. Regardless of how long a trial is retained and whether it is with-
drawn after a trial phase or introduced permanently, trials should be dealt with according to the same
procedure as all other changes. Before the trial phase starts, the company should implement an analo-
gous change control procedure.
Deviations
Deviations should not be treated as changes, not even when deviations become changes after the
company claries the failure. A deviation is an unplanned and undesirable variation from a requirement.
It does not correspond with the aim and procedure of change control and should be dealt with according
to a special procedure about handling deviations.
Change Control Committee
An important function as part of the change control program is fullled by the change control com-
mittee (also known as: change control team, change control panel). This permanent committee generally
consists of the head of quality assurance, who frequently also chairs and the heads of manufacturing,
quality control, sales, regulatory affairs and the information representative. If necessary, further depart-
ments (e.g. research/development, EDP, and engineering) also become involved. The task of the com-
mittee is to evaluate changes, determine the measures required, coordinate measures for the departments
affected by the change, and provide nal authorization.
A major problem, especially during the introduction phase of the change control system, is the issue of
which changes the change control committee should deal with rst. It is obvious that this committee can-
not deal with all changes in a company due to capacity reasons. As a matter of fact, the committee should
process only changes requiring control. These are changes relevant for the regulatory status and involve
reporting or authorization procedures. They are also changes that could have an inuence on the attributes
of a GMP-relevant system, facility, apparatus, material/product, or a procedure/process. This inuence may
be critical as it regards the product quality/process reliability and may require a revalidation/requalication
or the compilation/update of documentation. The change control committee is also involved in changes for
which their implementation is often extensive and coordinating measures are necessary. The committee
should also deal with all changes whose grade or implementation is unclear or questionable.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
11
The question of how the committee members communicate with each other is signicant. Not all
change control procedures urgently require the convening of a meeting, at which many important func-
tion heads must often be present in person. For cases that will involve an easy decision, it is also worth
considering traditional paper-based circulation procedures (serial or parallel), e-mail agreements, or
common access to Intranet-based forms.
The committee can only deal with change applications if they are actually assigned. A high involve-
ment is transferred to the other staff in the company. If no one evaluates a critical change and fails to
introduce a formalized change control procedure, this can cause severe problems with quality.
When a company has introduced a change control program, the committee can review the effective-
ness of the system using data, which is easy to determine. It includes:
• Number of completed change procedures/year
• Number of prematurely terminated procedures of procedure deviations
• Work expenditure/change type
• Duration of procedure: from application to completion
• Number of grading problems/total number of change procedures
• Number of OOS results/year
• Number of internal or external complaints/year
• Stability problems/batch reviews/recalls
It can measure the functionality of the system by taking into account the effort and speed of change
procedures, for example, or by examining the deviations. The effectiveness of a change control program
depends on the knowledge and experiences of the staff involved. The regular training about change con-
trol procedures is therefore extremely important. Committees should structure documentation procedures
and communication sequences as simply as possible to enable rapid implementation.
Deviations are unplanned and generally unwanted changes. They are expressed in the form of inter-
nal and external complaints, stability problems, or batch recalls. If they occur, this can be an indication
that the change control program has failed. It is possible that the change to a critical process parameter
or material specication has been overlooked or a negative trend in the development of process data has
not been noticed in time.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
12
Documentation
“All changes should be formally requested, documented and accepted by representa-
tives of Production, QC/QA, R&D, Engineering and Regulatory Affairs as appropriate.
The likely impact (risk assessment) of the change on the product should be evaluated and
the need for, and the extent of Re-validation discussed. The change control system should
ensure that all notied or requested changes are satisfactorily investigated, documented
and authorised.” (PIC/S document PI 006, section 6.7.2)
“All changes that may affect product quality or reproducibility of the process should
be formally requested, documented and accepted. The likely impact of the change of facil-
ities, systems and equipment on the product should be evaluated, including risk analysis.
The need for, and the extent of, re-qualication and re-validation should be determined.”
(Annex 15, no. 44)
Change control requires a written procedure (change control program) to regulate at least the follow-
ing points:
• What types of changes does change control take into account; for which areas does this operat-
ing instruction apply?
• Who can suggest/initiate changes?
• How are changes requested (forms, methods of communication)?
• How are changes graded, who is responsible for the grading?
• How are the measures necessary for carrying out the change determined; who compiles the
directions required?
• Who is responsible for the execution and monitoring of all necessary measures?
• How is the change control committee assembled; what are the duties of the committee?
• How is the change documented (format, content, storage)?
• Who is responsible for authorizing changes?
• What are the special regulations for urgent changes?
If the change affects a manufacturing or testing process, then the qualied person must take this into
consideration when releasing the batch (cf. PIC/S document PI 006, chapter 6.7.3).
He or she should document all quality-relevant changes in full to ensure that they are comprehen-
sible. The records can be archived in paper form or electronically. Keep in mind that when storing these
records, you need to have raw data and other relevant documents accessible.
Change Requests
Changes requiring control are generally documented in the form of a change request, in which the ap-
plicant for the change proposes the type of grade/evaluation of the change, species the time frames and

Pharma Change Control: Strategies for Successful Company-Wide Implementation
13
measures for carrying out the change, and requests that and the change is authorized or declined by the
change control committee. The documentation for the change procedure should prove that the change was
evaluated (risk analysis) and the subsequently dened measures were implemented as predetermined. When
implementing the change, you will need to coordinate several measures regarding timing and contents. A
clear display of the individual measures for a project plan is useful for the coordination of complex changes.
The “change control” operating instruction describes the procedure of change control and contains
an application form for documenting the change procedure. See the example below.
[Company] Change control procedures
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
File name/path:
[Enter le name/path]
Area of application:
Changes to materials, products, facilities/apparatus, processes/procedures and systems in the areas of pur-
chasing, manufacture, quality control, engineering, research/development, marketing authorization, sales,
EDP, contract manufacturing and external testing places, excluding changes to printed packaging materials
Key words:
Change, procedure
Replaces version: from:
[Enter version no.] [Enter date]
Changes made since last version:
[Enter main content of changes]
Cross references:
[Valid documents: Enter document no.]
References:
EU GMP Guideline, Annex 15, PIC/S document PI-006-2, Appendix I of the regulation (EC) no. 1084/2003
Distribution list:
[Enter recipients of document]
Compiled by:
[Name/signature of person
who compiled it]
on:
[Date of compilation]
Checked by:
[Name/signature of person
carrying out check]
on:
[Date of check]
Approved by:
[Name/signature of person giving au-
thorization]
on:
[Date
of approval]
Sample Change Control Document

Pharma Change Control: Strategies for Successful Company-Wide Implementation
14
[Company] Change control procedures
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
1. Purpose of the instruction:
Internal requirements are specied in order to comply with the legal drug product provisions and the
GMP Guideline, as well as to ensure that the quality of the medicinal product complies with the ap-
proval and is reproducible. These requirements cover the entire manufacturing process and the quality
control for a medicinal product, including the materials and facilities/apparatus used. The purpose of
this instruction is to ensure that the quality of the medicinal product, the safety of facilities/apparatus,
the safety of procedures/process and conformity with the applicable application les for marketing
authorization are maintained in the event of changes to these requirements.
2. Denitions/abbreviations:
Deviations: unplanned and undesirable deviation from a requirement
CCC: change control committee
Change: planned deviation (extension, replacement, removal, addition) as part of a requirement
Change control: system with which qualied representatives from corresponding departments evalu-
ate current or planned changes in terms of their effects with regard to a specic status. The aim is to
establish precautions that are necessary to prove and document compliance with the specic status.
Facility: total of all apparatus linked together with a common purpose.
Applicant: person who is initiating a change with a change request
Apparatus: object characterized by the technical processes carried out in it.
Minor change: change which fullls the conditions of Appendix I of Regulation (EC) no. 1084/2003
or which affects the attributes of a system, facility, apparatus, material/product or procedure/process.
Impairment of the product quality/process reliability is not likely. Minor changes may require notica-
tion to the regulatory or supervisory authorities.
Major change: change which cannot be classied as a minor change or which may affect the critical
attributes of a system, facility, apparatus, material/product or procedure/process. Impairment of the
product quality/process reliability is likely. Major changes may require authorization by the relevant
regulatory or supervisory authorities and/or prior revalidation or requalication.
Process: set of interrelated methods and activities which convert an input into results.
System: total of all facilities linked together with a common purpose.
Procedure: established way of carrying out an activity.
Trial: preliminary, temporary changes which are permanently established or revoked after a trial period

Pharma Change Control: Strategies for Successful Company-Wide Implementation
15
[Company] Change control procedures
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
3. Responsibilities:
3.1. Responsibilities for the established procedures
3.1.1. The legal medicinal product responsibility for proper planning, implementation and authoriza-
tion of changes is borne by the head of production, the head of quality control, the sales manager and
the information representatives for their relevant area. In particular, they must ensure that:
the qualication status of the rooms and facilities that are affected by a change is maintained or that a
requalication is implemented
the validation status of the processes/procedures that are affected by a change is maintained or that a
revalidation is implemented
changes to manufacturing, analysis and labeling of a medicinal product are based on a valid approval/
registration
the documentation required for the change is compiled or updated
3.1.2. The organizational processing and documentation of change control procedures is conferred
to the change control committee (CCC). The CCC comprises those in the roles mentioned above and
the head of regulatory affairs and the QA representative. Other heads of department or experts may be
called in at the wish of one of the members. The committee has the following tasks:
Risk evaluation of the change request
Authorization or rejection of the application
Establishing and scheduling necessary measures
3.1.3. The chairman of the CCC is the QA representative. He has the following tasks:
Calling the CCC meetings and taking minutes
Coordination of circulation procedure
Maintenance of the database of change control procedures
Formal control of the change requests
Monitoring compliance with deadlines
Archiving the completed change requests
3.2. Responsibility for the revision of this instruction
The QA representative is responsible for checking this instruction regularly to make sure it is up to
date and for revising it if necessary. He must release a new version at least every two years.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
16
[Company] Change control procedures
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
4. Procedure
4.1. Basic principles
4.1.1. GMP or approval-relevant changes must only be implemented if they have been previously
requested in writing and authorized.
4.1.2. Trials are also subject to this procedure.
4.1.3. Deviations are not subject to this procedure, but to operating instruction [enter doc. no. /ver-
sion no.] “Handling deviations”.
4.1.4. Changes can be requested by any staff member using the form in Appendix 1.
4.1.5. Changes of which the grade is debatable or unclear must also be requested using this procedure.
4.2. Implementation of change control procedures
4.2.1. Forms for change control procedure are issued by the QA department. If a new form is issued,
a change number is automatically allocated to the change procedure by the change database and is
entered on the form.
4.2.2. The applicant should specify the object of the change and the signicant reasons and circum-
stances of the change in no. 1 on the change request. He should then sign and date it.
4.2.3. The heads of area affected by the change should have the opportunity to give their opinion on
the intended change, to identify any risks and to suggest necessary measures and schedules. In any
case, they should be acquainted with the planned change. This is documented in no. 2 on the change
request.
4.2.4. The QA representative transfers the data required to identify the procedure into the change
control database.
4.2.5. The members of the CCC jointly carry out a risk analysis of the change request under no. 3.1,
classify the change under no. 3.2 and give their decision to authorize or reject the change application
under no. 3.3. Authorization may be associated with a time scale. The applicant and his head of area
are informed of the decision via a copy of the completed change request.
4.2.6. A decision by the change control committee in accordance with 4.2.5 may only be made if all
the information and documents relevant to the decision are submitted. If necessary, the request may be
returned to the applicant for completion.
4.2.7. If the change is authorized, the CCC can establish a measures plan in no. 3.4 of the change
request (tasks, responsibilities, schedule). This must be completed and authorized for the change pro-
cedure to be completed.
4.2.8. Those responsible in accordance with the measures plan shall each receive a copy of section
3.4 and shall inform the QA representative when the established measures have been completed.

Pharma Change Control: Strategies for Successful Company-Wide Implementation
17
[Company] Change control procedures
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
4.2.9. The QA representative shall collect all the completed measures plans, check them for
successful completion of each task and include them with the original change request. The completed
change request is archived and the result and date of the completion is entered in the change database.
4.2.10. In simple cases, in which the applicant can implement the change himself without
requiring any scheduling, an electronic copy of the request may be distributed to the CCC via the in-
ternal e-mail system instead of at a meeting (circulation procedure). Each CCC member then gives his
written vote regarding the change request via e-mail with electronic signature. The change request and
electronic voting from the CCC member are to be archived in accordance with chapter 4.3. If there is
no unanimous decision, the QA representative calls a meeting about the request.
4.3. Documentation
4.3.1. The procedure in accordance with chapter 4.2 must be documented on the “change request”
form in Appendix 1.
4.3.2. If necessary, documents relevant to the decision should be added to the change request.
4.3.3. The change request and documents relevant to the decision must be kept indenitely in the
“quality assurance” area of the department.
4.3.4. If necessary for capacity reasons, the paper copy of the change request and its associated docu-
ments may be replaced with an electronic archive le.
4.4. Deviation from the procedure
4.4.1. It is permissible to deviate from the regulations in chapter 4.2 only if:
an immediate change is urgently required for operational or staff safety or
an immediate change has considerable signicance for the unit, the need for it was not foreseeable and
it was not possible to comply with the formal procedure in chapter 4.2 in the time available.
4.4.2. In the cases in chapter 4.4.1, the consent of the responsible head of area or his representative
should be sought before the change is implemented.
4.4.3. Once the change has been implemented, the procedure in chapter 4.2 must be followed at the
earliest opportunity.
5. Appendices
Appendix 1: change request form

Pharma Change Control: Strategies for Successful Company-Wide Implementation
18
[Company] Change control procedure
Appendix 1
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
Company
Change request
[Cross reference to the operating instruction on which
it is based]
Page/pages
[enter page no.]
Change no.:
(to be entered by QA)
Change designation:
(to be entered by QA)
1. Applicant
Name … Department …
1.1. Object of the change (mark with a cross)
• Rooms
• Manufacturing facilities
• Quality control facilities
• Media supply
• Computer-assisted system
• Organization
• Contract acceptor/giver
• Supplier
• Marketing authorization
• Documentation
• Manufacturing procedure
• Test procedure
• Cleaning procedure
• Other procedure
• Starting materials
• Packaging material
• Semi-manufactured/bulk product
• Finished medicinal product
• Other material
• …
1.2. Description of change:
• Cause, reason for the change
• Products, procedures, facilities affected
• Necessary time frames
• Suggestion for implementing the change
• Estimated costs
• Alternatives
• Cross reference to ap-
pendices where necessary.
Date and signature of the
applicant: ...............................
2. Area head
Response/risk assessment of the affected areas
Area:
Date and signature of the person responsible:
Area:
Date and signature of the person responsible:
Area:
Date and signature of the person responsible:

Pharma Change Control: Strategies for Successful Company-Wide Implementation
19
[Company] Change control procedure
Appendix 1
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
3. Change control committee:
3.1. Additional risk assessments:
3.2. Grade of change
o Major change
o Minor change
o No major or minor change
3.3. Decision:
o The change is authorized. Time limit for the implementation:
o The measures list in no. 3.4 must be observed.
o The change is not authorized. Rationale:
Date and signatures of the CCC members
Head of Production:
Head of Quality Control:
Sales manager:
Information representative:
Head of regulatory affairs:
QA representative:
Other members:

Pharma Change Control: Strategies for Successful Company-Wide Implementation
20
[Company] Change control procedure
Appendix 1
Page/pages x of y
Document number:
[Enter document no.]
Version:
[Enter version no.]
valid from: [Enter date]
valid to: [Enter date]
3.4. Measures list
Directions for implementing and document-
ing the measure
Cross references to affected documents that
require revision
Extensive directions or project planning may
be recorded in separate documents
Responsible …
Time limit …
Completed on …
Signature …
4. QA head
Completion of procedure
• The procedure has been completed correctly.
• The procedure has not been completed correctly. It should be abandoned and not continued.
Rationale:
• The procedure has not been completed correctly. Deviations occurred and measures required
for procedure completion:
Date and signature of the QA head....................................................
Appendix A: CMS’ Change Control Management
Plan Version 1.0

Appendic A.docx Page 1 of 13 February 12, 2013
Change Control Management Plan
Version 1.0

Appendic A.docx Page 2 of 13 February 12, 2013
Revision History
DATE
REV
AUTHOR
DESCRIPTION
MM/DD/YY
1

Appendic A.docx Page 3 of 13 February 12, 2013
Table of Contents
1. Purpose ............................................................................................................4
2. Scope...............................................................................................................4
3. Definitions ........................................................................................................4
4. Roles and Responsibilities ...................................................................................5
5. Process.............................................................................................................5
Appendix A: Attributes Stored for Each Issue............................................................... 10
Appendix B: Change Control Board Charter Template ................................................... 12
List of Tables
Table 1: Roles and Responsibilities ..............................................................................5
Table 2: Process Summary ..........................................................................................6
Table 3: Change Management Process Steps .................................................................8
List of Figures
Figure 1: Change Management Process Flowchart ..........................................................7

Appendic A.docx Page 4 of 13 February 12, 2013
1. Purpose
This document describes the process to request and manage changes to work products created or
maintained by the ICD-10 project team members. These changes may apply to any area
impacted by ICD-10, including policies, processes, and systems. This process will allow for the
following:
• Facilitate communication regarding requested changes among the stakeholders of the
project team;
• Provide a common process for resolving requested changes and reported problems;
and
• Reduce the uncertainty around the existence, state, and outcome of a change that has
been requested in a work product.
2. Scope
Any stakeholder of the ICD-10 project can submit the following types of change requests for
consideration:
• Requests for scope, schedule, or resource changes that may affect program
administration;
• Requests for requirements changes (additions, deletions, modifications, deferrals) in
software currently under development;
• Reports of problems in current production or test environments;
• Requests for enhancements in current production systems; and
• Requests for new development projects.
This change control process applies to baselined deliverables/work products created or managed
by the members of the ICD-10 project team, including:
• Software that has been released to production or is in beta test;
• Requirements specifications for MMIS or other systems impacted by ICD-10;
• Group procedures and processes; and
• User and technical documentation.
The following work product classes are exempted from this change control process:
• Work products that are still under development, except for requirements changes
requested in new projects;
• Interim or temporary work products created during the course of a project; and
• Any work products intended for individual use only.
3. Definitions
Change Request (CR): An item submitted by a stakeholder for consideration through the change
control process.
Stakeholder: Someone who is affected by or who can influence the project.

Appendic A.docx Page 5 of 13 February 12, 2013
4. Roles and Responsibilities
All ICD-10 project team members play a role in change management and should understand the
impacts and opportunities of ICD-10 on their business areas (see Table 1). In addition, the
project manager, or designee, should include consideration of Change Control Board (CCB)
inputs and outputs on the ICD-10 project scope, cost, and schedule.
Table 1: Roles and Responsibilities
Role
Activity
ICD-10 Steering
Committee
The group that charters the CCB and approves or rejects ICD-10 project
changes where either CCB consensus is not reached or program
administration may be affected (e.g., scope, schedule, or costs).
CCB
The group that approves or rejects proposed changes for the project.
CCB Chair
Chairperson of the CCB; has decision-making authority as granted by
charter (e.g., may have final decision-making authority if the CCB does
not reach agreement); directs someone to be the Evaluator for each CR;
and directs someone to be the Modifier for each approved CR.
Evaluator
The person whom the CCB Chair asks to analyze the impact of a
proposed change.
Modifier
The person who is assigned responsibility for making changes in a work
product in response to an approved CR; updates the status of the request
over time.
Originator
The person who submits a new change request.
Project Manager
The person who is responsible for overall planning and tracking of the
development project activities.
Verifier
The person who determines whether a change was made correctly.
5. Process
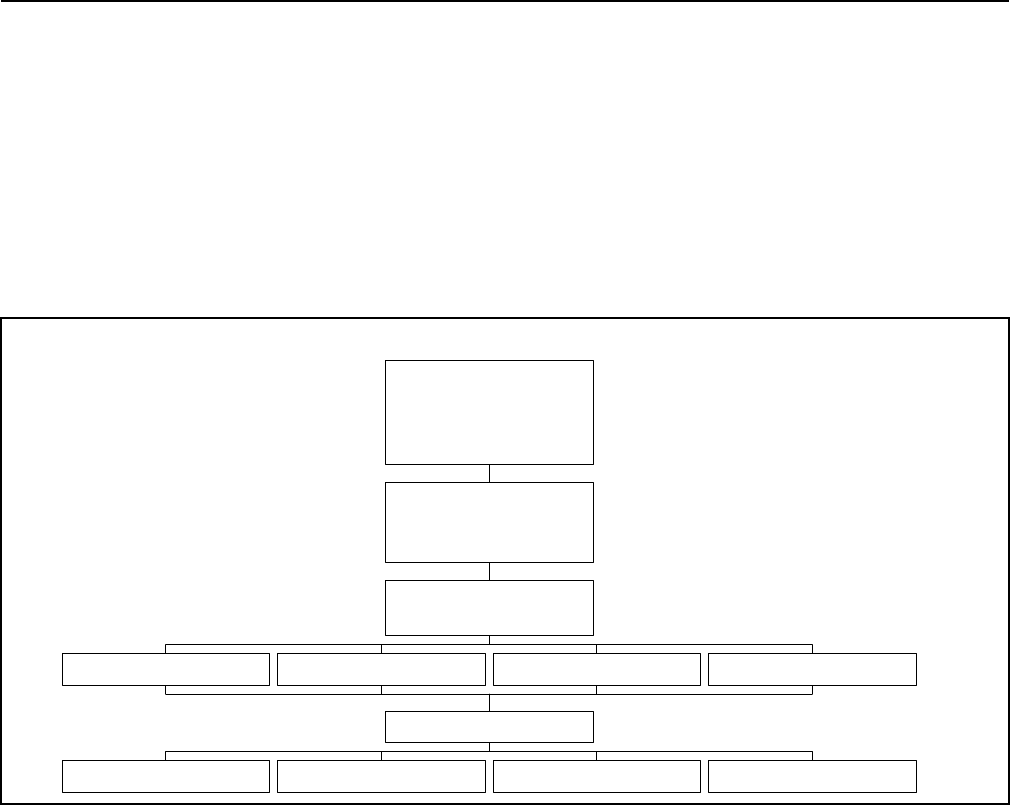
Outlined in Table 2 and Figure 1, the change management process begins when a stakeholder, at
any ICD-10 program level, submits a change request for consideration by the CCB.
Appendic A.docx Page 6 of 13 February 12, 2013
Table 2: Process Summary
Inputs
Process
Outputs
• Charters (e.g., Program,
Project, and CCB)
• Baselined work products
• Project Plan
• Risk Assessment Checklist
• Business/User
Requirements
• Schedule/WBS
• Valid CR submitted on
standard form
Regularly scheduled CCB
meetings to Evaluate CRs;
Modify policies, processes, and
systems; and Verify any
approved changes were made
correctly. [See Figure 1 and
Table 3]
• Change Management Log
reflecting status of issue
is either ‘Rejected’ or
‘Closed’
• Modified work products
• Updated requirements,
including traceability
information
• Status Reports
Note: The Change Management Log is managed at the Workgroup or Project Level.

Appendic A.docx Page 7 of 13 February 12, 2013
Submitted
Evaluated
Approved
Change
Made
Canceled
Verified
Closed
Originator
Submitted CR
Evaluator Performed
Impact Analysis
Rejected
CCB Decided
not to make
the change
CCB Decided to
Make the
Change
Change was
canceled,
back out of
modifications
Modifier has made
the change and
requested verification
Verification
Failed
Change Canceled,
back out of modifications
Verifier has confirmed
the change
No Verification Required,
Modifier has installed
refined work products
Change Canceled,
back out of modifications
Modifier has installed
refined work products
Figure 1: Change Management Process Flowchart

Appendic A.docx Page 8 of 13 February 12, 2013
Table 3: Change Management Process Steps
Role
Action
Originator
1. Documents CR using standard change request form
2. Stakeholder consults with project team lead (Workgroup/Project Level)
about CR
3. Stakeholder and/or project team lead submit CR to CCB
CCB
4. Sets initial CR status to ‘Submitted’
5. Assigns CR to Evaluator
Evaluator
6. Assesses the CR as to feasibility, whether it really pertains to the indicated
project, whether a reported problem can be reproduced, an estimate of the
labor hours needed to implement the change, and other criteria as decided
upon by CCB charter.
7. Change CR status to ‘Evaluated’
CCB
8. The CCB decides whether the requested change should be made (or the
reported problem fixed) at this time, at some point in the future, or not at all.
Input should be solicited from others potentially affected by the change
before making the decision.
9. If the change was accepted, the CCB Chair assigns a Modifier, sets the
status to ‘Approved,’ enters any explanation in the Response attribute, and
schedules the work. The Project Manager negotiates any necessary changes
in project commitments with affected stakeholders. Communication is sent
to the assigned Modifier and the Originator.
10. If the change was rejected, the CCB Chair sets the status to ‘Rejected’ and
enters an explanation of why in the Response attribute. Communication is
sent to the Originator and CCB.
11. The CCB Chair and the Originator determine whether formal verification of
the change will be required, following the procedure in the Verification
section. If so, they select the verification method to be used and the CCB
Chair assigns a Verifier.
Modifier
12. Makes the necessary changes in the affected work products and notifies any
other affected parties if corresponding changes need to be made, such as
user documentation, help screens, and tests.
Workgroup
Lead
Project
Manager
13. Updates the project plans, task lists, and schedules to reflect the impact of
the change on project work remaining to be done. The Project Manager
revises any task dependencies as necessary.

Appendic A.docx Page 9 of 13 February 12, 2013
Role
Action
Modifier
14. If it becomes apparent during the work that the requested change is not
feasible after all, the Modifier notifies the CCB Chair, who may then set the
status to ‘Canceled.’ The Modifier backs out of any modifications made,
restoring the work products to their previous baseline. Communication is
sent to the Originator, CCB Chair, Modifier, and Project Manager.
15. When the change is completed, the Modifier sets the status to ‘Change
Made,’ updates the issue in the database with appropriate notes in the
Response attribute, and enters the hours of effort that were required to make
the change in the Actual Hours attribute. Communication is sent to the
Originator and CCB Chair.
16. Notifies the Originator and Verifier (if one was assigned) that the change
has been made and makes all modified work products available to the
people responsible for verification.
Verifier
17. Performs the agreed-upon verification steps.
18. If verification is successful, the Verifier sets the status to ‘Verified.’
Communication is sent to the Originator and Modifier.
19. If verification is not successful, the Verifier sets the status back to
‘Approved’ and describes the problem in the Response attribute.
Communication is sent to the Originator and Modifier. The procedure
continues back at step 12.
Modifier
20. For a problem report CR or an enhancement request CR, the Modifier
installs the modified work product as appropriate and updates the product
baseline. For requirements changes, the Modifier updates version numbers
on all modified work products per the project’s version control procedure,
checks them back into the version control system, updates requirements
traceability information, and requirements status attributes as necessary, and
updates the requirements baseline.
21. Sets the status to ‘Closed.’ Communication is sent to the Originator and
CCB Chair.
Appendic A.docx Page 10 of 13 February 12, 2013
Appendix A: Attributes Stored for Each Issue
Field
How Set
Contents
Actual Hours
Modifier
Actual labor hours of effort needed to implement the change.
Description
Originator
Free-form text description of the change being requested. This
cannot be changed after it is entered. If reporting a problem,
enter the exact error message text observed here.
Date Submitted
System
Date this CR was submitted to the CCB.
Date Updated
System
Date this CR was most recently updated.
Estimated Hours
Modifier
Estimated labor hours of effort needed to implement the change.
Implementation
Priority
CCB Chair
Relative importance of making the change: Low (default),
Medium, High.
CR ID
System
Sequence number assigned to the CR.
CR Type
Originator
Type of change request: Scope, Schedule, Resources, Problem,
Enhancement, Requirement Change, New Project.
Modifier
CCB Chair
Person who is assigned responsibility for implementing change.
Originator
Originator
Originator’s name.
Originator E-
Mail
Originator
Originator’s e-mail address.
Originator
Phone
Originator
Originator’s phone number.
Originator
Priority
Originator
Originator’s relative importance of change: Low, Medium,
High.
Planned Release
CCB Chair
Product release number for which this approved change is
scheduled, determined by CCB.
Product
Originator
Name of the product or project in which a change is being
requested or a problem reported.

Appendic A.docx Page 11 of 13 February 12, 2013
Field
How Set
Contents
Problem
Severity
Originator
For a problem report, set severity of the change (see below).
Minor - Cosmetic problem, usability improvement, unclear error
messages; customer can live with the problem (default)
Major - Problem adversely affects product functioning, but a
workaround is available; customer will be annoyed; serious
usability impairment; problem blocks some testing
Critical - Product does not function at all or crashes; wrong
results are generated; further testing of application is not
possible
Emergency/Escalated - Anything that requires a change to be
made immediately, bypassing the change control process
temporarily for executive sponsor review
Response
CCB
Chair,
Modifier
Free-form text log of responses made to the change request.
Status
Originator,
Modifier
Update current status of the change request as it moves through
the states described in the Change Request Status section.
Title
Originator
One-line description of the CR.
Verifier
CCB Chair
Name of individual who is responsible for verifying that changes
were made correctly.

Appendic A.docx Page 12 of 13 February 12, 2013
Appendix B: Change Control Board Charter Template
Purpose
<Describe the objectives of the CCB. This section may read: “The Change Control Board
(CCB) represents the interests of program and project management by ensuring that a structured
process is used to consider proposed changes and incorporate them into a specified release of a
product. The CCB shall request that impact analysis of proposed changes be performed, review
change requests, make decisions, and communicate decisions made to affected groups and
individuals.” Define the relationship of this CCB to any other CCBs in the organization or other
decision-making bodies, such as a project steering committee.>
Scope of Authority
<Indicate the scope of decisions that the CCB makes. This scope could be over a specific
organizational range; a project, group of projects (program), or subproject; a maximum budget
or schedule impact. This scope boundary separates decisions that this CCB can make from those
that it must escalate to a higher-level CCB or manager for resolution.>
Membership
<List the members of this CCB. The CCB typically includes representatives from program
management, project management, software engineering, hardware engineering, testing,
documentation, customer support, and marketing. One individual is designated as the CCB
Chair. Keep the CCB as small as possible, to facilitate its ability to make rapid decisions, but
make sure that the critical perspectives are represented. Indicate who should be responsible for
escalated changes (e.g., scope, schedule, and resources).>
Operating Procedures
<State the frequency of regularly scheduled CCB meetings and the conditions that will trigger a
special meeting. Describe how meetings will be conducted, the number of CCB members who
constitute a quorum to make decisions at a meeting, and the roles that must be represented for
the meeting to proceed. Identify whether guest participants may attend, such as the individuals
who proposed the change requests being considered at a specific meeting.>
Decision-Making Process
<Describe how the CCB will make its decisions. Indicate whether voting, consensus, unanimity,
delegation to a specific individual, or some other decision rule is used to make decisions. State
whether the CCB Chair or another manager is permitted to overrule the CCB’s collective
decision.>
Communicating Status
<Describe how each decision that the CCB makes will be communicated to the individual who
requested the change, senior management, project management, affected team members who

Appendic A.docx Page 13 of 13 February 12, 2013
must implement the change, higher- or lower-level CCBs, and any other stakeholders. Indicate
where the decisions and any supporting information, rationale, or data will be stored.>
Appendix B: Change Management Plan Version
1.0

[Insert appropriate disclaimer(s)]
<PROJECT NAME>
CHANGE MANAGEMENT PLAN
Version Number: 1.0
Version Date: <mm/dd/yyyy>

<Project Name>
Change Management Plan (v1.0) Page 1 of 9
[Insert appropriate disclaimer(s)]
Notes to the Author
[This document is a template of a Change Management Plan document for a project. The template
includes instructions to the author, boilerplate text, and fields that should be replaced with the values
specific to the project.
• Blue italicized text enclosed in square brackets ([text]) provides instructions to the document
author, or describes the intent, assumptions and context for content included in this document.
• Blue italicized text enclosed in angle brackets (<text>) indicates a field that should be replaced
with information specific to a particular project.
• Text and tables in black are provided as boilerplate examples of wording and formats that may be
used or modified as appropriate to a specific project. These are offered only as suggestions to
assist in developing project documents; they are not mandatory formats.
When using this template, the following steps are recommended:
1. Replace all text enclosed in angle brackets (e.g., <Project Name>) with the correct field document
values. These angle brackets appear in both the body of the document and in headers and
footers. To customize fields in Microsoft Word (which display a gray background when selected)
select File->Properties->Summary and fill in the appropriate fields within the Summary and
Custom tabs.
After clicking OK to close the dialog box, update all fields throughout the document selecting
Edit>Select All (or Ctrl-A) and pressing F9. Or you can update each field individually by clicking
on it and pressing F9.
These actions must be done separately for any fields contained with the document’s Header and
Footer.
2. Modify boilerplate text as appropriate for the specific project.
3. To add any new sections to the document, ensure that the appropriate header and body text
styles are maintained. Styles used for the Section Headings are Heading 1, Heading 2 and
Heading 3. Style used for boilerplate text is Body Text.
4. To update the Table of Contents, right-click on it and select “Update field” and choose the option -
“Update entire table”.
5. Before submission of the first draft of this document, delete this instruction section “Notes to the
Author” and all instructions to the author throughout the entire document.

<Project Name>
Change Management Plan (v1.0) Page 2 of 9
[Insert appropriate disclaimer(s)]
VERSION HISTORY
[Provide information on how the development and distribution of the Change
Management Plan will be controlled and tracked. Use the table below to provide the
version number, the author implementing the version, the date of the version, the name
of the person approving the version, the date that particular version was approved, and
a brief description of the reason for creating the revised version.]
Version
Number
Implemented
By
Revision
Date
Approved
By
Approval
Date
Description of
Change
1.0
<Author name>
<mm/dd/yy>
<name>
<mm/dd/yy>
<description of change>

<Project Name>
Change Management Plan (v1.0) Page 3 of 9
[Insert appropriate disclaimer(s)]
TABLE OF CONTENTS
1 INTRODUCTION.................................................................................................................... 4
1.1 Purpose of The Change Management Plan....................................................... 4
2 CHANGE MANAGEMENT PROCESS ............................................................................... 4
2.1 Change Request Process Flow Requirements.................................................. 4
2.2 Change Request Form and Change Management Log ................................... 5
2.3 Evaluating and Authorizing Change Requests .................................................. 5
2.3.1 Change Control Board...................................................................................... 6
3 RESPONSIBILITIES .............................................................................................................. 6
APPENDIX A: CHANGE MANAGEMENT PLAN APPROVAL............................................7
APPENDIX B: REFERENCES ...................................................................................................8
APPENDIX C: KEY TERMS .......................................................................................................9
<Project Name>
Change Management Plan (v1.0) Page 4 of 9
[Insert appropriate disclaimer(s)]
1 INTRODUCTION
1.1 PURPOSE OF THE CHANGE MANAGEMENT PLAN
[Provide the purpose of the Change Management Plan. This document should be
tailored to fit the particular project needs.]
The Change Management Plan documents and tacks the necessary information
required to effectively manage project change from project inception to delivery.
The Change Management Plan is created during the Planning Phase of the
project. Its intended audience is the project manager, project team, project
sponsor and any senior leaders whose support is needed to carry out the plan.
2 CHANGE MANAGEMENT PROCESS
The Change Management process establishes an orderly and effective procedure
for tracking the submission, coordination, review, evaluation, categorization, and
approval for release of all changes to the project’s baselines.
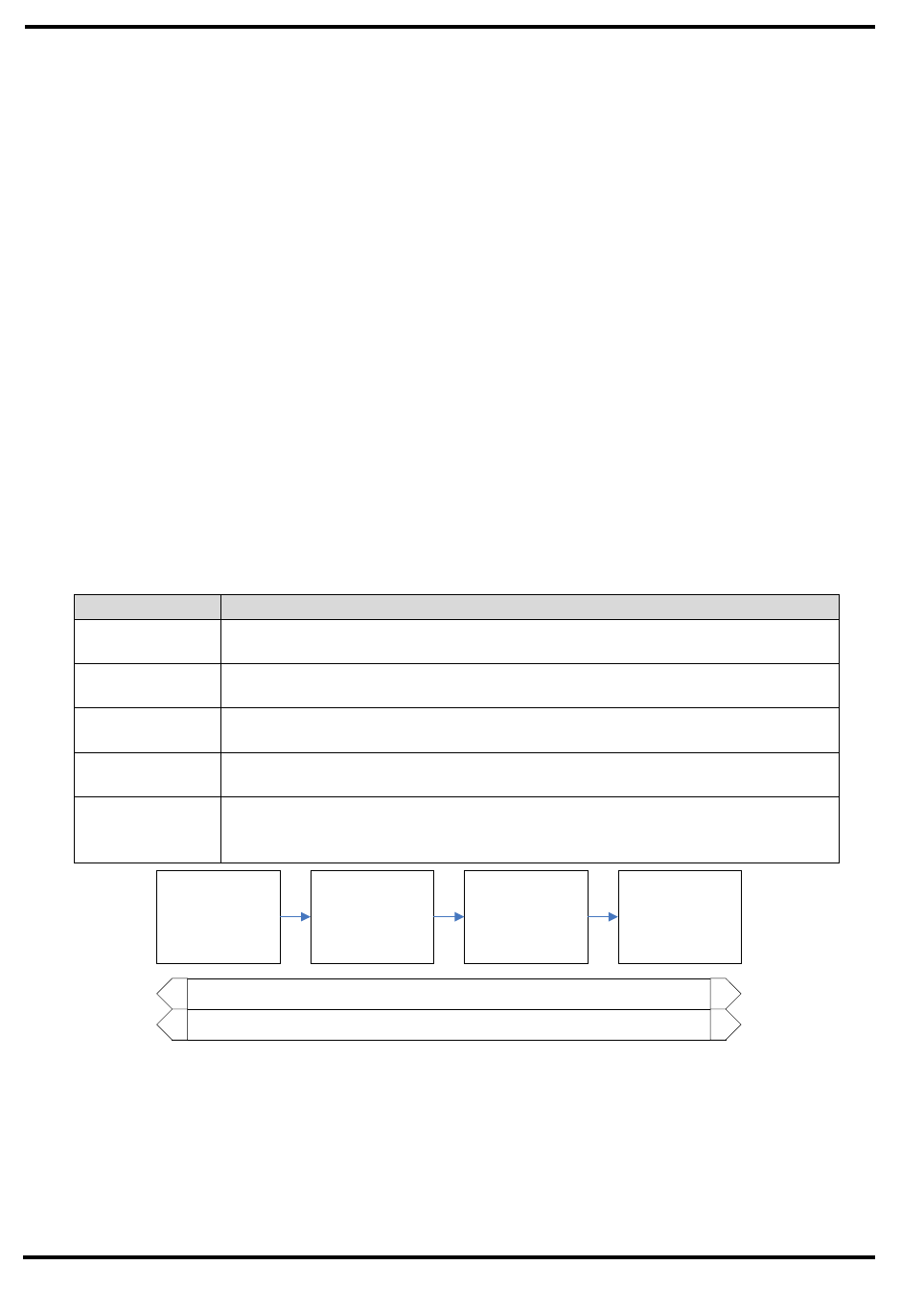
2.1 CHANGE REQUEST PROCESS FLOW REQUIREMENTS
[Outline the project team’s agreed upon change request (CR) process flow. The
following outlines a generic change request process flow.]
Step Description
Generate CR A submitter completes a CR Form and sends the completed form to the
Change Manager
Log CR Status The Change Manager enters the CR into the CR Log. The CR’s status is
updated throughout the CR process as needed.
Evaluate CR Project personnel review the CR and provide an estimated level of effort to
process, and develop a proposed solution for the suggested change
Authorize Approval to move forward with incorporating the suggested change into
the project/product
Implement If approved, make the necessary adjustments to carry out the requested
change and communicate CR status to the submitter and other
stakeholders
Generate CR
Report Status
Log Updated Status
Implement CR
Authorize CREvaluate CR

<Project Name>
Change Management Plan (v1.0) Page 5 of 9
[Insert appropriate disclaimer(s)]
2.2 CHANGE REQUEST FORM AND CHANGE MANAGEMENT LOG
[List and define the data elements the project team needs to include on the
Change Request Form and in the Change Management Log. At a minimum, the
following data should be included on the project’s Change Request Form and
Change Management Log.]
Element Description
Date
The date the CR was created
CR#
Assigned by the Change Manager
Title
A brief description of the change request
Description
Description of the desired change, the impact, or benefits of a change
should also be described
Submitter
Name of the person completing the CR Form and who can answer
questions regarding the suggested change
Phone
Phone number of the submitter
E-Mail
Email of the submitter
Product
The product that the suggested change is for
Version
The product version that the suggested change is for
Priority A code that provides a recommended categorization of the urgency of the
requested change (High, Medium, Low)
2.3 EVALUATING AND AUTHORIZING CHANGE REQUESTS
[In order to evaluate and prioritize a change request, the “priority” and “type” of the
change are taken into consideration. Use the first and second tables below to list
and define the “priority” and “type” data elements that are applicable for the
project. The third table provides examples of commonly used project status types.
The list of elements is at the discretion of the project manager.]
Change requests are evaluated using the following priority criteria:
Priority Description
High
<Insert the definition the project assigns to a high priority CR>
Medium
<Insert the definition the project assigns to a medium priority CR>
Low
<Insert the definition the project assigns to a low priority CR>
<priority>
<Insert the definition the project assigns to this level of priority CR>
Change requests are evaluated and assigned one or more of the following change
types:
Type Description
Scope
Change affecting scope
Time
Change affecting time
Duration
Change affecting duration
Cost
Change affecting cost
Resources
Change affecting resources
Deliverables
Change affecting deliverables
Product
Change affecting product
Processes
Change affecting process
Quality
Change affecting quality
<change type>
<define this change type>

<Project Name>
Change Management Plan (v1.0) Page 6 of 9
[Insert appropriate disclaimer(s)]
Change requests are evaluated and assigned one of the following status types:
Status Description
Open
Entered/Open but not yet approved or assigned
Work in
Progress
CR approved, assigned, and work is progressing
In Review
CR work is completed and in final review prior to testing
Testing
CR work has been reviewed and is being tested
Closed CR work is complete, has passed all tests, and updates have been
released.
<status type>
<define this status type CR>
2.3.1 Change Control Board
[A Change Control Board (CCB) is a formally constituted group of stakeholders
responsible for approving or rejecting changes to the project baselines. This group
may meet on a predefined schedule or on an as needed basis. The table below
provides a brief description of personnel acting as the Change Control Board
(CCB) and their role/level of authority within that group.]
Role
Name
Contact
Description
[Insert Role]
[Insert Name]
[Insert Contact #]
[Insert Role Description]
3 RESPONSIBILITIES
[Provide a brief description of persons responsible for each step of the change
management process for the project.]
Role
Name
Contact
Description
Project
Manager
Change
Manager
[Insert Role]
[Insert Name]
[Insert Contact #]
[Insert Role Description]

<Project Name>
Change Management Plan (v1.0) Page 7 of 9
[Insert appropriate disclaimer(s)]
Appendix A: Change Management Plan Approval
The undersigned acknowledge that they have reviewed the <Project Name>
Change Management Plan and agree with the information presented within this
document. Changes to this Change Management Plan will be coordinated with,
and approved by, the undersigned, or their designated representatives.
[List the individuals whose signatures are desired. Examples of such individuals
are Business Owner, Project Manager (if identified), and any appropriate
stakeholders. Add additional lines for signature as necessary.]
Signature:
Date:
Print Name:
Title:
Role:
Signature:
Date:
Print Name:
Title:
Role:
Signature:
Date:
Print Name:
Title:
Role:

<Project Name>
Change Management Plan (v1.0) Page 8 of 9
[Insert appropriate disclaimer(s)]
APPENDIX B: REFERENCES
[Insert the name, version number, description, and physical location of any
documents referenced in this document. Add rows to the table as necessary.]
The following table summarizes the documents referenced in this document.
Document Name
Description
Location
<Document Name and
Version Number>
<Document description>
<URL or Network path where document
is located>

<Project Name>
Change Management Plan (v1.0) Page 9 of 9
[Insert appropriate disclaimer(s)]
APPENDIX C: KEY TERMS
The following table provides definitions and explanations for terms and acronyms
relevant to the content presented within this document.
Term
Definition
[Insert Term]
<Provide definition of term and acronyms used in this document.>
CDER’s Enterprise Performance Lifecycle (EPLC) Initiative.
Appendix C: EMA Questions and Answers on
Post Approval Change Management Protocols

7 Westferry Circus ● Canary Wharf ● London E14 4HB ● United Kingdom
Telephone
+44 (0)20 7418 8400
Facsimile
+44 (0)20 7418 8416
E-mail
Website
www.ema.europa.eu
An agency of the European Union
© European Medicines Agency, 2012. Reproduction is authorised provided the source is acknowledged.
30 March 2012
EMA/CHMP/CVMP/QWP/586330/2010
Committee for Medicinal Products for Human Use (CHMP)
Questions and answers on post approval change
management protocols
Draft agreed by CHMP / CVMP Quality Working Party 9 September 2010
Adopted by CHMP for release for consultation 23 September 2010
Adopted by CVMP for release for consultation 14 October 2010
End of consultation (deadline for comments) 28 February 2011
Agreed by CHMP / CVMP Quality Working Party 2 February 2012
Adopted by CVMP 8 March 2012
Adopted by CHMP 15 March 2012
Date for coming into effect 1 October 2012
Keywords Post approval change management protocol; Variation; Control strategy
Use of Post Approval Change Management Protocols
1. Introduction:
The concept of post approval change management protocols has been introduced in the EU through the
Commission’s Guideline on the details of the various categories of variations to the terms of marketing
authorisations for medicinal products for human use and veterinary medicinal products (2010/C 17/01)
that supports the Variations Regulation (Commission Regulation (EC) No 1234/2008).
This Questions and Answers document sets some general principles about the content and future use
of these protocols and will be updated in the light of more experience, particularly for biological
products.
2. Scope:
This Questions and Answers document is intended to apply to all medicinal products for human and
veterinary use including biotechnological or biological products. It applies to all types of products,
irrespective of whether a traditional or enhanced Quality by Design (QbD) approach has been used for
product development. The use of Post Approval Change Management Protocols is optional.
3. What is a Post Approval Change Management Protocol?
A post-approval change management protocol describes specific changes that a company would like to
implement during the lifecycle of the product and how these would be prepared and verified. It is a
step-wise approach in the assessment of changes, which allows an early evaluation of the strategy for
the change and a later separate evaluation of the data produced based on the agreed strategy (Figure
1). Such a stepwise approach is expected to lead to faster and more predictable implementation of
changes post-approval, since the MAH will have obtained agreement from the Regulatory Authorities
about the proposed strategy and tests to verify the effect of the change on product quality.
Typically the variation category designated for reporting changes under an approved post approval
change management protocol is at least one category lower than would normally be the case.
Figure 1: Post Approval Change Management Protocols
+
Strategy
Results
+
Results
Strategy
Early Step 1: Fast Step 2:
Currently
Evaluation of a proposed
variation as a ‘whole’
(Strategy + Results)
Submission of a
Change
Management
Protocol
Reporting of
implementation of a
change in accordance
with an approved
protocol
Type II Variation
Type IA
IN
or IB
Questions and answers on post approval change management protocols
EMA/CHMP/CVMP/QWP/586330/2010 Page 2/6

Questions and answers on post approval change management protocols
EMA/CHMP/CVMP/QWP/586330/2010 Page 3/6
4. What should be in the content of a post approval change
management protocol?
In general, in order to support the proposed change, the company should submit all relevant
information that can demonstrate that it has acquired adequate knowledge to prepare and manage the
impact of the change.
The content of the protocol could include the following, depending on the nature of the change:
Justification that there is a recognised future need for the specific change within a reasonable
timeframe and that adequate knowledge has been acquired to define criteria to appropriately
evaluate and manage the change for the specific product concerned;
A detailed description of the proposed change. The differences with what is already approved should
be clearly highlighted (preferably in a tabular format). Depending upon the nature of the change, it
should be demonstrated, preferably with data from development or pilot scale studies, that the
proposed approach is feasible. If only lab-scale data are provided the potential scale up effect
should be discussed;
Risk assessment of the impact of the change on product quality. This should include identification of
the potential risks and detailed strategy of how these risks will be mitigated or managed;
Discussion on the appropriateness of the approved control strategy to identify and manage these
risks and, if required, description of the additional controls that might be needed to be put in place.
This should take into consideration the extent of the change and therefore the potential impact on
the quality of the active substance and/or finished product, as appropriate;
Description of the studies to be performed, and the test methods and acceptance criteria that will
be used to fully assess the effect of the proposed change on product quality
1
.The applicant should
justify the appropriateness of the methods proposed to assess the impact of the proposed change.
Data from development or pilot scale studies can provide assurance about the relevance and
adequacy of the proposed tests;
For biologics, the approach to be used to demonstrate the comparability of the pre- and post-
change product;
A plan for stability studies should be included, if appropriate;
Commitment to update the approved protocol, if this becomes invalid, due to significant changes to
the proposed test methods/acceptance criteria or a significant body of new knowledge or new
regulatory requirements;
In case that the protocol describes several changes, a justification showing that how the changes
are related, and that a simultaneous review under a single protocol is meaningful;
For chemical medicinal products, a proposal of how the implementation of the change will be
reported to the relevant competent authorities using the existing variation procedures, e.g., as a
Type IA / IAIN variation (implemented prior to notification) or Type IB variation (requires approval
before implementation);
1
Whenever a particular change is investigated it should be evaluated against the specified acceptance criteria in terms of
the relevant registered specification(s) and analytical methods at that point in time. It is recognised that these tests and
limits may have changed since the original protocol was presented and accepted.
However, the protocol will still remain valid provided the changes to the tests and limits are minor in nature and therefore
do not fundamentally impact the basis for the original protocol and have already been formally registered and, where
relevant, assessed and approved.

- If a Type IA/ IAIN variation has been chosen, then the conditions that need to be fulfilled by
the marketing authorisation holder (MAH) prior to the implementation of the change, as well as
a description of the amount and level of detail of the data to be provided, need to be clearly
stated;
- If a Type IB variation has been selected, then a description of the amount and level of detail of
the data to be provided should be included;
For biological medicinal products, in accordance with the Variations Classification Guideline, the
reporting will always be made as a Type IB variation.
If this is not the case, or if there are other fundamental changes to the protocol, they will need to be
separately updated as part of a Type IB variation before the change can be notified. However, in the
event that there are minor changes to an approved protocol that has already been agreed to be
notified as a Type IA (annual or immediate), where necessary, it will be possible to include the minor
changes to the protocol at the same time as the submission to implement the change.
These will need to be notified as Type IB variations and the change cannot be implemented prior to
approval.
5. What is the mechanism for the submission and evaluation
of a Post approval Change Management Protocol?
A post approval change management protocol may be included in an original marketing authorisation
application or (line) extension application, or may be submitted subsequently as a stand alone
variation. The Variations Classifications Guideline includes specific scopes for the introduction (Change
no. B.1e.2) or deletion (Change no. B.1e.3) of a protocol for the active substance and the finished
product (introduction B.2.g.2) (deletion B.2.g.3).
When submitted as part of the original marketing authorisation or a (line) extension application, the
evaluation of a post approval change management protocol will follow the rules of procedure applicable
to the actual marketing authorisation or extension application.
When submitted post approval, the evaluation of a post approval change management protocol will
follow the rules of procedure applicable for all Quality Type II variations with a 60 days timetable.
A change to an already approved protocol will be processed as a Type IB variation, unless it
fundamentally changes the content of the protocol. In the later case, the submission of a new protocol
would be required. This is very relevant for biological/biotech products where comparability principles
apply.
6. How will the change be implemented after all the studies
described in the approved protocol have been performed?
A prerequisite for the implementation of a change described in an approved protocol is that all studies
described in the protocol have been performed, and the results of the studies comply with the
predefined criteria set out in the protocol.
In all cases, a justification that the approved protocol is still valid should be provided, together with the
procedure number of the application that led to the approval of the protocol.
In the event that any of the relevant specifications (limits or analytical methods) that are to be used
for the evaluation have changed in a minor way since the original protocol was accepted, provided the
Questions and answers on post approval change management protocols
EMA/CHMP/CVMP/QWP/586330/2010 Page 4/6

changes have in the interim been formally registered, there is no requirement to submit an updated
protocol before the change is notified.
However, any changes to the specifications should be clearly highlighted as part of the reporting
variation. If this is not the case, or if there are other changes that fundamentally impact the basis of
the original protocol, the protocol itself should be formally updated before the change is notified (Type
IB variation) or a new protocol should be submitted (Type II variation) Minor deviations to the protocol
may be reported and justified in the variation (Type IB) submitted in support of the implementation of
the change. However, the change(s) cannot be implemented prior to approval.
The implementation of a change in accordance with an already approved protocol can be made via a
Type IAIN or Type IB variation (Change no. B.V.c.1 of the Variations Classification Guideline)
depending on whether it requires the evaluation of supportive data.
If a Type IAIN variation has been agreed during the evaluation of the protocol, then the applicant may
implement the change without any further regulatory evaluation prior to its approval. A notification of
the implementation should immediately be sent to the relevant competent authority.
If a Type IB variation has been agreed during the evaluation of the protocol, or is mandatory, as is the
case for biological medicinal products, then the applicant may only implement the change upon receipt
of a positive notification from the relevant Competent Authority. In both cases the respective timelines
and procedural requirements for Type IA and IB variations apply.
If the same protocol has been submitted for several products changes, the implementation of the
change can be made using the existing Grouping and Worksharing procedures.
7. Can applicants submit post approval change management
protocols for any type of change?
The types of changes that would benefit from, and consequently could be included in such a protocol,
depend on the complexity of the product and its manufacturing process, as well as the understanding
that the company has gained about them. It is not possible to set a priori a list of acceptable changes
since it depends on the type of the products, and the type of information presented in the dossier.
However, protocols should be specific to a product and should therefore not name multiple products,
although the proposed change(s) and management strategies may be applicable to other products and
processes.
In order for a change to be submitted and accepted as part of a protocol, the company should
demonstrate suitable scientific knowledge and understanding of the active/product and the process,
coupled with the use of an appropriate quality risk management and an efficient pharmaceutical quality
system.
Consequently, it is strongly recommended that companies submit post approval change management
protocols only for those changes that they are highly likely to implement and whose feasibility has
already been investigated and is supported by relevant data.
For a biological medicinal product where non-clinical/clinical data are needed as part of the
comparability exercise, a post approval management protocol will not be feasible.
Changes that should not be submitted in a post approval change management protocol include any
change that would result in a (line) extension to the original marketing authorisation.
Questions and answers on post approval change management protocols
EMA/CHMP/CVMP/QWP/586330/2010 Page 5/6

Questions and answers on post approval change management protocols
EMA/CHMP/CVMP/QWP/586330/2010 Page 6/6
8. Can a post approval change management protocol cover
multiple changes?
It is possible to cover more than one change in a single protocol provided that they are directly related
and a simultaneous review under a single protocol is meaningful. A justification should be provided in
the protocol.
Depending upon the specific nature of the change(s), it is also possible for the content of a protocol to
be applied on more than one occasion, e.g., new manufacturer of active substance starting material.
However, this will depend upon successful implementation each time, in line with the protocol. In
addition, the possibility will need to be transparent and therefore, if applicable, clearly stated in the
protocol itself.
9. Where should the requested documents (description of
change/change management protocol) be placed in the
application?
The protocol should be included in the marketing authorisation application as part of the Regional
Section 3.2.R of Module 3 (or in Part 2.G for veterinary applications). The relevant section(s) of
Module 3 (or Part II for veterinary), affected by the proposed change, should cross refer to the
protocol.
At the time of submission of the protocol (Type II) only 3.2.R/2.3.R (or Part 2.G for veterinary) is
affected. Update of the relevant section(s) of Module 3 (other than 3.2.R) (or Part II for veterinary) is
done with the Type I variation to implement the protocol.
A listing of the approved protocols should also be provided and maintained in Module 3.2.R (or Part
2.G for veterinary). This should include a listing of all the protocols with a description of the change,
the proposed reporting category and a reference to the relevant section in Module 3 (or Part 2 for
veterinary).
10. Are post approval change management protocols
applicable to all types of applications?
Change Management protocols are applicable to all types of applications, irrespective of the
development approach that has been followed, i.e., traditional or enhanced. However, it is expected
that if the applicant has applied Quality by Design principles during product development, then an
increased product and process understanding is achieved, thus making it easier to predict the impact
of a change on the active substance or finished product quality.

