
Medicaid Managed Care
Organization
2014 Annual Technical Report
Submitted by:
Delmarva Foundation
April 2015
HealthChoice and Acute Care Administration
Division of HealthChoice Quality Assurance

2014 Annual Technical Report
Delmarva Foundation
Table of Contents
Table of Contents
Executive Summary ............................................................................................................................................ i
Introduction ............................................................................................................................................... i
HACA Quality Strategy............................................................................................................................. ii
EQRO Program Assessment Activities ................................................................................................ iii
General Overview of Findings ............................................................................................................... iii
Assessment of Quality, Access, and Timeliness ........................................................................... iii
Recommendations and Corrective Action Plans for MCOs ........................................................ vi
Best and Emerging Practice Strategies .............................................................................................. ix
I. Systems Performance Review ............................................................................................................... 1
Introduction .............................................................................................................................................. 1
Purpose ..................................................................................................................................................... 1
Methodology ............................................................................................................................................. 1
Corrective Action Plan Process .............................................................................................................. 3
Findings ..................................................................................................................................................... 4
Conclusions ............................................................................................................................................ 26
II. Value Based Purchasing ...................................................................................................................... 27
Introduction ........................................................................................................................................... 27
Performance Measure Selection Process ......................................................................................... 27
Value Based Purchasing Validation ................................................................................................... 28
2013 VBP Incentive/Disincentive Target Setting Methodology .................................................... 32
2013 Value Based Purchasing Results ............................................................................................. 34
2013 VBP Financial Incentive and Disincentive Methodology ...................................................... 35
III. Performance Improvement Projects ................................................................................................. 36
Introduction ........................................................................................................................................... 36
Methodology .......................................................................................................................................... 37
Findings .................................................................................................................................................. 38
Recommendations ............................................................................................................................... 42
IV. Encounter Data Validation .................................................................................................................. 43
Introduction ........................................................................................................................................... 43
Encounter Data Validation Process ................................................................................................... 43
Medical Record Review Procedure .................................................................................................... 44
Analysis Methodology .......................................................................................................................... 45
Medical Record Sampling ................................................................................................................... 46
Results .................................................................................................................................................... 48
Conclusions and Recommendations ................................................................................................ 53

2014 Annual Technical Report
Delmarva Foundation
Table of Contents
V. EPSDT Medical Record Review ........................................................................................................... 54
Introduction ........................................................................................................................................... 54
Program Overview ................................................................................................................................ 54
Program Objectives .............................................................................................................................. 55
2013 EPSDT Review Process ............................................................................................................. 56
Findings .................................................................................................................................................. 58
Corrective Action Plan Process ........................................................................................................... 69
Conclusions ............................................................................................................................................ 69
VI. Healthcare Effectiveness Data and Information Set
®
.................................................................... 71
Introduction ........................................................................................................................................... 71
Measures Designated for Reporting .................................................................................................. 72
HEDIS
®
Methodology ............................................................................................................................ 77
Findings .................................................................................................................................................. 86
VII. Consumer Assessment of Health Providers and Systems
®
........................................................... 89
Introduction ........................................................................................................................................... 89
2014 CAHPS
®
5.0H Medicaid Survey Methodology ........................................................................ 89
Findings .................................................................................................................................................. 92
VIII. Consumer Report Card ........................................................................................................................ 99
Introduction ........................................................................................................................................... 99
Information Reporting Strategy .......................................................................................................... 99
Analytic Methodology ......................................................................................................................... 103
2014 Report Card Results................................................................................................................. 105
IX. Review of Compliance with Quality Strategy ................................................................................. 106
Recommendation for MCOs .............................................................................................................. 107
Recommendations for HACA ............................................................................................................ 107
Conclusion ............................................................................................................................................ 107
Appendices
Acronym List ....................................................................................................................................... A1-1
Adolescent Well Care (AWC) HEDIS
®
Specifications ................................................................... A2-1
Controlling High Blood Pressure (CBP) HEDIS
®
Specifications ................................................. A3-1
HEDIS
®
Result Tables ........................................................................................................................ A4-1
CY 2014 MD HealthChoice Performance Report Card ............................................................... A5-1

2014 Annual Technical Report
Delmarva Foundation
i
Executive Summary
Introduction
The Maryland Department of Health and Mental Hygiene (DHMH) is responsible for evaluating the quality
of care provided to eligible participants in contracted Managed Care Organizations (MCOs) through the
Maryland Medicaid Managed Care Program, known as HealthChoice. HealthChoice has been operational
since June 1997 and operates pursuant to Title 42 of the Code of Federal Regulations (CFR), Section 438.204
and the Code of Maryland Regulations (COMAR) 10.09.65. HealthChoice’s philosophy is based on providing
quality health care that is patient-focused, prevention-oriented, comprehensive, coordinated, accessible, and
cost-effective.
DHMH’s HealthChoice and Acute Care Administration (HACA) is responsible for coordination and
oversight of the HealthChoice program. HACA ensures that the initiatives established in 42 CFR 438,
Subpart D are adhered to and that all MCOs that participate in the HealthChoice program apply these
principles universally and appropriately. The mission of HACA is to continuously improve both the clinical
and administrative aspects of the HealthChoice Program. The functions and infrastructure of HACA support
efforts to identify and address quality issues efficiently and effectively. There is a systematic process where
DHMH identifies both positive and negative trends in service delivery and outcomes. Quality monitoring,
evaluation, and education through enrollee and provider feedback are integral parts of the managed care
process and help to ensure that health care is not compromised.
DHMH is required to annually evaluate the quality of care provided to HealthChoice participants by
contracting MCOs. In adherence to Federal law [Section 1932(c)(2)(A)(i) of the Social Security Act], DHMH
is required to contract with an External Quality Review Organization (EQRO) to perform an independent
annual review of services provided by each contracted MCO to ensure that the services provided to the
participants meet the standards set forth in the regulations governing the HealthChoice Program. For this
purpose, DHMH contracts with Delmarva Foundation to serve as the EQRO.
Delmarva Foundation is a non-profit organization established in 1973 as a Professional Standards Review
Organization. Over the years, the company has grown in size and in mission. Delmarva Foundation is
designated by the Centers for Medicare and Medicaid Services (CMS) as a Quality Improvement Organization
(QIO)-like entity and performs External Quality Reviews and other services to State of Maryland and
Medicaid agencies in a number of jurisdictions across the United States. The organization has continued to
build upon its core strength to develop into a well-recognized leader in quality assurance and quality
improvement.

2014 Annual Technical Report
Delmarva Foundation
ii
Delmarva Foundation is committed to supporting the Department’s guiding principles and efforts to provide
quality and affordable health care to its burgeoning population of Medicaid recipients. As the EQRO,
Delmarva Foundation maintains a cooperative and collaborative approach in providing high quality, timely,
and cost-effective services to the Department. Delmarva Foundation’s goal is to assist the Department in this
challenging economic environment.
The HealthChoice program served over 910,232 participants as of December 31, 2013 and contracted with
seven MCOs during this evaluation period. The seven MCOs evaluated during this period were:
AMERIGROUP Community Care (ACC)
Priority Partners (PPMCO)
Jai Medical Systems, Inc. (JMS)
Riverside Health of Maryland (RHMD)
Maryland Physicians Care (MPC)
UnitedHealthcare (UHC)
MedStar Family Choice, Inc. (MSFC)
RHMD began participating in the HealthChoice program in February 2013. The EQRO’s evaluation of
RHMD for calendar year (CY) 2013 included only the Systems Performance Review and Early and Periodic
Screening, Diagnosis, and Treatment (EPSDT) Medical Record Reviews, as the MCO did not have a full year
of participation in the HealthChoice system. Their participation in all EQRO activities will begin in CY 2015.
Pursuant to 42 CFR 438.364, this Annual Technical Report describes the findings from Delmarva
Foundation’s External Quality Review activities for years 2012-2013 which took place in CY 2014. The report
includes each review activity conducted by Delmarva Foundation, the methods used to aggregate and analyze
information from the review activities, and conclusions drawn regarding the quality, access, and timeliness of
healthcare services provided by the HealthChoice MCO.
HACA Quality Strategy
The overall goals of the Department’s Quality Strategy are to:
Ensure compliance with changes in Federal/State law and regulation;
Improve performance over time;
Allow comparisons to national and state benchmarks;
Reduce unnecessary administrative burden on MCOs; and,
Assist the Department with setting priorities and responding to identified areas of concern such as
children, pregnant women, children with special healthcare needs, adults with disabilities, and adults with
chronic conditions.
HACA works collaboratively with MCOs and stakeholders to identify opportunities for improvement and to
initiate quality improvement activities that will impact the quality of health care services for HealthChoice
participants.

2014 Annual Technical Report
Delmarva Foundation
iii
EQRO Program Assessment Activities
Federal regulations require that three mandatory activities be performed by the EQRO using methods
consistent with protocols developed by the CMS for conducting the activities. These protocols specify that
the EQRO must conduct the following activities to assess managed care performance:
1) Conduct a review of MCOs’ operations to assess compliance with State and Federal standards for quality
program operations;
2) Validate State required performance measures; and
3) Validate State required Performance Improvement Projects (PIPs) that were underway during the prior
12 months.
Delmarva Foundation also conducted an optional activity: validation of encounter data reported by the
MCOs. As the EQRO, Delmarva Foundation conducted each of the mandatory activities and the optional
activities in a manner consistent with the CMS protocols during CY 2014.
Additionally, the following two review activities were conducted by Delmarva Foundation:
1) Conduct the EPSDT Medical Record Reviews; and
2) Develop and produce an annual Consumer Report Card to assist participants in selecting an MCO.
In aggregating and analyzing the data from each activity, Delmarva Foundation allocated standards and/or
measures to domains indicative of quality, access, and timeliness of care and services. Separate report sections
address each review activity and describe the methodology and data sources used to draw conclusions for the
particular area of focus. The final report section summarizes findings and recommendations to HACA and
the MCOs to further improve the quality of, timeliness of, and access to health care services for
HealthChoice participants.
General Overview of Findings
Assessment of Quality, Access, and Timeliness
For the purposes of evaluating the MCOs, Delmarva Foundation has adopted the following definitions
for quality, access, and timeliness:
Quality, as it pertains to external quality review, is defined as “the degree to which an MCO or
Prepaid Inpatient Health Plan increases the likelihood of desired health outcomes of its participants
(as defined in 42 CFR 438.320[2]) through its structural and operational characteristics and through
the provision of health services that are consistent with current professional knowledge.” ([CMS],
Final Rule: Medicaid Managed Care; 42 CFR Part 400, et. al. Subpart D- Quality Assessment and Performance
Improvement, [June 2002]).

2014 Annual Technical Report
Delmarva Foundation
iv
Access (or accessibility), as defined by the National Committee for Quality Assurance (NCQA), is “the
extent to which a patient can obtain available services at the time they are needed. Such service refers to
both telephone access and ease of scheduling an appointment, if applicable. The intent is that each
organization provides and maintains appropriate access to primary care, behavioral health care, and
member services.” (2006 Standards and Guidelines for the Accreditation of Managed Care Organizations).
Timeliness, as it relates to utilization management decisions and as defined by NCQA, is whether “the
organization makes utilization decisions in a timely manner to accommodate the clinical urgency of the
situation. The intent is that organizations make utilization decisions in a timely manner to minimize any
disruption in the provision of health care.” (2006 Standards and Guidelines for the Accreditation of Managed
Care Organizations). An additional definition of timeliness given in the Institute of Medicine National
Health Care Quality Report refers to “obtaining needed care and minimizing unnecessary delays in
getting that care.” (Envisioning the National Health Care Quality Report, 2001).
Table 1 outlines the review activities conducted annually that assess quality, access, and timeliness.
Table 1. Review Activities that Assess Quality, Access, and Timeliness
Annual Review Activities that Assess Quality, Access, and Timeliness
Systems Performance Review
Quality
Access
Timeliness
Standard 1 - Systematic Process of Quality Assessment and Improvement
√
Standard 2 - Accountability to the Governing Body
√
Standard 3 - Oversight of Delegated Entities
√
Standard 4 - Credentialing and Recredentialing
√
√
√
Standard 5 - Enrollee Rights
√
√
√
Standard 6 - Availability and Accessibility
√
√
Standard 7 - Utilization Review
√
√
√
Standard 8 - Continuity of Care
√
√
√
Standard 9 - Health Education Plan
√
√
Standard 10 - Outreach Plan
√
√
Standard 11 - Fraud and Abuse
√
√
Value Based Purchasing
Quality
Access
Timeliness
Adolescent Well Care
√
√
√
Ambulatory Care Services for SSI Adults Ages 21–64 Years
√
√
Ambulatory Care Services for SSI Children Ages 0–20 Years
√
√
Cervical Cancer Screening for Women Ages 21–64 Years
√
√
Childhood Immunization Status (Combo 3)
√
√
Eye Exams for Diabetics
√
√
Immunizations for Adolescents
√
√

2014 Annual Technical Report
Delmarva Foundation
v
Value Based Purchasing
Quality
Access
Timeliness
Lead Screenings for Children Ages 12–23 Months
√
√
Postpartum Care
√
√
√
Well-Child Visits for Children Ages 3 – 6 Years
√
√
√
Performance Improvement Project
Quality
Access
Timeliness
Adolescent Well Care PIP
√
√
√
High Blood Pressure PIP
√
√
√
EPSDT Medical Record Review
Quality
Access
Timeliness
Health and Developmental History
√
√
Comprehensive physical examination
√
√
Laboratory tests/at-risk screenings
√
√
Immunizations
√
√
Health education and anticipatory guidance
√
√
Encounter Data Validation
Quality
Access
Timeliness
Inpatient, Outpatient, Office Visit Medical Record Review
√
HEDIS
®
Quality
Access
Timeliness
Childhood Immunization Status
√
√
Immunizations for Adolescents
√
√
Appropriate Treatment for Children with Upper Respiratory Infection
√
Appropriate Testing for Children with Pharyngitis
√
Breast Cancer Screening
√
√
Cervical Cancer Screening
√
√
Chlamydia Screening in Women
√
√
Comprehensive Diabetes Care
√
√
Use of Appropriate Medications for People with Asthma
√
Use of Imaging Studies for Low Back Pain
√
Avoidance of Antibiotic Treatment in Adults with Acute Bronchitis
√
Adult BMI Assessment
√
√
Controlling High Blood Pressure
√
√
Annual Monitoring for Patients on Persistent Medications
√
√
Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis
√
Medication Management for People with Asthma
√
Adults’ Access to Preventive/Ambulatory Health Services
√
√
Children and Adolescents’ Access to Primary Care Practitioners
√
√
Prenatal and Postpartum Care
√
√
Call Answer Timeliness
√
√

2014 Annual Technical Report
Delmarva Foundation
vi
HEDIS
Quality
Access
Timeliness
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment
√
√
Frequency of Ongoing Prenatal Care
√
√
√
Well-Child Visits in the First 15 Months of Life
√
√
√
Well-Child Visits in the 3
rd
, 4
th
, 5
th
and 6
th
Years of Life
√
√
√
Adolescent Well-Care Visits
√
√
√
Ambulatory Care
√
Identification of Alcohol and Other Drug Services
√
√
Weight Assessment and Counseling for Nutrition and Physical Activity for
Children/Adolescents
√
√
√
Use of Sprirometry Testing in the Assessment and Diagnosis of COPD
√
√
Pharmacotherapy Management of COPD Exacerbation
√
√
Asthma Medication Ratio
√
Persistence of Beta-Blocker Treatment After a Heart Attack
√
√
CAHPS
®
Quality
Access
Timeliness
Getting Needed Care
√
Getting Care Quickly
√
How Well Doctors Communicate
√
Customer Service
√
√
Shared Decision Making
√
Health Promotion and Education
√
Coordination of Care
√
Access to Prescription Medication*
√
Access to Specialized Services*
√
Family Centered Care: Personal Doctor Who Knows Your Child*
√
Family Centered Care: Getting Needed Information*
√
Coordination of Care for Children with Chronic Conditions*
√
*Additional Composite Measures for Children with Chronic Conditions
Recommendations and Corrective Action Plans for MCOs Prior Year Review Activities
Systems Performance Review
Although the Maryland (MD) MCO Aggregate rate was 99% in CY 2012, MCOs were required to submit
systems performance review Corrective Action Plans (CAPs) in areas where opportunities for improvement
were identified or in areas where non-conformance with federal and contractual operational systems were
noted.

2014 Annual Technical Report
Delmarva Foundation
vii
The following CAPs were required from the MCOs in the last review period (January 1, 2012 – December 31,
2012):
ACC provided evidence of compliance with preauthorization determination.
ACC provided evidence of compliance with adverse determination notification time frames including the
process for reporting compliance with notification time frames.
ACC revised the Utilization Management Timeliness Audit Policy to incorporate the process for
monitoring and reporting compliance with State-required notification time frames.
JMS provided evidence of meeting time frames set forth in the MCO’s policies regarding recredentialing
decision date requirements.
PPMCO revised the Delegation Policy to ensure that it was in compliance with committee review and
approval of delegate complaint, grievance, and appeal reports on a quarterly basis.
PPMCO provided evidence of the appropriate committee’s review and approval of the annual Utilization
Management Program (UMP) and Utilization Management (UM) criteria for each entity that has been
delegated UM, including Block Vision.
PPMCO provided evidence of meeting time frames set forth in the MCO’s policies regarding
recredentialing decision date requirements in all records reviewed.
PPMCO resolved inconsistency between the Clinical Review Criteria Policy and the UMP as it pertains to
the responsibility for the development of internal criteria.
PPMCO demonstrated compliance with determination and notification time frames for all
preauthorization requests consistent with State regulation or MCO standards if the latter are more
stringent than State regulation.
PPMCO demonstrated that it consistently includes all required components in its adverse determination
letters.
UHC demonstrated that it provides ongoing monitoring of vendor CAPs specific to the MCO, with
documentation to support progress and resolution or recommendation for termination.
UHC clarified in the Delegation Manual which committee is responsible for review and approval of
delegate quarterly complaints and grievances reports.
UHC provided evidence that the appropriate committee reviews and approves quarterly complaint and
grievance reports on a quarterly basis.
UHC provided clearer documentation of committee review and approval of delegate reports to identify
the time period being reviewed.
UHC provided evidence of compliance with the 95% threshold for meeting regulatory time frames for
preauthorization determinations and for adverse determination notifications for any service requiring pre-
authorization regardless of which unit conducts the review.
UHC provided documentation to support how compliance is measured and evidence of corrective action
when time frames are not met.

2014 Annual Technical Report
Delmarva Foundation
viii
UHC provided evidence of a CAP to meet the minimum thresholds for compliance consistent with those
established by the State.
UHC provided a CAP to come into compliance with the 100% threshold for meeting regulatory time
frames for resolution of all expedited and routine appeals, including medical, substance abuse (SA), and
pharmacy. Additionally, MCO minimum thresholds for compliance must be consistent with those
established by the State.
UHC provided evidence of the Compliance Committee’s review and approval of administrative and
management procedures, including mandatory compliance plans to prevent fraud and abuse, for each
delegate that the MCO contracts with.
Overall, the MCOs demonstrated a commitment to providing quality and comprehensive health care to
HealthChoice members. Although these CAPs were followed up on in CY 2013, opportunities still remain
primarily in the areas of delegation and utilization management.
Performance Improvement Projects
Multiple recommendations were made to the MCOs as a result of the CY 2012 PIP review activities:
Complete a thorough and annual barrier analysis which will direct where limited resources can be most
effectively used to drive improvement.
Develop system-level interventions, which include educational efforts, changes in policy, targeting of
additional resources, or other organization-wide initiatives. Face-to-face contact is usually most effective.
To improve outcomes, interventions should be systematic (affecting a wide range of members, providers
and the MCO), timely, and effective.
Assess interventions for their effectiveness, and make adjustments where outcomes are unsatisfactory.
Detail the list of interventions (who, what, where, when, how many) to make the intervention
understandable and so that there is enough information to determine if the intervention was effective.
Although these recommendations were addressed by the MCOs in the CY 2012 PIPs, continued
opportunities for improvements remain for MCOs to improve both qualitative and quantitative analyses of
the study populations.
EPSDT Medical Record Review
The result of the EPSDT review demonstrates strong compliance with the timely screening and preventive
care requirements of the HealthChoice/EPSDT Program. The results of the CY 2012 review demonstrated
that improvements were needed in the following areas:
Immunizations - this component continued to decline by two percentage points again this year.
Laboratory Tests/At-Risk Screenings - this component showed a slight increase of one percentage point.
The Laboratory Tests/At-Risk Screenings component represents an area in most need of improvement.
Recommendations for quality improvement continue to be shared with MCOs annually.

2014 Annual Technical Report
Delmarva Foundation
ix
Two MCOs (MPC and PPMCO) required CAPs in CY 2012 for the Laboratory Tests/At-Risk Screenings
component. Although these CAPs were followed up on in CY 2013, continued opportunities were seen in the
area of Laboratory Tests/At-Risk Screenings. Overall review scores demonstrated that the Primary Care
Physicians (PCPs) and MCOs are committed to providing care that is patient focused and prevention
oriented.
Best and Emerging Practice Strategies
The MCOs effectively addressed quality, timeliness, and access to care issues in their respective managed care
populations. The MCOs implemented the following best practice strategies:
ACC has a comprehensive policy and procedure for the identification, referral, assignment of severity and
action taken to address clinical quality of care issues.
ACC has an objective means for scoring provider office site visits. The scoring guideline provides a
threshold for performance so that reviewers are able to determine when a CAP is required.
ACC has a highly integrated approach to care management of its members designed to address their
somatic and behavioral health needs.
JMS utilizes a prompt evaluation and approval schedule of the QA Program which ensures that quality
improvement efforts are effective in order to identify the need for program change. For example, its
BOD reviews and approves the Quality Assurance (QA) Annual Evaluation, QAP Description and QA
Work Plan for the year within the first quarter of the operational year.
JMS provides a very detailed description of any additional information needed for reconsideration in all
adverse determination letters.
JMS health education classes/programs reflect the needs of the population based upon data analysis and
provider recommendations.
MPC has a well-documented process for performing the practice site reviews and what should be
addressed during the reviews.
MPC includes language in all adverse determination letters documenting the rationale for the
determination which is very clear and easy to understand for a layperson. Letters explain in detail the
reason for the determination, any authorization requirements, and any additional information needed for
reconsideration.
MPC consistently performed well above the State performance threshold for both determination and
notification time frames.
MSFC provides a very detailed and easily understandable explanation for the adverse determination as
well as additional information needed for reconsideration.
MSFC provides its members a very comprehensive menu of health education programs and support
groups throughout the community.

2014 Annual Technical Report
Delmarva Foundation
x
MSFC completes a comprehensive analysis of survey results from the Provider Health Education Survey
which supports the MCO in providing programs that are relevant and of value to the MCO population.
PPMCO continues to demonstrate excellent discovery methods for capturing, reporting, and tracking
QOC issues by provider.
PPMCO conducts thorough reviews of all provider applications and reviews 100% of practitioners for
malpractice history, independent of the outcome. Both, deliberations and committee decisions are clearly
documented in the meeting minutes.
UHC has a very engaged Provider Advisory Committee lead by the MCO’s Chief Medical Officer.
Meeting minutes reflect active provider discussion on operational issues that affect both members and
providers.
UHC completed a comprehensive analysis of CAHPS
®
and Provider Satisfaction survey results including
comparing results to goals/benchmarks and identifying barriers, opportunities for improvement, and
related interventions.

2014 Annual Technical Report
Delmarva Foundation
1
Section I
Systems Performance Review
Introduction
As the EQRO, Delmarva Foundation performed an independent annual review of services provided under
each MCO contract in order to ensure that the services provided to the participants meet the standards set
forth in the regulations governing the HealthChoice Program. COMAR 10.09.65 requires that all
HealthChoice MCOs comply with the Systems Performance Review (SPR) standards and all applicable
federal and state laws and regulations. This section describes the findings from the SPR for CY 2013,
conducted in January and February of 2014. All seven MCOs were evaluated during this review period.
The SPRs were conducted at the MCO’s corporate offices and performed by a review team consisting of
health professionals, a nurse practitioner, and two masters prepared reviewers. The team has combined
experience of more than 45 years in managed care and quality improvement systems, 33 years of which are
specific to the HealthChoice program.
Purpose
The purpose of the SPR is to provide an annual assessment of the structure, process, and outcome of each
MCO’s internal quality assurance programs. Through the systems review, the team is able to identify, validate,
quantify, and monitor problem areas. The team completed the reviews and provided feedback to the Division
of HealthChoice Quality Assurance (DHQA) and each MCO with the goal of improving the care provided to
HealthChoice participants.
Methodology
For CY 2013, COMAR 10.09.65.03 required that all HealthChoice MCOs comply with the SPR standards
established by the Department and all applicable federal and state laws and regulations.
The following eleven performance standards were included in the CY 2013 review cycle:
Systematic Process of Quality Assessment*
Accountability to the Governing Body
Oversight of Delegated Entities
Credentialing and Recredentialing
Enrollee Rights
Availability and Accessibility

2014 Annual Technical Report
Delmarva Foundation
2
Utilization Review (UR)
Continuity of Care
Health Education*
Outreach*
Fraud and Abuse
*Note: These standards were exempt from the CY 2013 review cycle for all MCOs except for RHMD, as this was the MCO’s first SPR.
For CY 2013, all MCOs (except for RHMD) were expected to meet the compliance rate of 100% for all
standards. RHMD’s compliance rate was set at 80% for its first SPR. The MCOs were required to submit a
CAP for any standard that did not meet the minimum compliance rate.
In September 2013, Delmarva provided the MCOs with a “Medicaid Managed Care Organization Systems
Performance Review Orientation Manual” for Calendar Year 2013 and invited the MCOs to direct any
questions or issues requiring clarification to specific Delmarva and DHQA staff. The manual included the
following information:
Overview of External Quality Review Activities
CY 2013 Review Timeline
External Quality Review Contact Persons
Pre-site Visit Overview and Survey
Pre-site SPR Document List
Systems Performance Review Standards, including CY 2013 changes
System Performance Standards and Guidelines
Prior to the on-site review, the MCOs were required to submit a completed pre-site survey form and provide
documentation for various processes such as quality and UM, delegation, credentialing, enrollee rights,
continuity of care, outreach, and fraud and abuse policies. The documents provided were reviewed by
Delmarva staff prior to the on-site visit.
During the on-site reviews in January and February of 2014, the team conducted structured interviews with
key MCO staff and reviewed all relevant documentation needed to assess the standards. At the conclusion,
exit conferences were held with the MCOs. The purpose of the conferences was to provide the MCOs with
preliminary findings, based on interviews and all documentation reviewed. Notification was also provided
during the exit conferences that the MCOs would receive a follow-up letter describing potential issues that
could be addressed by supplemental documents, if available. The MCOs were given 10 business days from
receipt of the follow-up letter to submit any additional information to Delmarva; documents received were
subsequently reviewed against the standard(s) to which they related.

2014 Annual Technical Report
Delmarva Foundation
3
After completing the on-site review, Delmarva documented its findings for each standard by element and
component. The level of compliance for each element and component was rated with a review determination
of met, partially met, or unmet, as follows:
Met
100%
Partially Met
50%
Unmet
0%
Each element or component of a standard was of equal weight. A CAP was required for each performance
standard that did not meet the minimum required compliance rate, as defined for the CY 2013 review.
If an MCO chose to have standards in their policies and procedures that were higher than what was required
by DHMH, the MCO was held accountable to the standards which were outlined in their policies and
procedures during the SPR.
The Department had the discretion to change a review finding to “Unmet” based on the fact that it has been
found “Partially Met” for more than one consecutive year.
Preliminary results of the SPR were compiled and submitted to DHMH for review. Upon the Department’s
approval, the MCOs received a report containing individual review findings. After receiving the preliminary
reports, the MCOs were given 45 calendar days to respond to Delmarva with required CAPs. The MCOs could
have also responded to any other issues contained in the report at its discretion within this same time frame,
and/or requested a consultation with DHMH and Delmarva to clarify issues or ask for assistance in preparing
a CAP.
Corrective Action Plan Process
Each year the CAP process is discussed during the annual review meeting. This process requires that each
MCO must submit a CAP which details the actions to be taken to correct any deficiencies identified during
the SPR. CAPs must be submitted within 45 calendar days of receipt of the preliminary report. CAPs are
reviewed by Delmarva and determined to be adequate only if they address the following required elements
and components:
Action item(s) to address each required element or component
Methodology for evaluating the effectiveness of actions taken
Time frame for each action item, including plans for evaluation
Responsible party for each action item

2014 Annual Technical Report
Delmarva Foundation
4
In the event that a CAP is deemed unacceptable, Delmarva Foundation will provide technical assistance to
the MCO until an acceptable CAP is submitted. Three MCOs were required to submit CAPs for the CY 2013
SPR. All CAPs were submitted, reviewed, and found to adequately address the standard in which the
deficiencies occurred.
Delmarva reviewed any additional materials submitted by the MCO, made appropriate revisions to the
MCO’s final report, and submitted the report to the DHMH for review and approval. The Final MCO
Annual System Performance Review Reports were mailed to the MCOs.
Corrective Action Plan Review
CAPs related to the SPR can be directly linked to specific components or standards. The annual SPR for CY
2014 will determine whether the CAPs from the CY 2013 review were implemented and effective. In order to
make this determination, Delmarva Foundation will evaluate all data collected or trended by the MCO
through the monitoring mechanism established in the CAP. In the event that an MCO has not implemented
or followed through with the tasks identified in the CAP, DHMH will be notified for further action.
Findings
The HealthChoice MCO annual SPR consists of 8 to 11 standards, depending on the MCO. The compliance
threshold established by DHMH for all standards for CY 2013 is 100% for all MCOs, except for RHMD for
which the compliance threshold is set at 80% for its first SPR.
All seven HealthChoice MCOs participated in the SPR. In areas where deficiencies were noted, the MCOs
were provided recommendations that, if implemented, should improve their performance for future reviews.
If the MCO’s score was below the minimum threshold, a CAP was required. Four MCOs (ACC, JMS, MPC,
and MSFC) received perfect scores in all standards. Three MCOs (PPMCO, RHMD, and UHC) were
required to submit CAPs for CY 2013. All CAPs were submitted, reviewed, and found to adequately address
the standard in which the deficiencies occurred.
Table 2 provides for a comparison of SPR results across MCOs and the MD MCO Compliance for the CY
2013 review.

2014 Annual Technical Report
Delmarva Foundation
5
Table 2. CY 2013 MCO Compliance Rates
Standard
Description
Elements
Reviewed
MD MCO
Compliance
Rate
ACC
JMS
MPC
MSFC
PPMCO
RHMD**
UHC
1
Systematic Process
33
100%
Exempt
Exempt
Exempt
Exempt
Exempt
100%
Exempt
2
Governing Body
10
100%
100%
100%
100%
100%
100%
100%
100%
3
Oversight of Delegated
Entities
7
83%*
100%
100%
100%
100%
100%
36%*
71%*
4
Credentialing
38
98%*
100%
100%
100%
100%
100%
98%*
100%
5
Enrollee Rights
21
96%*
100%
100%
100%
100%
90%*
94%*
90%*
6
Availability and Access
10
96%*
100%
100%
100%
100%
95%*
80%*
100%
7
Utilization Review
24
90%*
100%
100%
100%
100%
80%*
67%*
85%*
8
Continuity of Care
4
100%
100%
100%
100%
100%
100%
100%
100%
9
Health Education Plan
12
88%*
Exempt
Exempt
Exempt
Exempt
Exempt
88%*
Exempt
10
Outreach Plan
14
93%*
Exempt
Exempt
Exempt
Exempt
Exempt
93%*
Exempt
11
Fraud and Abuse
19
98%*
100%
100%
100%
100%
100%
89%*
100%
*Denotes that the minimum compliance rate of 100% was unmet.
**RHMD’s minimum compliance threshold is set at 80%, as this was the MCO’s first SPR.

2014 Annual Technical Report
Delmarva Foundation
6
For each standard assessed for CY 2013, the following section describes the requirements reviewed; the
results, including the MD MCO compliance rate; the overall MCO findings; the individual MCO
opportunities for improvement and CAP requirements, if applicable; and follow up, if required.
STANDARD 1: Systematic Process of Quality Assessment/Improvement
Requirements: The Quality Assurance Program (QAP) objectively and systematically monitors/evaluates the
quality of care (QOC) and services to participants. Through QOC studies and related activities, the MCO pursues
opportunities for improvement on an ongoing basis. The QAP studies monitor QOC against clinical practice
guidelines which are based on reasonable evidence based practices. The QAP must have written guidelines for its
QOC studies and related activities that require the analysis of clinical and related services. The QAP must include
written procedures for taking appropriate corrective action whenever inappropriate or substandard services are
furnished. The QAP must have written guidelines for the assessment of the corrective actions. The QAP
incorporates written guidelines for evaluation of the continuity and effectiveness of the QAP. A comprehensive
annual written report on the QAP must be completed, reviewed, and approved by the MCO governing body. The
QAP must contain an organizational chart that includes all positions required to facilitate the QAP.
Results:
All MCOs (except for RHMD) were exempt from this standard. This standard was exempt as each MCO has
received compliance ratings of 100% for the past three consecutive years.
RHMD met the minimum compliance threshold for this standard.
Findings: This was RHMD’s first review of their QAP. It was found to be comprehensive in scope and to
appropriately monitor and evaluate the quality of care and service to members using meaningful and relevant
performance measures. Clinical care standards and/or practice guidelines are in place which the MCOs monitor
performance against annually, and clinicians monitor and evaluate quality through review of individual cases where
there are questions about care. Additionally, there is evidence of development, implementation, and monitoring of
corrective actions.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
7
STANDARD 2: Accountability to the Governing Body
Requirements: The governing body of the MCO is the Board of Directors or, where the Board’s participation with
the quality improvement issues is not direct; a committee of the MCO’s senior management is designated. The
governing body is responsible for monitoring, evaluating, and making improvements to care. There must be
documentation that the governing body has oversight of the QAP. The governing body must approve the overall
QAP and an annual QAP. The governing body formally designates an accountable entity or entities within the
organization to provide oversight of quality assurance, or has formally decided to provide oversight as a committee.
The governing body must routinely receive written reports on the QAP that describe actions taken, progress in
meeting quality objectives, and improvements made. The governing body takes action when appropriate and directs
that the operational QAP be modified on an ongoing basis to accommodate review of findings and issues of concern
within the MCO. The governing body is active in credentialing, recredentialing, and utilization review activities.
Results: The overall MD MCO Compliance Rate was 100% for CY 2013.
Findings: Overall, MCOs continue to have appropriate oversight by their governing boards. Evidence was provided
of the oversight provided by the governing body, along with ongoing feedback and direction of quality improvement
activities and operational activities of the MCO.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
8
STANDARD 3: Oversight of Delegated Entities
Requirements: The MCO remains accountable for all functions, even if certain functions are delegated to other
entities. There must be a written description of the delegated activities, the delegate's accountability for these
activities, and the frequency of reporting to the MCO. The MCO has written procedures for monitoring and
evaluating the implementation of the delegated functions and for verifying the quality of care being provided. The
MCO must also provide evidence of continuous and ongoing evaluation of delegated activities.
Results:
The overall MD MCO Compliance Rate was 83% for CY 2013.
ACC, JAI, MPC, MSFC, and PPMCO met the minimum compliance threshold for this standard.
RHMD and UHC were required to submit CAPs.
Findings: MCOs continue to demonstrate opportunities for improvement in this standard regarding delegation
policies and procedures and in the monitoring and evaluation of delegated functions.
MCO Opportunity/CAP Required
RHMD Opportunities/CAPs:
Element 3.1 – There is a written description of the delegated activities, the delegate’s accountability for these
activities, and the frequency of reporting to the MCO.
RHMD received a finding of partially met because the delegated agreements provided a detailed listing of specific
delegated claims processing activities and procedures; however, no specific performance measures or reporting
requirements were identified. Additionally, formalized responsibilities, which had been delegated to the vendor and
clearly outlined in amendments, were not found for functions such as complaints, grievances, and appeals.
In order to receive a finding of met in the CY 2014 SPR, RHMD must ensure that all delegation agreements
accurately reflect responsibility for specific delegated activities. Additionally, specific reporting requirements and
performance measures need to be included in all delegation agreements.
Component 3.3b – There is evidence of continuous and ongoing evaluation of delegated activities, including
quarterly review and approval of reports from the delegates that are produced at least quarterly regarding
complaints, grievances, and appeals, where applicable.
RHMD received a finding of unmet because there was no evidence of Quality Imporvement Committee (QIC)
quarterly review and approval of two delegated vendors’ quarterly complaint, grievance, and appeal reports for the
first, second, or third quarter of 2013. The MCO did not commence operations until February of 2013; therefore,
there were no delegated activities for the fourth quarter of 2012.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of formal review and
approval of delegate quarterly complaint, grievance, and appeal reports on a quarterly basis by the appropriate
committee designated in the MCO's policy.

2014 Annual Technical Report
Delmarva Foundation
9
Component 3.3c - There is evidence of continuous and ongoing evaluation of delegated activities, including
review and approval of claims payment activities, where applicable.
RHMD received a finding of unmet because there was no evidence of the QIC’s review and approval of three
delegated vendors’ claims activities reports since the MCO’s commencement of operations in mid-February 2013.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of formal review and
approval of delegate claims activities reports by the appropriate committee designated in the MCO's policy and
according to the stated frequency.
Component 3.3d - There is evidence of continuous and ongoing evaluation of delegated activities, including
review and approval of the delegated entities’ UM plan, which must include evidence of review and approval
of UM criteria by the delegated entity, where applicable.
RHMD received a finding of unmet because there was no evidence of QIC review and approval of the annual UMP
and UM criteria from two of the delegated vendors in 2013.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of formal review and
approval of each delegate's annual UMP and UM criteria by the appropriate committee designated in the MCO's
policy.
Component 3.3e - There is evidence of continuous and ongoing evaluation of delegated activities, including
review and approval of over and underutilization reports, where applicable.
RHMD received a finding of unmet because there was no evidence of QIC review and approval for two delegated
vendors over and underutilization reports since the MCO’s commencement of operations in mid-February 2013.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of formal review and
approval of each delegate's over/under utilization report(s) by the appropriate committee designated in the MCO's
policy and according to the stated frequency.
UHC Opportunities/CAPs:
Component 3.3a – There is evidence of continuous and ongoing evaluation of delegated activities, including
oversight of delegated entities’ performance to ensure the quality of the care and/or service provided, through
the review of regular reports, annual reviews, site visits, etc.
UHC received a finding of unmet because this was the second year that there were opportunities for improvement
identified in this area of review. As a result of the CY 2012 SPR finding, UHC was required to submit a CAP to
provide evidence of ongoing oversight and monitoring of delegated entities. The CAP was not fully implemented
and continuing opportunities for improvement exist. According to the Director of Marketing, routine monitoring of
delegated entities occurred informally through ad hoc meetings convened in response to identified issues. There was
no documentation of these meetings. There was no evidence of review of delegated vendors’ annual audit findings.

2014 Annual Technical Report
Delmarva Foundation
10
Evidence was provided supporting an annual credentialing audit; however, there was no evidence of a claims audit,
which is also a delegated activity.
In order to receive a finding of met in the CY 2014 SPR, UHC must provide ongoing evidence of routine monitoring
and oversight of each delegated entity that includes documented review of annual audit findings of delegated
activities and monitoring of any CAPs.
Component 3.3e - There is evidence of continuous and ongoing evaluation of delegated activities, including
review and approval of over and underutilization reports, where applicable.
UHC received a finding of unmet because the QMC did not review over and underutilization comparisons on an
annual basis.
In order to receive a finding of met in the CY 2014 SPR, UHC must provide evidence of review/approval of any
UM delegated entity’s over/under utilization report(s) by the appropriate committee at intervals consistent with the
MCO's policy.
Follow-up:
RHMD and UHC were required to submit CAPs for the above elements/components. Delmarva Foundation
reviewed and approved the submissions.
The approved CAPs will be reviewed during the CY 2014 SPR.

2014 Annual Technical Report
Delmarva Foundation
11
STANDARD 4: Credentialing and Recredentialing
Requirements: The QAP must contain all required provisions to determine whether physicians and other health
care professionals licensed by the State and under contract with the MCO are qualified to perform their services. The
MCO must have written policies and procedures for the credentialing process that govern the organization’s
credentialing and recredentialing. There is documentation that the MCO has the right to approve new providers and
sites and to terminate or suspend individual providers. The MCO may delegate credentialing/recredentialing
activities with a written description of the delegated activities, a description of the delegate’s accountability for
designated activities, and evidence that the delegate accomplished the credentialing activities. The credentialing
process must be ongoing and current. There must be evidence that the MCO requests information from recognized
monitoring organizations about the practitioner. The credentialing application must include information regarding
the use of illegal drugs, a history of loss of license and loss or limitation of privileges or disciplinary activity, and an
attestation to the correctness and completeness of the application. There must be evidence of an initial visit to each
potential PCP’s office with documentation of a review of the site and medical record keeping practices to ensure
compliance with the American’s with Disabilities Act and the MCO’s standards.
There must be evidence that recredentialing is performed at least every three years and includes a review of enrollee
complaints, results of quality reviews, hospital privileges, current licensure, and office site compliance with
Americans with Disabilities Act of 1990 (ADA) standards, if applicable.
Results:
The overall MD MCO Compliance Rate was 99% for CY 2013.
All MCOs met the minimum compliance threshold for this standard.
RHMD received a compliance rate of 98%, which exceeds its minimum compliance threshold of 80% for its
first review.
Findings: Overall, MCOs have appropriate policies and procedures in place to determine whether physicians and
other health care professionals, licensed by the State and under contract to the MCO, are qualified to perform their
services. Evidence in credentialing and recredentialing records demonstrated that those policies and procedures are
functioning effectively. There were issues identified with the recredentialing process over the past year which
represented the slight decline in the overall MCO compliance rate.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
12
STANDARD 5: Enrollee Rights
Requirements: The organization demonstrates a commitment to treating participants in a manner that acknowledges
their rights and responsibilities. The MCO must have a system linked to the QAP for resolving participants’
grievances. This system must meet all requirements in COMAR 10.09.71.02 and 10.09.71.04. Enrollee information
must be written to be readable and easily understood. This information must be available in the prevalent non-
English languages identified by the Department. The MCO must act to ensure that the confidentiality of specified
patient information and records are protected. The MCO must have written policies regarding the appropriate
treatment of minors. The MCO must, as a result of the enrollee satisfaction surveys, identify and investigate sources
of enrollee dissatisfaction, implement steps to follow-up on the findings, inform practitioners and providers of
assessment results, and reevaluate the effectiveness of the implementation steps at least quarterly. The MCO must
have systems in place to assure that new participants receive required information within established time frames.
Results:
The overall MD MCO Compliance Rate was 96% for CY 2013.
ACC, JAI, MPC, and MSFC met the minimum compliance threshold for this standard.
PPMCO, RHMD, and UHC were required to submit CAPs.
Findings: Overall, MCOs have policies and procedures in place that demonstrate their commitment to treating
members in a manner that acknowledges their rights and responsibilities. Evidence of enrollee information was
reviewed and found to be easily understood and written in Spanish as required by the Department.
Additionally, all MCOs provided evidence of their complaint, grievance, and appeals processes. However,
opportunities for improvement did exist regarding policies and procedures, complaints/grievances, and satisfaction
surveys.
MCO Opportunity/CAP Required
PPMCO Opportunities/CAPs:
Component 5.1d – The grievance policy and procedure describes the process for aggregation and analysis of
grievance data and the use of the data for QI. There is documented evidence that this process is in place and
is functioning.
PPMCO received a finding of partially met because its Member Complaint/Grievance Policy did not reflect the
correct committee reporting structure.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must revise the Member Complaint/Grievance
Policy to reflect the correct reporting structure.
Component 5.1f - There is complete documentation of the substance of the grievances and steps
taken.

2014 Annual Technical Report
Delmarva Foundation
13
PPMCO received a finding of partially met because after a review of 35 complaint/grievance records, it was found
that the documentation of the substance of the complaint/grievance in the electronic system, along with the letters to
members regarding the complaint/grievance and its resolution, was not complete in several records. Additionally,
the documentation in the complaint/grievance records did not match up to the dates noted in the system: start dates,
completion dates, dates on customer service call notes, and response letter dates.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must provide complete and clear documentation
of the substance of the grievances and steps taken in each record.
Component 5.1g – The MCO adheres to the time frames set forth in its policies and procedures for resolving
grievances.
PPMCO received a finding of unmet because a review of 35 complaint/grievance records found that the current
electronic system did not clearly track the dates of resolution activity for all records.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must adhere to the time frames set forth in its
policies and procedures for resolving grievances in all records.
RHMD Opportunities/CAPs:
Component 5.6a - Policies and procedures are in place that address the content of new enrollee packets of
information and specify the time frames for sending such information to the enrollee.
RHMD received a finding of partially met because the MCO does not have a formal written policy and procedure
that includes the content of new enrollee packets and the regulatory time frames for mailing such information to new
participants. Currently, welcome packet fulfillment reports are reviewed daily, along with the use of Health Risk
Assessments and Welcome Calls to confirm receipt of new enrollee packets.
In order to receive a finding of met in the CY 2014 SPR, RHMD must develop a policy and procedure that includes
the content of new enrollee packets and the regulatory time frames for mailing such information to new participants.
UHC Opportunities/CAPs:
Component 5.1g – The MCO adheres to the time frames set forth in its policies and procedures for resolving
grievances.
UHC received a finding of unmet because grievance records demonstrated that resolution letters were absent from
almost all case records due to staffing changes and training issues during 2013. Therefore, the reviewer was unable
to determine whether or not resolutions met the required time frames. UHC proactively developed a CAP prior to
the review to rectify the noncompliant situation, including a new tracking grid, implementation of weekly and
quarterly audits, and secured electronic record keeping. These activities will begin in February 2014.
In order to receive a finding of met in the CY 2014 SPR, UHC must adhere to the time frames set forth in the
MCO’s policies and procedures for resolving grievances.

2014 Annual Technical Report
Delmarva Foundation
14
Component 5.5c - As a result of the enrollee satisfaction surveys, the MCO informs practitioners and
providers of assessment results.
UHC received a finding of unmet because the MCO did not notify providers of the annual satisfaction survey
results. UHC would normally publish the results and analysis of the 2013 CAHPS® survey (measuring data from
CY 2012) in the fourth quarter 2013 provider newsletter.
In order to receive a finding of met in the CY 2014 SPR, UHC must inform practitioners and providers of
assessment results.
Follow-up:
PPMCO, RHMD, and UHC were required to submit CAPs for the above elements/components. Delmarva
Foundation reviewed and approved the submissions.
The approved CAPs will be reviewed for compliance during the CY 2014 SPR.

2014 Annual Technical Report
Delmarva Foundation
15
STANDARD 6: Availability and Accessibility
Requirements: The MCO must have established measurable standards for access and availability. The MCO must
have a process in place to assure MCO service, referrals to other health service providers, and accessibility and
availability of health care services. The MCO must have a list of providers that are currently accepting new
participants. The MCO must implement policies and procedures to assure that there is a system in place for notifying
participants of due dates for wellness services.
Results:
The overall MD MCO Compliance Rate was 96% for CY 2013.
ACC, JAI, MPC, MSFC, RHMD, and UHC met the minimum compliance threshold for this standard.
PPMCO was required to submit a CAP.
Findings: Overall, MCOs have established appropriate standards for ensuring access to care and have fully
implemented a system to monitor performance against these standards. All MCOs have current provider directories
that list providers that are currently accepting new participants along with websites and helplines that are easily
accessible to members as well. Each MCO has an effective system in place for notifying members of wellness
services. However, opportunities exist regarding consistency in policies and procedures and corrective action
planning.
MCO Opportunity/CAP Required
Component 6.1c - The MCO has established policies and procedures for the operations of its
customer/enrollee services and has developed standards/indicators to monitor, measure, and report on its
performance.
PPMCO received a finding of partially met because the MCO’s Access, Availability and Performance Standards
Policy cited performance standards that were inconsistent with their call center metrics. Additionally, the policy was
silent as to how to rectify ongoing noncompliance of call center performance.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must revise either the Access, Availability and
Performance Standards Policy or the Call Center Metric goal so that both documents state the same calls answered
within 30 seconds (availability rate for customer service representatives) goal. Currently, the policy states 90% and
the matrix spreadsheet states 85%.
Follow-up:
PPMCO was required to submit a CAP for the above element/component. Delmarva Foundation reviewed and
approved the submission.
The approved CAP will be reviewed for compliance during the CY 2014 SPR.

2014 Annual Technical Report
Delmarva Foundation
16
STANDARD 7: Utilization Review
Requirements: The MCO must have a comprehensive Utilization Management Program, monitored by the
governing body, and designed to evaluate systematically the use of services through the collection and analysis of
data in order to achieve overall improvement. The Utilization Management Plan must specify criteria for Utilization
Review/Management decisions. The written Utilization Management Plan must have mechanisms in place to detect
over utilization and underutilization of services. For MCOs with preauthorization or concurrent review programs,
the MCO must substantiate that: preauthorization, concurrent review, and appeal decisions are made and supervised
by appropriate qualified medical professionals; efforts are made to obtain all necessary information, including
pertinent clinical information, and to consult with the treating physician as appropriate; the reasons for decisions are
clearly documented and available to the enrollee; there are well publicized and readily available appeal mechanisms
for both providers and participants; preauthorization and concurrent review decisions are made in a timely manner
as specified by the State; appeal decisions are made in a timely manner as required by the exigencies of the
situation; and the MCO maintains policies and procedures pertaining to provider appeals as outlined in COMAR
10.09.71.03. Adverse determination letters must include a description of how to file an appeal and all other required
components. The MCO must also have policies, procedures, and reporting mechanisms in place to evaluate the
effects of the Utilization Management Program by using data on enrollee satisfaction, provider satisfaction, or other
appropriate measures.
Results:
The overall MD MCO Compliance Rate was 90% in CY 2013.
ACC, JAI, MPC, and MSFC met the minimum compliance threshold for this standard.
PPMCO, RHMD and UHC were required to submit CAPs.
Findings: Overall, MCOs have strong Utilization Management Plans that describe procedures to evaluate medical
necessity criteria used, information sources, procedures for training and evaluating staff, monitoring of the
timeliness and content of adverse determination notifications, and the processes used to review and approve the
provision of medical services. The MCOs provided evidence that qualified medical personnel supervise pre-
authorization and concurrent review decisions. The MCOs have implemented mechanisms to detect over and
underutilization of services. Overall, policies and procedures are in place for providers and participants to appeal
decisions. However, continued opportunities were present in the areas of monitoring compliance of UR decision.
MCO Opportunity/CAP Required
PPMCO Opportunities/CAPs:
Component 7.3a - Services provided must be reviewed for over and underutilization.
PPMCO received a finding of partially met because the Over and Under Utilization Policy outlines procedures for
monitoring of potential over and underutilization and development of interventions, as indicated. Monitoring is to
occur on a quarterly basis with results reported to the Quality Improvement Work Group (QIWG). Although it is
evident that the UM Close Committee was reviewing utilization trends for some inpatient services, this component
was only partially met as there was no evidence that the UM Close Committee reported results to the QIWG in a
manner consistent with the MCO's policy. Additionally, there was no evidence of follow-up on action items
requiring further investigation of identified trends to assess for over or under utilization.

2014 Annual Technical Report
Delmarva Foundation
17
In order to receive a finding of met in the CY 2014 SPR, PPMCO must provide evidence that the MCO is following
its policies for monitoring and reporting of potential over and underutilization. There must also be evidence of
follow-up on identified action plans requiring further investigation of potential over and underutilization.
Component 7.3b – Utilization review reports must provide the ability to identify problems and take the
appropriate corrective action.
PPMCO received a finding of unmet because there was no evidence that the MCO identified problems of
over/under utilization and implemented corrective action based upon review of QIWG meeting minutes from 2013.
The MCO did provide two examples of BH meeting minutes that primarily focused on the State-required SA
performance improvement project and noted that reports had been presented to the QIWG. Use of a State-required
performance improvement project does not meet the intent of this standard/component.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must provide evidence that the MCO takes
corrective action in response to identified over/under utilization problems as documented in the appropriate
committee meeting minutes.
Component 7.3c - Corrective measures implemented must be monitored.
PPMCO received a finding of unmet because there was no documentation in appropriate committee meeting
minutes that corrective measures to address over/under utilization were monitored in 2013.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must provide evidence that corrective measures
have been implemented to address over/under utilization problems are monitored by the appropriate committee.
Component 7.4c - The reasons for decisions are clearly documented and available to the enrollee.
PPMCO received a finding of partially met because a review of a sample of member adverse determination letters
demonstrated unclear language from the criteria used to make the determination included in the letters. For example,
the letters included the use of standard medical terminology such as "functional plateau" and "decline in speech
intelligibility," terms. These terms could not be easily understood and are inappropriate in a letter to a member.
In order to receive a finding of met in the CY 2014 SPR, PPMCO must document the reasons for decisions in
clearly understandable language for the member.
Component 7.4e - Preauthorization and concurrent review decisions are made in a timely manner as specified
by the State.
In response to the CY 2012 SPR findings, PPMCO was required to develop a CAP to demonstrate consistent
compliance with determination and notification time frames specified by the State. In CY 2013, continued
opportunities for improvement exist; therefore PPMCO received a finding of unmet in that component. The
Inpatient Preauthorizations document identified compliance with turnaround times by month throughout 2013.
Compliance exceeded the 95% threshold with the exception of June, which was slightly below at 94.8%.

2014 Annual Technical Report
Delmarva Foundation
18
In order to receive a finding of met in the CY 2014 SPR, PPMCO must consistently demonstrate compliance at the
95% threshold in response to State-required time frames for preauthorization determinations and adverse
determination notifications for medical, pharmacy, and SA.
Component 7.4f - Appeal decisions are made in a timely manner as required by the exigencies of the situation.
PPMCO received a finding of partially met because the MCO failed to meet the required resolution time frames
throughout 2013. The Appeals Process Management Team Report evidences tracking of compliance by month for
expedited pre-service, non-urgent pre-service, and post-service appeals. For expedited pre-service appeals,
compliance was consistently reported as 100%. For non-urgent pre-service appeals, compliance ranged from 91% to
100%, with four months out of compliance (June, July, August, and September).
In order to receive a finding of met in the CY 2014 SPR, PPMCO must consistently demonstrate compliance with
State-required time frames for appeal resolution.
RHMD Opportunities/CAPs:
Component 7.1a – The comprehensive Utilization Review Plan includes procedures to evaluate medical
necessity, criteria used, information sources, and the process used to review and approve the provision of
medical services.
RHMD received a finding of partially met because there was there was no description of the data and information
the MCO uses to make determinations regarding SA and there was no evidence that the UMP was approved by the
QIC in 2013.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of the processes the MCO
follows and the information it uses to make determinations in response to requests for preauthorization, concurrent,
and retrospective reviews for SA. This can be included in the UMP or in specific preauthorization, concurrent, and
retrospective review policies. Additionally, the UMP must be approved by the QIC annually.
Component 7.1b - The scope of the Utilization Review Plan includes a review of all covered services in all
settings, admissions in all settings, and collateral and ancillary services.
RHMD received a finding of unmet because the UMP did not state that its review activities included all covered
services in all settings, admissions in all settings, and collateral and ancillary services.
In order to receive a finding of met in the CY 2014 SPR, RHMD must ensure that the UMP explicitly includes the
scope of its review activities.
Component 7.1c - There is documentation that ensures that utilization determinations made by an individual
or entity are not directly influenced by financial incentive or compensation.

2014 Annual Technical Report
Delmarva Foundation
19
RHMD received a finding of partially met because there was no evidence that the Affirmative Statement regarding
UM decision making that is required at initial hire and annually thereafter was included in the December 2012
edition of the RHMD Provider Manual.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence that the MCO fully
implements its Affirmative Statement Policy, which requires inclusion of the affirmative statement in the Provider
Manual as well as annual publication in the provider newsletter.
Component 7.2d - There must be evidence that the criteria for UR/UM decisions are reviewed and updated
according to MCO policies and procedures.
RHMD received a finding of unmet because there was no evidence of Provider Advisory Committee review and
approval of Milliman Care Guidelines found, from the time the MCO commenced operations to the SPR review.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence of Provider Advisory
Committee annual review and approval of all medical necessity criteria used by the MCO, consistent with its policy.
Component 7.4c - The reasons for decisions are clearly documented and available to the enrollee.
RHMD received a finding of partially met because adverse determination letters did not consistently provide a clear,
full, complete explanation of the reason for the adverse determination in easily understandable language. Seven out
of the 30 letters reviewed (23%) provided an inadequate explanation for the reason for the adverse determination.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence that reasons for adverse
determinations are communicated in writing to the member and provide a clear, full, and complete explanation for
the decision in easily understandable language.
Component 7.4e - Preauthorization and concurrent review decisions are made in a timely manner as specified
by the State.
RHMD received a finding of unmet because there was no documented process that describes the methodology for
reporting compliance with preauthorization determination and adverse determination notification time frames.
However, compliance was tracked on a routine basis and reported to the QIC. Although sampling was used, the
required sample size calculator was not used to ensure a statistically valid sample size. Compliance with the 95%
threshold could, therefore, not be assessed.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence that the MCO has a
documented methodology for determining compliance with preauthorization determination and adverse
determination notification time frames consistent with state requirements, including use of the sample size calculator
to ensure a statistically valid sample size if the total population is not used.
Component 7.4g - The MCO maintains policies and procedures pertaining to provider appeals as outlined in
COMAR 10.09.71.03.

2014 Annual Technical Report
Delmarva Foundation
20
RHMD received a finding of partially met because the Provider Appeals Policy includes some, but not all,
requirements as outlined in COMAR 10.09.71.03.
In order to receive a finding of met in the CY 2014 SPR, RHMD must revise the Provider Appeal Policy to be
consistent with the requirements outlined in COMAR 10.09.71.03.
Element 7.5 - Adverse determination letters include a description of how to file an appeal and all
other required components.
RHMD received a finding of unmet because none of the sample adverse determination letters reviewed evidenced
that the PCP was copied, although there was evidence that the PCP was copied on the requesting provider letter. The
component that requires a clear, full, complete factual explanation for the reasons for denial, reduction or
termination in understandable language was not met in two (20%) of the 10 letters. Additionally, the component
requiring description of any additional information the MCO needs for reconsideration was stated as N/A in each of
these letters. Another 20 adverse determination letters were reviewed for these two components. Five of the 20
(25%) additional letters reviewed for compliance with these components were found to provide an inadequate
explanation of the reason for the adverse determination and were also lacking a description of additional information
needed for reconsideration.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence that the PCP is copied on
all member adverse determination letters. Additionally, all adverse determination letters must include a clear, full,
complete factual explanation of the reasons for denial, reduction, or termination in understandable language and
describe any additional information the MCO needs for reconsideration.
Component 7.6a - The MCO has a process in place to evaluate the effects of the UR program by
using enrollee satisfaction, provider satisfaction, and/or other appropriate measures.
RHMD received a finding of unmet because there was no policy that addresses member and provider satisfaction
with the UMP.
In order to receive a finding of met in the CY 2014 SPR, RHMD must provide evidence that the MCO has
developed a comprehensive policy that addresses the process for evaluating member and provider satisfaction with
the UMP on an annual basis.
UHC Opportunities/CAPs:
Component 7.4c - The reasons for decisions are clearly documented and available to the enrollee.
UHC received a finding of unmet as it has not met compliance with State required time frames for determinations
and notifications for preauthorization requests since 2007 with the exception of 2011 when the component was
scored as baseline. The CAPs that have been implemented to date have left continued opportunities for
improvement.
In order to receive a finding of met in the CY 2014 SPR, UHC must demonstrate consistent tracking and compliance
with State-required time frames for determinations and notifications for preauthorization requests for medical,
pharmacy, and SA services.

2014 Annual Technical Report
Delmarva Foundation
21
Component 7.4f – Appeal decisions are made in a timely manner as required by the exigencies of the
situation.
UHC received a finding of unmet because as a result of the CY 2011 and 2012 SPR findings, the MCO was required
to submit a CAP each year to address compliance with regulatory time frames for appeal processing on a consistent
basis. These CAPs were partially implemented and continued opportunities for improvement exist in demonstrating
routine compliance with State-required time frames.
In order to receive a finding of met in the CY 2014 SPR, there must be evidence that the MCO consistently meets
the State required resolution time frames for all medical, pharmacy, and SA appeals.
Element 7.5 - Adverse determination letters include a description of how to file an appeal and all other
required components.
UHC received a finding of partially met because in seven of the 10 denial letters reviewed, the requesting provider
rather than the PCP was copied at the bottom of the letter.
In order to receive a finding of met in the CY 2014 SPR, UHC must consistently demonstrate inclusion of all
required components in adverse determination letters. Specifically, all letters must include evidence that a copy was
sent to the member's PCP.
Component 7.6c - The MCO demonstrates review of the data on enrollee satisfaction, provider satisfaction,
and/or other appropriate data by the appropriate oversight committee.
UHC received a finding of partially met because there was no evidence of review of 2013 Provider Satisfaction
Survey results related to UMP satisfaction by the appropriate oversight committee.
In order to receive a finding of met in the CY 2014 SPR, UHC must provide evidence of committee review of 2014
Provider Satisfaction Survey results, specifically in relation to UM processes. Additionally, there needs to be a more
detailed review of CAHPS
®
satisfaction results relating specifically to the UMP.
Component 7.6d - The MCO acts upon identified issues as a result of the review of the data.
UHC received a finding of partially met because there were no specific interventions related to specific UM-related
Provider Satisfaction issues.
In order to receive a finding of met in the CY 2014 SPR, UHC must provide evidence that the MCO acts upon
identified issues in response to both the 2014 CAHPS
®
and the provider satisfaction surveys, specifically relating to
the UMP.

2014 Annual Technical Report
Delmarva Foundation
22
Follow-up:
PPMCO, RHMD and UHC were required to submit CAPs for the above elements/components. Delmarva
Foundation reviewed and approved the submissions.
The approved CAPs will be reviewed during the CY 2014 SPR.

2014 Annual Technical Report
Delmarva Foundation
23
STANDARD 8: Continuity of Care
Requirements: The MCO must put a basic system in place that promotes continuity of care and case management.
Participants with special needs and/or those with complex health care needs must have access to case management
according to established criteria and must receive the appropriate services. The MCO must have policies and
procedures in place to coordinate care with other appropriate agencies or institutions (e.g., school health programs).
The MCO must monitor continuity of care across all services and treatment modalities. This must include an
ongoing analysis of referral patterns and the demonstration of continuity of individual cases (timeliness and follow-
up of referrals). The MCO must ensure appropriate initiation of care based on the results of the Health Risk
Assessment (HRA) data supplied to the MCO. This must include a process for gathering HRA data, an ongoing
analysis, and a process that calls for appropriate follow-up on results of the analysis.
Results:
The overall MD MCO Compliance Rate was 100% for CY 2013.
All MCOs met the minimum compliance threshold for this standard.
Findings: Overall, the findings, conclusions, actions taken, and results of actions taken as a result of the MCO's
quality assurance activities are documented and reported to appropriate individuals within the MCO’s structure and
through the established quality assurance channels. All MCOs have allocated resources, such as automated tracking
methodologies, that facilitate communication between members, PCPs, other health care professionals, and the
MCO’s care coordinators.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
24
STANDARD 9: Health Education Plan Review
Requirements: The MCO must have a comprehensive educational plan and have mechanisms in place to oversee
that appropriate health education activities are provided or are available at each provider site. The educational
activities must include health education on subjects that affect the health status of the enrollee population. The
Health Education Plan must incorporate activities that address needs identified through the analysis of enrollee data
and have a written methodology for an annual evaluation of the impact of the Health Education Plan on process
and/or outcome measures, such as emergency room (ER) utilization, avoidable hospital admissions, utilization of
preventive services, and clinical measures. The Health Education Plan must provide for qualified staff or contract
with external organizations to develop and conduct educational sessions to support identified needs of the members.
The Health Education Plan must contain a provision addressing how the MCO will notify providers of the
availability and contact information for accessing a health educator/educational program for member referrals. The
MCO must have mechanisms in place to identify participants in special need of educational efforts. Documentation
must support that these mechanisms are in place and functioning. The MCO must make the education program
available to the enrollee population and demonstrate that participants have attended.
Results:
All MCOs (except for RHMD) were exempt from this standard. This standard was exempt as each MCO has
received compliance ratings of 100% for the past three consecutive years.
RHMD received 88%, which met the minimum compliance threshold for this standard.
Findings: This area of review was exempt for all MCOs except for RHMD. This was RHMD’s first review of their
Health Education Plan. It was found to be comprehensive and include policies and procedures for internal staff
education, provider education and continuing education units, and enrollee health education.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
25
STANDARD 10: Outreach Plan Review
Requirements: The MCO must have developed a comprehensive written Outreach Plan to assist participants in
overcoming barriers in accessing health care services. The Outreach Plan must adequately describe the populations
to be served, activities to be conducted, and the monitoring of those activities. There must be evidence that the MCO
has implemented the Outreach Plan, appropriately identified the populations, monitored outreach activities, and
made modifications as appropriate.
Results:
All MCOs (except for RHMD) were exempt from this standard. This standard was exempt as each MCO has
received compliance ratings of 100% for the past three consecutive years.
RHMD received 93%, which met the minimum compliance threshold for this standard.
Findings: This area of review was exempt for all MCOs except for RHMD. This was RHMD’s first review of their
Outreach Plan. Overall, it was found to have adequately described their populations served, an assessment of
common health problems, and barriers to outreach within the MCO’s membership. RHMD described the
organizational capacity to provide both broad-based and enrollee specific outreach in the plan. The unique features
of the MCO’s enrollee education initiatives, community partnerships, and the roles of the provider network and local
health departments were also included in the Outreach Plan. Appropriate supporting evidence of the outreach
activities was also provided.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.

2014 Annual Technical Report
Delmarva Foundation
26
STANDARD 11: Fraud and Abuse
Requirements: The MCO maintains a Medicaid Managed Care Compliance Program that outlines its internal
processes for adherence to all applicable Federal and State laws and regulations, with an emphasis on preventing
fraud and abuse. The program also includes guidelines for defining failure to comply with these standards.
Results:
The overall MD MCO Compliance Rate was 98% for CY 2013.
All MCOs met the minimum compliance threshold for this standard.
RHMD received a compliance rate of 89%, which exceeds its minimum compliance threshold of 80% for its
first review.
Findings: All MCOs were found to have comprehensive compliance programs designed to support organizational
standards of integrity in identifying and addressing inappropriate and unlawful conduct, fraudulent activities, and
abusive patterns. Fraud and abuse plans articulated the organization’s commitment to comply with all applicable
Federal and State laws, regulations, and standards. The MCO also demonstrated procedures for timely investigation,
and tracking of reported suspected incidence of fraud and abuse. There were designated Compliance Officers and
active Compliance Committees. All staff, subcontractors, and participants were clearly communicated to regarding
disciplinary guidelines and sanctioning of fraud and abuse. Additionally, the MCO demonstrated it has a process
which allows employees, subcontractors, and participants to report fraud and abuse without the fear of reprisal.
MCO Opportunity/CAP Required
No CAPs were required.
Follow-up: No follow-up is required.
Conclusions
All MCOs have demonstrated the ability to design and implement effective QA systems. The CY 2013 review
provided evidence of the continuing progression of the HealthChoice MCOs as each MCO demonstrated
their ability to ensure the delivery of quality health care for their participants.
Maryland has set high standards for MCO QA systems. In general, HealthChoice MCOs continue to make
improvements in their QA monitoring policies, procedures, and processes while working to provide the
appropriate levels and types of health care services to managed care participants. This is evident in the
comparison of annual SPR results demonstrated throughout the history of the HealthChoice Program.

2014 Annual Technical Report
Delmarva Foundation
27
SECTION II
Value Based Purchasing
Introduction
DHMH began working with the Center for Health Care Strategies in 1999 to develop a Value Based
Purchasing Initiative (VBPI) for HealthChoice, Maryland’s Medicaid managed care program. VBP improves
quality by awarding business and incentives to contractors based on their performance along a range of
dimensions. The goal of Maryland’s purchasing strategy is to achieve better participant health through
improved MCO performance. Appropriate service delivery is promoted by aligning MCO incentives with the
provision of high-quality care, increased access, and administrative efficiency. Maryland’s VBP strategy aims
to better coordinate a variety of quality improvement efforts toward a shared set of priorities that focus on
the core populations served by HealthChoice. In addition, the state’s strategy meets the requirements of the
Balanced Budget Act of 1997 (BBA).
Delmarva Foundation and HealthcareData Company, LLC (HDC), a NCQA-Licensed Organization, were
contracted by DHMH to perform a validation of the CY 2013 VBP measurement data. Validation is the
process by which an independent entity evaluates the accuracy of reported performance measure data by or
on behalf of, another entity and determines the extent to which specific performance measures calculated by
an entity (or one acting on behalf of another) followed established calculation specifications. A validation (or
audit) determination is assigned to each measure, indicating whether the measure and its result is fully
compliant, substantially compliant, or not valid. DHMH contracted with HDC to perform the validation of
HEDIS
®
measures for the HealthChoice MCOs. HDC performed the validation of the HEDIS
®
-based VBP
measurement data for all seven of the HealthChoice MCOs using the NCQA’s HEDIS
®
Volume 5: HEDIS
®
Compliance Audit: Standards, Policies, and Procedures.
Performance Measure Selection Process
The HealthChoice VBP program emphasizes continuous quality improvement and evidence-based medicine,
making it consistent with trends in the larger health care market. The program increases the comparability of
Maryland’s performance to that of other states, enabling the sharing of best practices. In addition,
performance evaluation based on administrative and encounter data rather than on the review of a small
sample of medical records means that the quality indicators are representative of more participants.
In its performance measure selection process, DHMH solicits input from stakeholders, including MCOs and
the Maryland Medicaid Advisory Committee. Together, they identified legislative priorities in selecting the
performance measures. Measures may be added or removed, based upon evolving DHMH priorities and
participant health care needs.

2014 Annual Technical Report
Delmarva Foundation
28
The measures address several aspects of plan performance which fall into one of the following three
categories:
Access to Care: The ability of patients to get access to needed services.
Quality of Care: The ability to deliver services to improve health outcomes.
Timeliness of Care: The ability of patients to get needed services in a timely manner.
DHMH selects measures that are:
1. relevant to the core populations served by HealthChoice, including children, pregnant women, special
needs children, adults with disabilities, and adults with chronic conditions;
2. prevention-oriented and associated with improved outcomes;
3. measurable with available data;
4. comparable to national performance measures for benchmarking;
5. consistent with how CMS is developing a national set of performance measures for Medicaid MCOs; and
6. possible for MCOs to affect change.
Value Based Purchasing Validation
Several sources of measures (Table 3) are included in the CY 2013 VBP program. They are chosen from
NCQA’s HEDIS
®
data set, encounter data, and data supplied by the HealthChoice MCOs, and subsequently
validated by Delmarva Foundation. The measure type and the presence of an existing audit or validation
process determined the validation activities undertaken.
Table 3. CY 2013 VBP Measures
Performance Measure
HEDIS
®
Domain
Measure
Reporting
Entity
Adolescent Well Care
Use of Services
HEDIS
®
MCO
Ambulatory Care Services for SSI Adults Ages 21-64
Access to Care
Encounter Data
DHMH
Ambulatory Care Services for SSI Children Ages 0-20
Access to Care
Encounter Data
DHMH
Cervical Cancer Screening for Women Ages 21–64
Effectiveness of Care
HEDIS
®
MCO
Childhood Immunization Status (Combo 3)
Effectiveness of Care
HEDIS
®
MCO
Eye Exams for Diabetics Ages 18-75
Effectiveness of Care
HEDIS
®
MCO
Lead Screenings for Children Ages 12–23 Months
Effectiveness of Care
Encounter , Lead
Registry, & Fee For
Service Data
DHMH
Postpartum Care
Access to Care
HEDIS
®
MCO
Immunizations for Adolescents
Effectiveness of Care
HEDIS
®
MCO
Well Child Visits for Children Ages 3–6
Use of Services
HEDIS
®
MCO
HEDIS
®
Measure Validation
HealthChoice MCOs are required to produce and report audited HEDIS
®
data under COMAR
10.09.65.03.B(2). Seven of the CY 2013 VBP measures are HEDIS
®
measures and are validated under the

2014 Annual Technical Report
Delmarva Foundation
29
provisions of the HEDIS
®
Compliance Audit. The goal of the HEDIS
®
audit is to ensure accurate, reliable,
and publicly reportable data.
HDC completed the HEDIS
®
audits in three phases: offsite, onsite, and post onsite (reporting). The offsite
audit phase includes a review of each MCO’s HEDIS
®
Record of Administration, Data Management and
Processes (Roadmap). The Roadmap is used to supply information about an MCO’s data systems and
HEDIS
®
data reporting structure and processes. Other activities of the offsite audit process include the
selection of HEDIS
®
measures to audit in detail (results are then extrapolated to the rest of the HEDIS
®
measures), investigation of measure rotation strategies, and validation of the medical record review process by
the certified audit firm.
Prior to the onsite phase, HDC holds annual auditor conference calls with all MCOs for the purpose of
addressing any NCQA changes or updates to the audit guidelines. HDC also responds to each MCO’s
questions.
During the onsite phase, auditors investigate issues identified in the Roadmap and observe the systems used
to collect and produce HEDIS
®
data. The audit team interviews MCO staff; reviews MCO information
system structure, protocols, and processes; and reviews MCO measure-specific data collection processes with
the MCO staff.
The post onsite and reporting phase of the HEDIS
®
Compliance Audit includes the issuance of a follow-up
letter to the MCO that lists any items the auditors still require to complete the audit; a list of corrective
actions for problems found in the Roadmap or onsite, as well as the necessary completion dates; and
preliminary audit findings specifically indicating the measures at risk for a Not Report designation. When the
MCO has provided all requested documents and performed the recommended corrective actions, the auditor
completes a final audit report and assigns audit designations indicating the suitability of measures for public
reporting. The four possible audit designations are explained in Table 4. The final activity of the post onsite
phase of the audit consists of the MCO submitting data to NCQA, using NCQA’s Interactive Data
Submission System (IDSS).

2014 Annual Technical Report
Delmarva Foundation
30
Table 4. HEDIS
®
Compliance Audit Designations
Audit Findings
Description
Rate/Result
The MCO produced a reportable rate or count for the
measure and followed the HEDIS
®
technical specifications.
Reportable Measure
0-XXX
The MCO followed the specifications but the denominator
was too small to report a valid rate.
Not Applicable
NA
The MCO did not offer the health benefits required by
the measure (e.g., Mental Health/Chemical Dependency).
No Benefit
NB
The MCO calculated the measure but the rate
was materially biased, or
the MCO chose not to report the measure.
Not Report
NR
In order to avoid duplicating efforts and placing undue administrative burden on the HealthChoice MCOs,
DHMH used seven of the HEDIS
®
audit measure determinations as VBP measure determinations. The
HEDIS
®
measures in the VBP program are:
Well-Child Visits in the Third, Fourth, Fifth, and Sixth Years of Life
Adolescent Well Care
Childhood Immunization Status (Combo 3)
Cervical Cancer Screening
Postpartum Care
Immunizations for Adolescents
Comprehensive Diabetes Care (eye exam indicator only)
EQRO’s Data Measure Validation
Three CY 2013 VBP measures were calculated by The Hilltop Institute of University of Maryland Baltimore
County (Hilltop) for DHMH, using encounter data submitted by the MCOs for January 1 – December 31,
2012, Maryland Department of the Environment’s Lead Registry data, and Fee-for-Service data. The
measures calculated utilizing encounter data are:
Ambulatory Care Services for SSI Adults
Ambulatory Care Services for SSI Children
Lead Screenings for Children Ages 12–23 Months
Delmarva Foundation validated the measurement data for each of the above VBP measures, including the
specifications for each encounter data-based measure, source code to determine algorithmic compliance with
the measure specifications, information regarding the encounter data processing system, and analysis of the
encounter data process. Clarifications and corrections to source code were conducted to ensure algorithmic
compliance with VBP measure specifications.
Validation determinations were used to characterize the findings of the EQRO. Table 5 indicates the possible
determinations of the EQRO-validated measures. To validate the rates calculated, two analysts and an analytic

2014 Annual Technical Report
Delmarva Foundation
31
scientist with the Delmarva Foundation reviewed and approved the measure creation process and source
code.
Table 5. Possible Validation Findings for EQRO-Validated Measures (encounter data)
Validation Determination
Definition
Fully Compliant (FC)
Measure was fully compliant with State specifications and reportable.
Substantially Compliant (SC)
Measure was substantially compliant with State specifications and had only
minor deviations that did not significantly bias the reported rate.
Not Valid (NV)
Measure deviated from state specifications such that the reported rate was
significantly biased. This designation is also assigned to measures where
no rate was reported, although reporting of the rate was required.
Not Applicable (NA)
Measure was not reported because the entity did not have any Medicaid
participants that qualified for the denominator.
Validation Results
Validation of the methodologies, criteria, and processes employed in creating the VBP measures results in a
determination of the effect of bias on the resulting statistic. Validation determinations by HDC are reported
using the audit designations and rationales outlined by NCQA as part of the HEDIS
®
Compliance Audit.
All of the VBP measures audited by HDC were determined to be reportable for all MCOs.
Table 6 shows the results of the EQRO-led validation activities related to the VBP measures. DHMH was
responsible for producing these VBP measures at the MCO level and working with the EQRO to validate the
measurement data. During the validation process undertaken by Delmarva Foundation, no issues were
identified that could have introduced bias to the resulting statistics.
Table 6. EQRO VBP Measure Validation Determinations
Measure
Validation Determinations
Ambulatory Care Services for SSI Adults
Fully Compliant
Ambulatory Care Services for SSI Children
Fully Compliant
Lead Screenings for Children Ages 12–23 Months
Fully Compliant

2014 Annual Technical Report
Delmarva Foundation
32
CY 2013 VBP Incentive/Disincentive Target Setting Methodology
The Hilltop Institute of University of Maryland Baltimore County (Hilltop) developed a target setting
methodology at the request of DHMH for VBP.
The incentive target is calculated as follows:
Determine the mean of the highest score for the measure in CY 2011 and the overall average of all
MCOs
Add 15 percent of the difference between the new mean determined above and 100 percent
The disincentive target is calculated as follows:
Determine the mean of the highest score for the measure in CY 2011 and the overall average of all
MCOs
Subtract 15 percent of the difference between the new mean determined above and 100 percent
The neutral range includes all scores following between the incentive and disincentive targets.
The above target setting methodology was used to calculated all targets, except the following:
The CY 2013 calculated ranges for the Cervical Cancer Screening for ages 21-64 and Postpartum Care
measures were lower than the CY 2012 ranges, therefore the CY 2012 ranges were used.
The CY 2013 calculated targets for Adolescent Well Care, Diabetic Eye Exams, and Well Child 3-6 were
greater than the HEDIS national 90th percentile, therefore the CY 2012 targets were used.

2014 Annual Technical Report
Delmarva Foundation
33
Table 7 shows the CY 2013 VBP measures and their targets.
Table 7. CY 2013 VBP Measures
Performance Measure
Data Source
2013 Target
Adolescent Well Care:
% of adolescents ages 12-21 (enrolled 320 or more days) receiving at
least one comprehensive well-care visit with a PCP or an OB/GYN
practitioner during the measurement year
HEDIS
®
Incentive: ≥ 77%
Neutral: 68%–76%
Disincentive: ≤ 67%
Ambulatory Care Services for SSI Adults Ages 21–64 Years:
% of SSI adults (enrolled 320 or more days) receiving at least one
ambulatory care service during the measurement year
Encounter Data
Incentive: ≥ 86%
Neutral: 82%–85%
Disincentive: ≤ 81%
Ambulatory Care Services for SSI Children Ages 0–20 Years:
% of SSI children (enrolled 320 or more days) receiving at least one
ambulatory care service during the measurement year
Encounter Data
Incentive: ≥ 83%
Neutral: 78%–82%
Disincentive: ≤ 77%
Cervical Cancer Screening for Women Ages 21–64 Years:
% of women ages 21–64 (continuously enrolled during reporting year)
receiving at least one PAP test during the last 3 years, consistent with U.S.
Preventive Services Task Force recommendations
HEDIS
®
Incentive: ≥ 80%
Neutral: 74%–79%
Disincentive: ≤ 73%
Childhood Immunization Status (Combo 3):
% of children who turned 2 years old during the measurement year who
were continuously enrolled for 12 months immediately preceding their
second birthday and who had 4 DTaP, 3 IPV, 1 MMR, 2 H influenza type B,
3 hepatitis B, 1 chicken pox vaccine (VZV), and pneumococcal conjugate by
the time period specified and by the child’s second birthday
HEDIS
®
Incentive: ≥ 86%
Neutral: 82%–85%
Disincentive: ≤81%
Eye Exams for Diabetics:
% of diabetics ages 18-75 (continuously enrolled during measurement
year) receiving a retinal or dilated eye exam during the measurement year,
consistent with American Diabetes Association recommendations
HEDIS
®
Incentive: ≥ 80%
Neutral: 71%–79%
Disincentive: ≤ 70%
Lead Screenings for Children Ages 12–23 Months:
% of children ages 12–23 months (enrolled 90 or more days) who receive a
lead test during the current or prior calendar year
Lead Registry,
Encounter & Fee
for Service Data
Incentive: ≥ 72%
Neutral: 63%–71%
Disincentive: ≤ 62%
Postpartum Care:
% of deliveries that had a postpartum visit on or
between 21 and 56 days after delivery
HEDIS
®
Incentive: ≥ 78%
Neutral: 72%–77%
Disincentive: ≤ 71%
Immunizations for Adolescents:
% of adolescents 13 years of age during the measurement year who had
one dose of meningococcal vaccine and either one Tdap or Td vaccine by
their 13
th
birthday
HEDIS
®
Incentive: ≥ 71%
Neutral: 61%–70%
Disincentive: ≤ 60%
Well-Child Visits for Children Ages 3 – 6 Years:
% of children ages 3–6 (enrolled 320 or more days) receiving at least one
well-child visit during the measurement year, consistent with American
Academy of Pediatrics & EPSDT recommended number of visits
HEDIS
®
Incentive: ≥ 89%
Neutral: 84%–88%
Disincentive: ≤ 83%

2014 Annual Technical Report
Delmarva Foundation
34
2013 Value Based Purchasing Results
The CY 2013 performance results presented in Table 8 were validated by Delmarva Foundation and
DHMH’s contracted HEDIS
®
Compliance Audit™ firm, HDC. The contractors determined the validity and
the accuracy of the performance measure results. All measures were calculated in a manner that did not
introduce bias, allowing the results to be used for public reporting and the VBP program. In CY 2013, six
HealthChoice MCOs qualified to participate in the initiative:
AMERIGROUP Community Care (ACC)
MedStar Family Choice, Inc. (MSFC)
Jai Medical Systems (JMS)
Priority Partners (PPMCO)
Maryland Physicians Care (MPC)
UnitedHealthcare (UHC)
Table 8. MCO CY 2013 VBP Performance Summary
Performance Measure
CY 2013
Target
ACC
JMS
MPC
MSFC
PPMCO
UHC
Incentive (I); Neutral (N); Disincentive (D)
Adolescent
Well Care
Incentive: ≥ 77%
Neutral: 68%–76%
Disincentive: ≤ 67%
68%
(N)
77%
(I)
69%
(N)
68%
(N)
62%
(D)
61%
(D)
Ambulatory Care Services
for SSI Adults
Incentive: ≥ 86%
Neutral: 82%–85%
Disincentive: ≤ 81%
81%
(D)
85%
(N)
84%
(N)
83%
(N)
84%
(N)
82%
(N)
Ambulatory Care Services
for SSI Children
Incentive: ≥ 83%
Neutral: 78%–82%
Disincentive: ≤ 77%
80%
(N)
86%
(I)
84%
(I)
81%
(N)
83%
(I)
77%
(D)
Cervical Cancer Screening
for Women Ages
21–64
Incentive: ≥ 80%
Neutral: 74%–79%
Disincentive: ≤ 73%
80%
(I)
80%
(I)
80%
(I)
74%
(N)
76%
(N)
63%
(D)
Childhood Immunization
Status—Combo 3
Incentive: ≥ 86%
Neutral: 82%–85%
Disincentive: ≤81%
78%
(D)
86%
(I)
72%
(D)
86%
(I)
81%
(D)
71%
(D)
Eye Exams for Diabetics
Ages 18-75
Incentive: ≥ 80%
Neutral: 71%–79%
Disincentive: ≤ 70%
65%
(D)
80%
(I)
72%
(N)
71%
(N)
71%
(N)
57%
(D)
Lead Screenings for
Children Ages 12–23
Months
Incentive: ≥ 72%
Neutral: 63%–71%
Disincentive: ≤ 62%
63%
(N)
79%
(I)
58%
(D)
63%
(N)
57%
(D)
53%
(D)
Postpartum
Care
Incentive: ≥ 78%
Neutral: 72%–77%
Disincentive: ≤ 71%
72%
(N)
79%
(I)
72%
(N)
72%
(N)
76%
(N)
64%
(D)
Immunizations for
Adolescents
Incentive: ≥ 71%
Neutral: 61%–70%
Disincentive: ≤ 60%
69%
(N)
76%
(I)
63%
(N)
71%
(I)
75%
(I)
63%
(N)
Well-Child Visits for Children
Ages 3–6
Incentive: ≥ 89%
Neutral: 84%–88%
Disincentive: ≤ 83%
84%
(N)
89%
(I)
89%
(I)
84%
(N)
84%
(N)
75%
(D)
™
NCQA HEDIS
®
Compliance Audit™ is a trademark of the National Committee for Quality Assurance.

2014 Annual Technical Report
Delmarva Foundation
35
2013 VBP Financial Incentive and Disincentive Methodology
As described in COMAR 10.09.65.03, DHMH uses financial incentives and disincentives to promote
performance improvement. There are three levels of performance for all measures: incentive, neutral and
disincentive. Financial incentives are earned when performance meets or exceeds the incentive target for a
measure. Conversely, disincentives are assessed when performance is at or below the minimum target. All
measures are evaluated separately and are of equal weight in the methodology. For any measure that the
MCO does not meet the minimum target, a disincentive of 1/10 of 1 percent of the total capitation amount
paid to the MCO during the measurement year shall be collected. For any measure that the MCO meets or
exceeds the incentive target, the MCO shall be paid an incentive payment of up to 1/10 of 1 percent of the
total capitation amount paid to the MCO during the measurement year. The amounts are calculated for each
measure and the total incentive payments made to the MCOs each year may not exceed the total amount of
disincentives collected from the MCOs in the same year plus any additional funds allocated by the DHMH
for a quality initiative. MCOs’ CY 2013 performance is shown in Table 9.
Table 9. MCO CY 2013 VBP Incentive/Disincentive Amounts
Performance
Measure
MCO
ACC
JMS
MPC
MSFC
PPMCO
UHC
Adolescent Well Care
$0
$87,967.11
$0
$0
($847,113.81)
($579,128.97)
Ambulatory Care
Services for SSI Adults
($704,655.35)
$0
$0
$0
$0
$0
Ambulatory Care
Services for SSI
Children
$0
$87,967.11
$668,670.79
$0
$847,113.81
($579,128.97)
Cervical Cancer
Screening for Women
Ages 21–64
$704,655.35
$87,967.11
$668,670.79
$0
$0
($579,128.97)
Childhood
Immunization
Status—Combo 3
($704,655.35)
$87,967.11
($668,670.79)
$138,043.09
($847,113.81)
($579,128.97)
Eye Exams for
Diabetics
Ages 18-75
($704,655.35)
$87,967.11
$0
$0
$0
($579,128.97)
Lead Screenings for
Children Ages
12–23 Months
$0
$87,967.11
($668,670.79)
$0
($847,113.81)
($579,128.97)
Postpartum Care
$0
$87,967.11
$0
$0
$0
($579,128.97)
Immunizations for
Adolescent
$0
$87,967.11
$0
$138,043.09
$847,113.81
$0
Well-Child Visits for
Children Ages 3–6
$0
$87,967.11
$668,670.79
$0
$0
($579,128.97)
Total Incentive/
Disincentive Amount
($1,409,310.70)
$791,703.99
$668,670.79
$276,086.18
($847,113.81)
($4,633,031.76)

2014 Annual Technical Report
Delmarva Foundation
36
SECTION III
Performance Improvement Projects
Introduction
COMAR 10.09.65.03 requires that all HealthChoice MCOs conduct PIPs that focus on clinical or nonclinical
areas. As the EQRO, Delmarva Foundation is responsible for evaluating the two PIPs from each of the
HealthChoice MCOs according to CMS’ External Quality Review Protocol 3: Validating Performance Improvement
Projects. The PIPs are designed to achieve, through ongoing measurements and interventions, significant
improvement sustained over time in clinical care or non-clinical care areas that are expected to have a
favorable effect on health outcomes. The PIPs include measurements of performance using objective quality
indicators, the implementation of system interventions to achieve improvement in quality, evaluation of the
effectiveness of the interventions, and planning and initiation of activities for increasing or sustaining
improvement.
In addition to improving the quality, access, or timeliness of service delivery, the process of completing a PIP
functions as a learning opportunity for the MCO. The processes and skills required in PIPs, such as indicator
development, root cause analysis, and intervention development, are transferable to other projects that can
lead to improvement in other health areas.
As designated by DHMH, six MCOs conducted PIPs in CY 2013 for submission in 2014:
AMERIGROUP Community Care (ACC)
MedStar Family Choice, Inc. (MSFC)
Jai Medical Systems (JMS)
Priority Partners (PPMCO)
Maryland Physicians Care (MPC)
UnitedHealthcare (UHC)
The Adolescent Well Care PIP continued and a new Controlling High Blood Pressure PIP began in CY 2013.
Six MCOs submitted PIPs in September 2014, which included CY 2013 data and results. Since RHMD had
not been operating a full year in CY 2013, they were unable to provide the sufficient data to participate in the
PIPs.
Topics Selected
DHMH initiated the Adolescent Well Care PIP in March 2012 using HEDIS
®
2012 measurement rates as the
baseline measurement for MCOs in developing interventions due in fall 2012. The measure seeks to increase
the percentage of adolescents 12-21 years of age in receiving at least one comprehensive well-care visit with a
PCP or OB/GYN practitioner during the measurement year. Maryland’s Early and Periodic Screening,
Diagnosis, and Treatment (EPSDT) Medical Record Review program measures health and developmental
history; comprehensive physical exam; laboratory tests/at-risk screening; immunizations; and health

2014 Annual Technical Report
Delmarva Foundation
37
education and anticipatory guidance for children and adolescents through age 20. The EPSDT 12-20 year age
group consistently scores lower than the other four age groups in each of these categories. In addition, the
underutilization of an adolescent well-care visit yields missed opportunities for prevention, early detection,
and treatment; therefore, increasing routine adolescent utilization is an important health care objective for the
Department.
DHMH initiated the Controlling High Blood Pressure PIP in March 2014 using HEDIS
®
2014 measurement
rates as the baseline measurement for MCOs in developing interventions due in fall 2014. The measure seeks
to increase the percentage of members 18–85 years of age who had a diagnosis of hypertension and whose
blood pressure was adequately controlled during the measurement year.
Delmarva Foundation was responsible for providing technical assistance, validation of results, education, and
oversight of the MCOs’ PIPs. All PIP submissions were made using an approved project submission tool.
Methodology
The guidelines utilized for PIP review activities were CMS’ External Quality Review Protocol 3: Validating
Performance Improvement Projects (PIPs). The protocol assists in evaluating whether or not the PIP was designed,
conducted, and reported in a sound manner and the degree of confidence a state agency could have in the
reported results.
Each MCO was required to provide the study framework and project description for each PIP. This
information was reviewed to ensure that each MCO was using relevant and valid study techniques. The
MCOs were required to provide annual PIP submissions in September 2014. The submissions included
results of measurement activities, a status report of intervention implementations, analysis of the
measurement results using the defined data analysis plan, as well as information concerning any modifications
to (or removal of) intervention strategies that may not be yielding anticipated improvement. If an MCO
decided to modify other portions of the project, updates to the submissions were permitted in consultation
with Delmarva Foundation and the Department.
Reviewers evaluated each project submitted using a standard validation tool that employed the CMS
validation methodology, which included assessing each project in the following ten critical areas:
Step 1: Review of the selected study topics.
Step 2: Review of the study questions.
Step 3: Review of the selected study indicator(s).
Step 4: Review of the identified study population.
Step 5: Review of sampling methods.

2014 Annual Technical Report
Delmarva Foundation
38
Step 6: Review of the MCO’s data collection procedures.
Step 7: Assessment of the MCO’s improvement strategies.
Step 8: Review of data analysis and interpretation of study results.
Step 9: Assessment of the likelihood that reported improvement is real improvement.
Step 10: Assessment of whether the MCO has sustained its documented improvement.
As Delmarva Foundation staff conducted the review, each of the components within a step was rated as
“Yes”, “No”, or “N/A” (Not Applicable). Components were then aggregated to create a determination of
“Met”, “Partially Met”, “Unmet”, or “Not Applicable” for each of the 10 steps.
Table 10 describes the criteria for reaching a determination in the scoring methodology.
Table 10. Rating Scale for PIP Validation
Determination
Criteria
Met
All required components were present.
Partially Met
One but not all components were present.
Unmet
None of the required components were present.
Not Applicable
None of the required components are applicable.
Findings
This section presents an overview of the findings from the validation activities completed for each PIP
submitted by the MCOs. Each MCO’s PIP was reviewed against all components contained within the 10
steps.
Adolescent Well Care PIPs
All Adolescent Well Care PIPs focused on increasing the number of adolescents ages 12-21 who receive at
least one comprehensive well-care visit with a PCP or an OB/GYN practitioner during the measurement
year, according to HEDIS
®
technical specifications.
Table 11 represents the PIP Validation Results for all Adolescent Well Care PIPs for CY 2013.

2014 Annual Technical Report
Delmarva Foundation
39
Table 11. Adolescent Well Care PIP Validation Results for CY 2013
Step/Description
Adolescent Well Care PIP Review Determinations
ACC
JMS
MPC
MSFC
PPMCO
UHC
1. Assess the Study Methodology
Met
Met
Met
Met
Met
Met
2. Review the Study Question(s)
Met
Met
Met
Met
Met
Met
3. Review the Selected Study Indicator(s)
Met
Met
Met
Met
Met
Met
4. Review the Identified Study Population
Met
Met
Met
Met
Met
Met
5. Review Sampling Methods
Met
Met
N/A
Met
Met
Met
6. Review Data Collection Procedures
Met
Met
Met
Met
Met
Met
7. Assess Improvement Strategies
Partially
Met
Met
Met
Met
Met
Met
8. Review Data Analysis & Interpretation of Study
Results
Partially
Met
Met
Met
Met
Met
Met
9. Assess Whether Improvement is Real
Improvement
Partially
Met
Partially
Met
Met
Partially
Met
Partially
Met
Met
10. Assess Sustained Improvement
N/A
N/A
N/A
N/A
N/A
N/A
MPC received a rating of “Not Applicable” for Step 5 (Review Sampling Methods) because sampling was not
utilized. MPC’s entire eligible population was used for this study.
ACC received a rating of “Partially Met” for Step 7 (Assess Improvement Strategies) because for the second
year in a row, the submission form was not completed appropriately. Additionally, a rating of “Partially Met”
was received for Step 8 because follow-up activities were not provided in the Qualitative Analysis.
Four MCOs received a rating of “Partially Met” for Step 9 (Assess Whether Improvement is Real
Improvement) because their rates decreased for this measurement period.
A rating of “Not Applicable” was received for all MCOs for Step 10 as sustained improvement cannot be
assessed until the second remeasurement year of the PIP has been assessed.
The following are examples of interventions which were implemented by the HealthChoice MCOs for the
Adolescent Well Care PIPs:
Nurse Medical Record Reviews to confirm that well child visits did not occur for non compliant
members
Provider visits to top 20 high volume PCPs to share non compliance member reports

2014 Annual Technical Report
Delmarva Foundation
40
Home visits offered to SSI population
Home visits to adolescents that have not been seen in the past two years
Onsite appointment scheduling
Birthday card reminders sent to members
Wellness letter sent to members
Automated telephone call reminders to non compliant members
Member incentives
Provider pay for performance program/provider incentives
School based clinic collaboration
Back to school flyers
Hiring of outreach representative
Piloting use of Facebook to communicate need for Adolescent Well Care (AWC) visits
Offer pediatric health fairs, with entertainment, games, food, and gifts at pediatric offices
Provider focus groups
Controlling High Blood Pressure PIPs
All Controlling High Blood Pressure PIPs focused on increasing the percentage of members 18-85 years of
age who had a diagnosis of hypertension and who blood pressure was adequately controlled during the
measurement year.
Table 12 represents the PIP Validation Results for all Controlling High Blood Pressure PIPs for CY 2013.
Table 12. Controlling High Blood Pressure PIP Validation Results for CY 2013
Step/Description
Controlling High Blood Pressure
PIP Review Determinations
ACC
JMS
MPC
MSFC
PPMCO
UHC
1. Assess the Study Methodology
Met
Met
Met
Met
Met
Met
2. Review the Study Question(s)
Met
Met
Met
Met
Met
Met
3. Review the Selected Study Indicator(s)
Met
Met
Met
Met
Met
Met
4. Review the Identified Study Population
Met
Met
Met
Met
Met
Met
5. Review Sampling Methods
Met
Met
Met
Met
Met
Met
6. Review Data Collection Procedures
Met
Met
Met
Met
Met
Met
7. Assess Improvement Strategies
Met
Met
Met
Met
Met
Met
8. Review Data Analysis & Interpretation of
Study Results
Met
Met
Met
Met
Met
Met
9. Assess Whether Improvement Is Real
Improvement
N/A
N/A
N/A
N/A
N/A
N/A
10. Assess Sustained Improvement
N/A
N/A
N/A
N/A
N/A
N/A

2014 Annual Technical Report
Delmarva Foundation
41
All Controlling High Blood Pressure PIPs received a rating of “Not Applicable” for Steps 9 and 10 (Assess
Whether Impovement Is Real Improvement and Assess Sustained Improvement) because this was the
baseline year of data collection, and neither real improvement nor sustained improvement could be assessed.
The following are examples of interventions which are planned for the CY 2014 by the HealthChoice MCOs
for the Controlling High Blood Pressure PIPs:
Hypertention Disease Management Program
Physician education dinner series
Provider work groups
Pharmacy reminder programs for providers
Social media to education members
Follow up on ER encounters to ensure appointments with PCP
Education materials to members and providers
Transportation for members
Medical record reviews
Annual health fairs
Increase staff for outreach to members
Member incentives
Adolescent Well Care Indicator Results
This is the first remeasurement year for the Adolescent Well Care PIP. Table 13 represents the indicator rates
for all MCOs for the PIP.
Table 13. Adolescent Well Care PIP Indicator Rates
Measurement Year
Indicator 1: Adolescent Well Care
ACC
JMS
MPC
MSFC
PPMCO
UHC
Baseline Year
1/1/12-12/31/12
68.06%
76.85%
60.20%
69.40%
67.59%
59.71%
Measurement Year 1
1/1/13-12/31/13
67.93%
76.72%
68.75%
67.80%
61.57%
60.80%
Remeasurement Year 2
1/1/14-12/31/14
N/A
N/A
N/A
N/A
N/A
N/A
Remeasurement Year 3
1/1/15-12/31/15
N/A
N/A
N/A
N/A
N/A
N/A
The rate for the 2012 HEDIS
®
Medicaid 90th Percentile measure for Adolescent Well Care was 64.72%.
MPC and UHC are performing below the 90th percentile, and the remaining four MCOs are performing
above the 90th percentile.
Two MCO’s indicator rates increased over baseline measurement. Those increases included a 1.09 percentage
point increase for UHC and an 8.55 percentage point increase for MPC. Four MCO’s indicator rates

2014 Annual Technical Report
Delmarva Foundation
42
decreased (ACC, JMS, MSFC, and PPMCO) over baseline measurement. Those decreases ranged from .13
percentage points to 6.02 percentage points.
Controlling High Blood Pressure Indicator Results
This is the baseline year of data collection for the Controlling High Blood Pressure PIP. Table 14 represents
the Controlling High Blood Pressure PIP indicator rates for all MCOs for the PIP.
Table 14. Controlling High Blood Pressure PIP Indicator Rates
Measurement Year
Indicator 1: Controlling High Blood Pressure
ACC
JMS
MPC
MSFC
PPMCO
UHC
Baseline Year
1/1/13 - 12/31/13
49.00%
56.20%
46.78%
65.52%
56.97%
42.34%
Measurement Year 1
1/1/14 - 12/31/14
N/A
N/A
N/A
N/A
N/A
N/A
Remeasurement Year 2
1/1/15 - 12/31/15
N/A
N/A
N/A
N/A
N/A
N/A
Remeasurement Year 3
1/1/16 - 12/31/16
N/A
N/A
N/A
N/A
N/A
N/A
The rate for the 2014 HEDIS
®
Medicaid 90th Percentile for Controlling High Blood Pressure measure is
69.79%. All MCOs are performing below the benchmark set for this PIP which is the 90th percentile. MSFC
is close to approaching this benchmark, however, two MCO’s rates (JMS and PPMCO) are performing at or
around the 50
th
Percentile which is 56.20%. The remaining MCO’s rates (ACC, MPC, and UHC) are close to
or below the 25
th
Percentile which is 48.53% for this measure.
Recommendations
Delmarva Foundation recommends that the MCOs continue to concentrate on the following:
Completing thorough and annual barrier analysis, which will direct where limited resources can be most
effectively used to drive improvement.
Developing system-level interventions, which include educational efforts, changes in policy, targeting of
additional resources, or other organization-wide initiatives. Face-to-face contact is usually most effective.
To improve outcomes, interventions should be systematic (affecting a wide range of members, providers
and the MCO), timely, and effective.
Assessing interventions for their effectiveness, and making adjustments where outcomes are
unsatisfactory.
Detailing the list of interventions (who, what, where, when, how many) to make the intervention
understandable and so that there is enough information to determine if the intervention was effective.

2014 Annual Technical Report
Delmarva Foundation
43
Section IV
Encounter Data Validation
Introduction
The Medicaid Managed Care Provisions of the BBA directed the U.S. Department of Health and Human
Services to develop protocols to serve as guidelines for conducting EQRO activities. In 1995, CMS began
developing a series of tools to help State Medicaid agencies collect, validate, and use encounter data for
managed care program management and oversight. Among the functions that Delmarva Foundation
performs as EQRO for the Maryland HealthChoice Program is the medical record review component for
encounter data validation (EDV). Delmarva Foundation completes encounter data validation according to
CMS’ EQR Protocol 4: Validation of Encounter Data Reported by the MCO. The Department required all
HealthChoice MCOs to submit CY 2013 encounter data by June 2014.
Encounter Data Validation Process
The CMS approach to EDV
1
includes the following three core activities:
Assessment of MCO information system (IS).
Analysis of MCO electronic encounter data for accuracy and completeness.
Review of medical records for additional confirmation of findings.
The EDV protocol also makes the following assumptions:
An encounter refers to the electronic record of a service provided to an MCO enrollee by both
institutional and non-institutional providers.
The State specifies the types of encounters (e.g., physician, hospital, dental, vision, laboratory) for which
encounter data are to be provided. In addition, the type of data selected for review (e.g., inpatient,
outpatient, office visits) is directly proportionate to the total percent of encounter types per calendar year.
Encounter data is considered “complete” when the data can be used to describe the majority of services
that have been provided to Medicaid beneficiaries who are MCO participants.
Encounter data completeness and accuracy requires continued monitoring and improvement. States need
to develop encounter data standards and monitor for accuracy and completeness. Ultimately, it is the
State that establishes standards for encounter data accuracy and completeness.
1
Department of Health and Human Services, Centers for Medicare and Medicaid Services. Validation of Encounter
Data Reported by the MCO, A Voluntary Protocol for External Quality Review (EQR), September 2012

2014 Annual Technical Report
Delmarva Foundation
44
The EDV protocol consists of five sequential activities:
Review of State requirements for collection and submission of encounter data
Review of MCO’s capability to produce accurate and complete encounter data
Analysis of MCO’s electronic encounter data for accuracy and completeness
Review of medical records for additional confirmation of findings
Analysis and submission of findings
Medical Record Review Procedure
Medical Record Validation
Medical record documentation for services provided from January 2013 through December 2013 was
compared to the encounter data for the same time period. The medical record was validated as the correct
medical record requested by verifying the patient name, date of birth (DOB), and gender.
Encounter Data Validation
After completing medical record reviewer training and achieving an inter-rater reliability score of 95%,
reviewers entered data from the medical record reviews into the Delmarva Foundation EDV Tool/Database.
The medical record was reviewed by either a certified coder or a nurse with coding experience to determine if
the submitted encounter data (diagnosis, procedure, or revenue codes) could be validated against the findings
in the medical record (see Table 15 for definition of terms). Where the diagnosis, procedure, and revenue
codes could be substantiated by the medical record, the review decision was “yes” or “a match.” Conversely,
if the medical record could not support the encounter data, the review decision was “no” or “no match.” For
inpatient encounters, the medical record reviewers also matched the principal diagnosis code to the primary
sequenced diagnosis. The review included validation of a maximum of 9 diagnosis codes, 6 procedure codes,
and 23 revenue codes per record.
Table 15. EDV Definition of Terms
Term
Definition
Encounter
A unique date of service with coded diagnoses and procedures for a single provider
or care/service provided on a unique date of service by the provider.
Review element
Specific element in the encounter data which is being compared to the medical
record; elements in this review include diagnosis, procedure, and revenue codes.
Match rate
Rate of correct record elements to the total elements presented as a percent.
The following reviewer guidelines were used to determine agreement or “match” between the encounter data
and the medical record findings:
As directed by the CMS Protocol, medical record reviewers could not infer a diagnosis from the medical
record documentation. Reviewers were required to use the diagnosis listed by the provider. For example,

2014 Annual Technical Report
Delmarva Foundation
45
if the provider recorded “fever and chills” in the medical record, and the diagnosis in the encounter data
was “upper respiratory infection,” the record did not match for diagnosis even if the medical record
documentation would support the use of that diagnosis.
For inpatient encounters with multiple diagnoses listed, the medical record reviewers were instructed to
match the first listed diagnosis (as the principal diagnosis) with the primary diagnosis in the encounter
data.
Procedure data was matched to the medical record regardless of sequencing.
Analysis Methodology
Data from the EDV Tool/Database were used to analyze the consistency between submitted encounter data
and corresponding medical records. Results were analyzed and presented separately by encounter type and
review element. Match rates and reasons for “no match” errors for diagnosis code, procedure code, and
revenue code elements are presented for Inpatient, Outpatient, and Office Visit encounter types in the
results. Delmarva Foundation recommended that DHMH set the standard for accuracy of match rates
between encounter data and medical records at 90% based on rates obtained in previous years.
Exclusion Criteria
Cases where a match between the medical record and encounter data could not be verified by DOB, gender,
and name were excluded from analyses. If information for DOB, gender, or name were missing, the record
could not be validated and was excluded from analyses.

2014 Annual Technical Report
Delmarva Foundation
46
Medical Record Sampling
Delmarva Foundation received a random sample of HealthChoice encounter data for hospital inpatient,
hospital outpatient, and physician office services that occurred in CY 2013 from Hilltop. The sample size,
determined to achieve a 95% confidence interval, was 384 medical records (Table 16). Oversampling for CY
2013 continued in order to ensure adequate numbers of medical records were received to meet the required
sample size. The hospital inpatient encounter types were oversampled by 500%, while the hospital outpatient
and office visit encounter types were oversampled by 200%.
Table 16. Maryland EDV Sample Size by Encounter Type, CY 2011 – CY 2013
CY 2011
CY 2012
CY 2013
Encounter
Type
Total
Encounters
% of
Encounters
Sample
Size
Total
Encounters
% of
Encounters
Sample
Size
Total
Encounters
% of
Encounters
Sample
Size
Inpatient
107,202
1.00%
4
116,434
1.60%
6
114,236
1.50%
6
Outpatient
1,030,121
9.50%
36
1,117,949
15.30%
59
1,143,752
15.05%
58
Office Visit
9,702,064
89.50%
344
6,090,237
83.10%
319
6,340,051
83.44%
320
Total
10,839,387
100.00%
384
7,324,620
100.00%
384
7,598,039
100.0%
384
The shift in the proportion of encounter types of the random sample as seen in Table 16:
Office Visits decreased 6.4 percentage points from 89.5% in CY 2011 to 83.1% in CY 2012 and then
increased by 0.34 percentage points to 83.44% in CY 2013.
Outpatient increased by 5.8 percentage points from 9.5% in CY 2011 to 15.3% in CY 2012 and then
declined by 0.25 percentage points to 15.05 in CY 2013.
Inpatient increased by 0.6 percentage points from 1.0% in CY 2011 to 1.6% in CY 2012 and then
declined by 0.1 percentage points to 1.5% in CY 2013.
From the information provided in Table 16, the following conclusions can be drawn:
Office Visit encounters make up the majority of the random sample of encounter data in all three years.
Inpatient encounters comprise a very small part of the random sample at less than two percent in all
three years.
The percentage of Office Visit encounters in the sample declined from CY 2011 to CY 2012 and then
increased slightly in CY 2013.
The increase in percentage of Office Visit encounters in the sample in CY 2013 was offset by a decline in
the percentage of the sample of inpatient and outpatient encounters.
With the approval of DHMH, Delmarva Foundation mailed requests for medical records to the providers of
service. Non-responders were contacted by telephone and fax. The efforts to obtain adequate records to meet
the minimum sample in CY 2013 were impacted by:
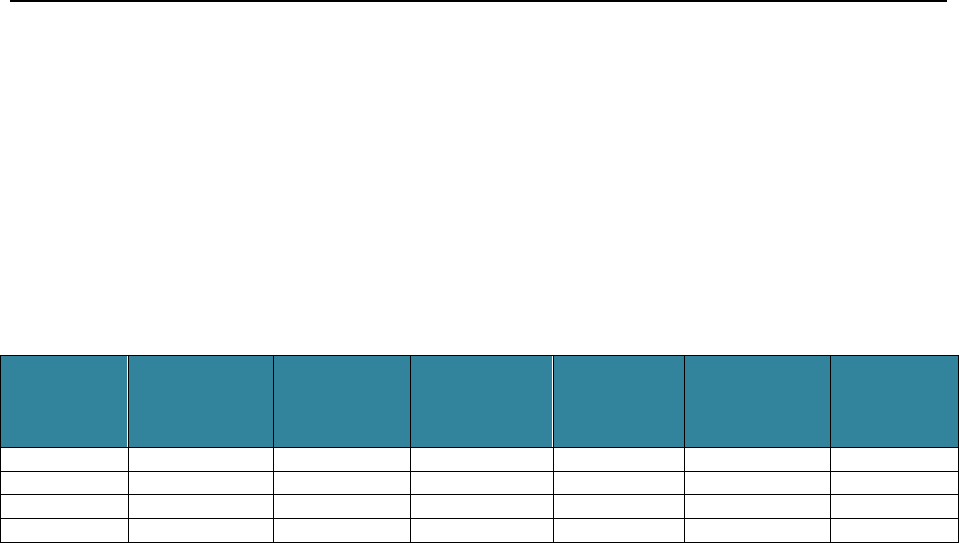
2014 Annual Technical Report
Delmarva Foundation
47
Many outpatient records were submitted without the patient’s DOB included (7%). Since DOB was one
of the critical elements needed to determine a record to be valid, these records either were not included in
the review or required additional follow-up to obtain the missing information.
There continued to be an issue with outpatient and office visit requests being returned due to bad
addresses (8%).
Response rates by encounter type are outlined in Table 17.
Table 17. Maryland EDV Medical Record Response Rates by Encounter Type, CY 2011 - CY 2013
Encounter
Type
CY 2011
Total Records
Received and
Reviewed
CY 2011
Sample Size
Achieved?
Yes/No
CY 2012
Total Records
Received and
Reviewed
CY 2012
Sample Size
Achieved?
Yes/No
CY 2013
Total Records
Received and
Reviewed
CY 2013
Sample Size
Achieved?
Yes/No
Inpatient
4
Yes
7
Yes
7
Yes
Outpatient
38
Yes
60
Yes
61
Yes
Office Visit
352
Yes
326
Yes
324
Yes
Total
394
393
392
Review sample sizes were achieved for each encounter type for all three calendar years.

2014 Annual Technical Report
Delmarva Foundation
48
Results
The analysis of the data was organized by review elements including diagnosis, procedure, and revenue codes.
A total of 392 medical records were reviewed. The overall element match rate (medical record review
supporting the encounter data submitted) increased by 1.2 percentage points for CY 2013 as compared to CY
2012, but remained lower than the CY 2011 match rate. The results for CY 2011 - CY 2013 EDV are
displayed in the following tables and the findings are discussed.
Table 18. Maryland EDV Results by Encounter Type, CY 2011 – CY 2013
Encounter
Type
Records Received &
Reviewed
Total Elements
Possible*
Total Matched Elements
Percentage of Matched
Elements
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
Inpatient
4
7
7
67
152
65
66
147
64
98.5%
96.7%
98.5%
Outpatient
38
60
61
435
614
666
394
588
630
90.6%
95.8%
94.6%
Office Visit
352
326
324
1,075
1,084
1,014
1,063
1,018
982
98.9%
93.9%
96.8%
TOTAL
394
393
392
1,577
1,850
1,745
1,523
1,753
1,676
96.6%
94.8%
96.0%
*Possible elements include diagnosis, procedure, and revenue codes.
The overall match rate (medical record review supporting the encounter data submitted) in CY 2013 was
96.0% which represents an increase of 1.2 percentage points from CY 2012, and almost reaches the CY 2011
match rate of 96.6%.
From CY 2012 to CY 2013 the inpatient encounter data match rate increased by 1.8 percentage points to
98.5%, equivalent to the match rate achieved in CY 2011.
The outpatient encounter data match rate was 94.6% for CY 2013, representing a decrease of 1.2 percentage
points compared to CY 2012 for this encounter type (95.8%), and remaining above the 2011 rate of 90.6%.
Finally, office visit encounters registered a rate of 96.8%, an increase of almost 3 percentage points compared
to CY 2012 (93.9%), and approaching the match rate of 98.9% for CY 2011.

2014 Annual Technical Report
Delmarva Foundation
49
Results by Review Element
Tables 19 through 21 illustrate EDV results by review element for each encounter type. The elements
reviewed were diagnosis codes, procedure codes, and revenue codes. (Note: Revenue codes are not applicable
for office visit encounters.)
Inpatient Encounters
Table 19. Maryland EDV Results by Element by Inpatient Encounter Type, CY 2011 – CY 2013
Encounter
Type
Diagnosis Codes
Procedure Codes
Revenue Codes
Total
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
Match
17
43
39
5
15
4
44
89
21
66
147
64
No Match
1
4
0
0
0
0
0
1
1
1
5
1
Total
Elements
18
47
39
5
15
4
44
90
22
67
152
65
Match
Percent
94.4%
91.5%
100%
100%
100%
100%
100%
98.9%
95.5%
98.5%
96.7%
98.5%
In CY 2013, diagnosis codes were matched at a 100% rate when compared to the content of the inpatient
medical record, an increase of 8.5 percentage points compared to 2012. Inpatient procedure codes maintained
a 100% match rate when compared to inpatient medical records in each measurement year.
Again in CY 2013, one revenue code failed to match in the review resulting in a match rate of 95.5%,
declining 3.4 percentage points from the CY 2012 match rate.
Overall, the Total match rate for Inpatient encounters across all elements in CY 2013 rose by 1.8 percentage
points from CY 2012 to register a rate of 98.5%, the same match rate reported in CY 2011.
Outpatient Encounters
Table 20. Maryland EDV Results by Element by Outpatient Encounter Type, CY 2011 – CY 2013
Encounter
Type
Diagnosis Codes
Procedure Codes
Revenue Codes
Total
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
Match
91
162
182
101
171
154
202
255
294
394
588
630
No Match
5
17
9
35
7
26
1
2
1
41
26
36
Total
Elements
96
179
191
136
178
180
203
257
295
435
614
666
Match
Percent
94.8%
90.5%
95.3%
74.3%
96.1%
85.6%
99.5%
99.2%
99.7%
90.6%
95.8%
94.6%

2014 Annual Technical Report
Delmarva Foundation
50
In CY 2013, the diagnosis code element match rate increased by 4.8 percentage points to 95.3% compared to
90.5% in CY 2012.
Consistent with 2011 Outpatient encounter comparisons, the procedure code element had the lowest match
rate of all elements in CY 2013 at 85.6%. This represents a decrease of 10.5 percentage points from the CY
2012 match rate for the procedure code element of 96.1%. In CY 2012, the procedure code match rate rose
21.8 percentage points to 96.1%.
In CY 2013 revenue codes showed an increase in match rate from 99.2% in CY 2012 to 99.7%, an increase of
0.5 percentage points.
Overall, the Total match rate for Outpatient encounters across all of the element types decreased slightly by
1.2 percentage points, from 95.8% in CY 2012 to 94.6% in CY 2013.
Office Visit Encounters
Table 21. Maryland EDV Results by Element by Office Visit Encounter Type, CY 2011 – CY 2013
Encounter
Type
Diagnosis Codes
Procedure Codes
Revenue Codes
Total
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
CY
2011
CY
2012
CY
2013
Match
714
707
673
349
311
309
NA
NA
NA
1,063
1,018
982
No Match
9
29
17
3
37
15
NA
NA
NA
12
66
32
Total
Elements
723
736
690
352
348
324
NA
NA
NA
1,075
1,084
1014
Match
Percent
98.8%
96.1%
97.5%
99.1%
89.4%
95.4%
NA
NA
NA
98.9%
93.9%
96.8%
The Total match rate increased 2.9 percentage points to 96.8% in CY 2013, from 93.9% in CY 2012.
Diagnosis code and procedure code match rates both rose from CY 2012 to CY 2013, increasing by 6
percentage points for procedure codes and 1.4 percentage points for diagnosis codes.

2014 Annual Technical Report
Delmarva Foundation
51
“No Match” Results by Element and Reason
Diagnosis Code Element Review
Tables 22 through 24 illustrate the principal reasons for “no match” errors. The reasons for determining a
“no match” for the diagnosis code element were:
Lack of medical record documentation
Incorrect principal diagnosis (inpatient encounters) or incorrect diagnosis codes
Table 22. Maryland EDV CY 2013 “No Match” Results for Diagnosis Code Element
CY 2013
“No Match” for Diagnosis Code Element
Encounter Type
Total Elements
Lack of Medical Record
Documentation
Incorrect Principal Diagnosis (Inpatient)
or Incorrect Diagnosis Codes
Inpatient
0
0
0
% of Total
0%
0%
Outpatient
9
2
7
% of Total
22.2%
77.8%
Office Visit
17
3
14
% of Total
17.6%
82.4%
There were no inpatient procedure codes that did not match in CY 2013. In prior calendar year comparisons,
all inpatient diagnosis code “no match” errors were due to incorrect diagnosis codes. Of the 9 “no match”
errors for outpatient encounters in CY 2013, 22.2% resulted from a lack of medical record documentation,
whereas 77.8% “no match” errors resulted from incorrect diagnosis codes. Similarly, the majority of the 17
“no matches” for CY 2012 outpatient encounters (88.2%) were due to incorrect diagnosis codes.
For office visit encounters, 27.6% of the 17 “no match” errors in CY 2013 resulted from a lack of medical
record documentation, compared to 82.4% of the “no match” errors in CY 2012. In CY 2013, 82.4% “no
match” errors for office visit encounters were the result of incorrect diagnosis codes.

2014 Annual Technical Report
Delmarva Foundation
52
Procedure Code Element Review
The reasons for determining a “no match” for the procedure code element were:
Lack of medical record documentation
Incorrect procedure codes
Table 23. Maryland EDV CY 2013 “No Match” Results for Procedure Code Element
In all three contract years, there were no inpatient procedure codes that did not match.
In CY 2013, 96.2% of “no match” errors for Outpatient encounters were due to incorrect procedure codes.
In CY 2012, 100% of the procedure code “no match” errors for office visits were due to incorrect procedure
codes.
All of the procedure code “no match” errors for Office Visit encounters in CY 2013 were the result of
incorrect procedure codes. By contrast, of the 37 “no match” errors detected in CY 2012, 56.8% were due to
lack of medical documentation and 43% were due to incorrect procedure codes.
Revenue Code Element Review
The reasons for determining a “no match” for the revenue code element were:
Lack of medical record documentation
Incorrect revenue codes
Table 24. Maryland EDV CY 2013 “No Match” Results for Revenue Code Element
CY 2013
“No Match” for Revenue Code Element *
Encounter Type*
Total Elements
Lack of Medical Record
Documentation
Incorrect Revenue Code
Inpatient
1
0
1
% of Total
0%
100%
Outpatient
1
1
1
% of Total
100%
0%
*Note – Revenue Codes do not apply to Office Visit encounters.
CY 2013
“No Match” for Procedure Code Element
Encounter
Type
Total Elements
Lack of Medical Record
Documentation
Incorrect Procedure Code
Inpatient
0
0
0
% of Total
0%
0%
Outpatient
26
1
25
% of Total
3.8%
96.2%
Office Visit
15
0
15
% of Total
0%
100%

2014 Annual Technical Report
Delmarva Foundation
53
Incorrect revenue codes and lack of medical record documentation respectively were the reasons for the one
revenue code “no match” error for Inpatient and Outpatient encounters in CY 2013. In CY 2012, one of the
Outpatient “no match errors” was due to an incorrect revenue code, while the other was due to lack of
medical documentation. Similar to CY 2013, the Inpatient “no match” error was due to an incorrect revenue
code.
Conclusions and Recommendations
For CY 2013, overall encounters matched the medical records 96% of the time. This match rate exceeds
Delmarva Foundation’s recommended standard of 90% for accuracy of match rates between encounter data
and medical records. The overall match rate for CY 2013 registered an increase of 1.2 percentage points
above the match rate for 2012, which had declined 1.8 percentage points from CY 2011. The rate for 2013 is
approaching but remains slightly below the 2011 rate of 96.6%. Therefore, the encounter data submitted for
CY 2013 can be considered reliable for reporting purposes.
The match rates for Inpatient encounters were 98.5% and Office Visits were 96.8%. Outpatient encounters
had the lowest match rate of all encounter types at 94.6%. Amongst all Outpatient visit encounters, the
procedure code element had the lowest match rate of all elements at 85.6%, as compared to the highest
match rate of all elements at 99.7% for revenue codes.
Based on our encounter data validation, we concluded that the primary reason for “no match” results in the
Inpatient, Outpatient, and Office Visit encounters for the diagnosis code element was due to incorrect
diagnosis codes. Only two of the nine “no match” errors for Outpatient encounters for the diagnosis code
element were due to a lack of medical record documentation. Only three of the 17 “no match” errors for
Office Visit encounters for the diagnosis code element were due to a lack of medical record documentation.
By contrast, all of the records matched for Inpatient diagnosis code.
The primary reason for all the “no match” results in the Outpatient encounter data for the procedure code
element was due to incorrect procedure codes (25 out of 26 records). All 15 of the Office Visit encounter “no
match” errors were due to incorrect procedure codes. All Inpatient encounter data procedure code elements
were matched.
It is recommended that the current rate of oversampling be continued in order to ensure adequate numbers
of medical records are received to meet the required sample size. Communication with provider offices
reinforcing the requirement to supply all supporting medical record documentation for the encounter data,
including the patient’s date of birth, has mitigated the impact of lack of documentation on meeting the
minimum sample. Outpatient and Office Visit requests being returned due to bad addresses continues to be
an issue in obtaining adequate records to meet the minimum sample.

2014 Annual Technical Report
Delmarva Foundation
54
Section V
Early and Periodic Screening, Diagnosis,
and Treatment (EPSDT) Medical Record Review
Introduction
As the EQRO, Delmarva Foundation annually completes an EPSDT medical record review. The medical
records review findings assist the Department in evaluating the degree to which HealthChoice children and
adolescents through 20 years of age are receiving timely screening and preventive care.
This section summarizes the findings from the EPSDT medical record review for CY 2013. Approximately
578,039 children were enrolled in the HealthChoice Program during this period.
The seven MCOs evaluated for CY 2013 were:
AMERIGROUP Community Care (ACC)
Priority Partners (PPMCO)
Jai Medical Systems (JMS)
Riverside Health of Maryland (RHMD)
Maryland Physicians Care (MPC)
MedStar Family Choice, Inc. (MSFC)
UnitedHealthcare (UHC)
Program Overview
The EPSDT Program is the federally mandated Medicaid program for screening, prevention, diagnosis, and
treatment of physical and mental health conditions in children and adolescents through 20 years of age (as
defined by Omnibus Budget Reconciliation Act [OBRA] 1989). Each State determines its own periodicity
schedule for services, including periodic physical and mental health screening, vision, dental, and hearing
services.
The Program’s philosophy is to provide quality health care that is patient focused, prevention oriented,
coordinated, accessible, and cost effective. The foundation of this philosophy is based on providing a
“medical home” for each enrollee, by connecting each enrollee with a PCP who is responsible for providing
preventive and primary care services, managing referrals, and coordinating all necessary preventive care for
the enrollee. The Program emphasizes health promotion and disease prevention, and requires that
participants be provided health education and outreach services.

2014 Annual Technical Report
Delmarva Foundation
55
Program Objectives
The Maryland EPSDT Program’s mission is to promote access to and assure availability of quality health care
for Medical Assistance children, and adolescents through 20 years of age. In support of the program’s
mission, the primary objective of the EPSDT medical record review is to collect and analyze data to assess
the timely delivery of EPSDT services to children and adolescents enrolled in an MCO. The review includes
an assessment of MCO performance for the following EPSDT components and their respective
subcategories:
Health and developmental history requires a comprehensive evaluation and includes documentation of:
Annual medical, immunization, family, and psychosocial histories with yearly updates.
Peri-natal history up through 2 years of age.
Developmental history/surveillance through 20 years of age.
Mental health assessment beginning at 3 years of age.
Substance abuse screening beginning at 12 years of age, younger if indicated.
Development screening using a standardized screening tool at the 9, 18, and 24-30 month visits.
Comprehensive, unclothed, physical exam requires evaluation and includes documentation of:
A complete assessment of no fewer than five body systems.
Age-appropriate vision and hearing assessments at every visit.
Nutritional assessment at every age.
Oral assessment at all ages.
Height and weight measurement with graphing through 20 years of age.
Head circumference measurement and graphing through 2 years of age.
BMI calculation and graphing for ages 2 through 20.
Blood pressure measurement beginning at 3 years of age.
Laboratory tests/at-risk screenings require documentation of:
Hereditary/metabolic screening test results at birth and again by 1 month* of age.
Age-appropriate risk assessment results for tuberculosis, cholesterol, and sexually transmitted diseases.
Counseling and/or laboratory test results for at-risk recipients.
Anemia tests at 12** and 24*** months of age.
Lead risk assessment beginning at 6 months through 6 years of age.
Referral to the lab for lead testing at appropriate ages.
Blood lead tests results at 12** and 24*** months of age.
Baseline blood lead test results for ages 3 through 5 when not done at 12 or 24 months of age.
Children with a blood level greater than 5 ug/dL must have a blood level drawn within 3 months of the
initial test.
NOTES: *accepted until 8 weeks of age, **accepted from 9-23 months of age, ***accepted from 24-35 months of age

2014 Annual Technical Report
Delmarva Foundation
56
Immunizations require assessment of need and documented administration that:
The DHMH Immunization Schedule is being implemented in accordance with the Advisory Committee
on Immunization Practices (ACIP) guidelines.
Age-appropriate vaccines are not postponed for inappropriate reasons.
Children and/or adolescents who are delayed in their immunizations are brought current with the
DHMH Immunization Schedule.
Health education and anticipatory guidance requires documentation of:
Age-appropriate guidance, with a minimum of three anticipatory guidance items or two major topics
documented per visit.
Counseling and/or referrals for health issues identified by the parent(s) or provider during the visit.
Oral health assessment following eruption of teeth, yearly dental education, and referrals are required
beginning at 12 months of age.
Educating recipient and/or parent regarding schedule of preventive care visits.
Return appointment documents, according to Maryland Schedule of Preventive Health Care.
CY 2013 EPSDT Review Process
Sampling Methodology
The sample frame was drawn from preventive care encounters occurring during calendar year 2013 for
children from birth through 20 years of age. The sampling methodology includes the following criteria:
A random sample of preventive care encounters per MCO including a 10% over sample.
Sample size per MCO provides a 95% confidence level and 5% margin of error.
Sample includes only recipients through 20 years of age as of the last day of CY 2013.
Sample includes encounter data for recipients enrolled on last day of CY 2013, and for at least 320 days
in the same MCO.
Exception
– If the recipient’s age on the last day of selected period is less than 365 days, the criteria is
modified to read same MCO for 180 days, with no break in eligibility.
Sample includes recipients who had a preventive care encounter (Current Procedural Terminology [CPT]
99381-85 or 99391-95) with a diagnostic code of V20 or V70 (For children less than 2 years of age who
may have had 4-6 preventive visits within a 12-month period, only one date of service was selected.)
Sample includes recipients when visits with CPT 99381-85 or 99391-95 were provided by primary care
providers and clinics with the following specialties:
Pediatrics
Family Practice
Internal Medicine
Nurse Practitioner
General Practice
Federally Qualified Health Center

2014 Annual Technical Report
Delmarva Foundation
57
Medical Record Review Process
Medical records were randomly selected in order to assess compliance with the program standards. Nurse
reviewers conducted all medical record reviews in the provider offices, with the exception of providers with
only one or two children in the sample. These providers were given the option to mail or fax a complete copy
of the medical record to Delmarva Foundation for review. In total, 2,366 medical records were reviewed for
CY 2013.
The review criteria used by Delmarva Foundation’s review nurses were the same as those developed and used
by the Department’s EPSDT review nurses. Delmarva Foundation review nurses completed annual training
and conducted Inter-Rater Reliability. The review nurses achieved a score of 90% prior to the beginning of
the CY 2013 EPSDT Medical Record Review.
Scoring Methodology
Data from the medical record reviews were entered into Delmarva Foundation’s EPSDT Evaluation Tool.
The analysis of the data was organized by the following age groupings:
birth through 11 months,
12 through 35 months,
3 through 5 years,
6 through 11 years, and
12 through 20 years of age.
The following scores were provided to the specific elements within each age group based on medical record
documentation:
Score
Finding
2
Complete
1
Incomplete
0
Missing
Exception
– When an element is not applicable to a child, such as a vision assessment for a blind child or a
documented refusal for a flu vaccine by a parent, a score of two was given.
Elements, each weighted equally, within a component were scored and added together to derive the final
component score. Similarly, the composite score (or overall score) follows the same methodology.

2014 Annual Technical Report
Delmarva Foundation
58
The scoring methodology produced a result that reflected the percentage of possible points obtained in each
component, for each age group, and for each MCO. The minimum per component compliance score is 75%.
If the minimum compliance score is not met, a CAP is required.
Findings
EPSDT review indicators are based on current pediatric preventive care guidelines and DHMH-identified
priority areas. The guidelines and criteria are divided into five component areas. Each MCO was required to
meet a minimum compliance rate of 75% for each of the five components. If an MCO did not achieve the
minimum compliance rate, the MCO was required to submit a CAP. Five of the seven MCOs (ACC, JMS,
MPC, MSFC, and PPMCO) met the minimum compliance rate of 75% in each of the five component areas
for the CY 2013 review. RHMD participated in the CY 2013 EPSDT review as a baseline review. The MCO’s
sample was limited (48 records) as the MCO had not participate in the HealthChoice system for the full
calendar year. Therefore, RHMD was not required to submit CAPs in the baseline year of review. CAPs for
the Laboratory Tests/At-Risk Screenings component were required from one MCO (UHC).
Findings for the CY 2013 EPSDT review by component area are described in Table 25.
Table 25. CY 2013 EPSDT Component Results by MCO
Component
Number of
Elements
Reviewed
ACC
JMS
MPC
MSFC
PPMCO
RHMD
UHC
HealthChoice
Aggregate
CY 2013
Health &
Developmental History
9
86%
97%
87%
91%
87%
92%
84%
89%
Comprehensive Physical
Examination
14
90%
95%
89%
92%
91%
95%
89%
91%
Laboratory Tests/At-
Risk Screenings
10
76%
94%
76%
78%
75%
58%*
66%*
77%
Immunizations
13
84%
84%
81%
87%
85%
95%
79%
84%
Health Education/
Anticipatory Guidance
4
89%
94%
88%
87%
90%
96%
86%
89%
*Denotes that the minimum compliance score of 75% was unmet and a CAP was required
The following section provides a description of each component along with a summary of HealthChoice
MCOs’ performance.

2014 Annual Technical Report
Delmarva Foundation
59
Health and Developmental History
Rationale: A comprehensive medical and family history assists the provider in determining health risks and
providing appropriate laboratory testing and anticipatory guidance.
Components: Medical history includes family, peri-natal, developmental, psychosocial, and mental health
information, as well as the immunization record. Psychosocial history assesses support systems and exposure
to family and/or community violence, which may adversely affect the child’s mental health. Developmental,
mental health, and substance abuse screenings determine the need for referral and/or follow-up services. The
mental health assessment provides an overall view of the child’s personality, behaviors, social interactions,
affect, and temperament.
Documentation: Annual updates for personal, family, and psychosocial histories are required to ensure the
most current information is available. The use of a standard age-appropriate history form (such as the
Maryland Healthy Kids Program Medical/Family History) or a similarly comprehensive history form (such as
the CRAFFT Assessment Tool from Children’s Hospital Boston) is recommended.
Table 26. CY 2013 Health and Developmental History Element Scores
Maryland Schedule of
Preventive Health Care
Health and Development
History Elements
ACC
CY 2013
JMS
CY 2013
MPC
CY 2013
MSFC
CY 2013
PPMCO
CY 2013
RHMD
CY 2013
UHC
CY 2013
Substance Abuse Assessment
80%
98%
78%
87%
83%
N/A
79%
Psychosocial History
89%
99%
89%
94%
92%
95%
86%
Mental Health Assessment
80%
98%
85%
90%
86%
N/A
84%
Family History
77%
97%
80%
85%
76%
89%
74%
Peri-natal History
83%
92%
96%
93%
87%
94%
83%
Health History
92%
99%
90%
95%
92%
93%
87%
Developmental Assessment/
History/Surveillance (0-5 yrs)
90%
91%
94%
97%
91%
92%
95%
Developmental Assessment/
History/Surveillance (6-20 yrs)
94%
88%
88%
95%
93%
N/A
92%
Developmental Screening
Using Standardized Tool at 9,
18, 24-30 Month Visits
71%
96%
86%
57%
75%
75%
72%
Recorded Autism Screening
using Standardized Tool*
30%
77%
23%
67%
42%
N/A
52%
Aggregate Element Rate
86%
97%
87%
91%
87%
92%
84%
__Denotes that the element score is below 75% which may impact the minimum level compliance score for the component.
*Baseline for CY 2012 and CY 2013 and was not used in the calculation of the overall component score.

2014 Annual Technical Report
Delmarva Foundation
60
Findings
All MCO aggregate scores exceeded the minimum compliance rate of 75% for the Health and
Developmental History component in CY 2013.
The CY 2013 HealthChoice Aggregate score for the Health and Developmental History component is
89% which is equal to the CY 2012 aggregate score.

2014 Annual Technical Report
Delmarva Foundation
61
Comprehensive Physical Examination
Rationale: The comprehensive physical exam uses a systems method review which requires documentation
of a minimum of five systems (example - heart, lungs, (HEENT or EENT), eyes, ears, nose, throat,
abdominal, genitals, skeletal-muscle, neurological, skin, head, face) to meet EPSDT standards.
Components & Documentation: A comprehensive physical exam includes documentation of:
Subjective or objective vision and hearing assessments at every well-child visit.
Measuring and graphing head circumference through 2 years of age.
Recording blood pressure annually for children 3 years of age and older.
Oral assessment, including a visual exam of the mouth, gums, and teeth.
Nutritional assessment, including typical diet, physical activity, and education provided with graphing of
weight and height through 20 years of age on the growth chart.
Calculating and graphing Body Mass Index (BMI) for 2 through 20 years of age.
Appropriate referrals for nutrition services and/or counseling due to identified nutrition or growth
problems.
Table 27. CY 2013 Comprehensive Physical Examination Element Scores
Maryland Schedule of
Preventive Health Care
Comprehensive Physical
Examination
ACC
CY 2013
JMS
CY 2013
MPC
CY 2013
MSFC
CY 2013
PPMCO
CY 2013
RHMD
CY 2013
UHC
CY 2013
Graphed Height
91%
100%
84%
94%
90%
92%
88%
Measured Height
99%
100%
99%
99%
99%
100%
100%
Graphed Weight
92%
100%
85%
95%
90%
92%
88%
Measured Weight
100%
100%
99%
100%
100%
100%
100%
Graphed Head
Circumference
78%
97%
68%
78%
74%
85%
72%
Measured Head
Circumference
84%
98%
87%
87%
83%
94%
84%
Measured Blood Pressure
97%
99%
98%
97%
99%
N/A
98%
Documentation Of
Minimum 5 Systems
88%
68%
88%
89%
92%
92%
89%
Assessed Hearing
87%
99%
90%
91%
89%
98%
83%
Assessed Vision
90%
99%
88%
90%
90%
98%
85%
Assessed Nutritional
Status
93%
97%
91%
93%
94%
96%
94%
Conducted Oral Screening
91%
86%
89%
88%
95%
95%
90%
Calculated BMI
83%
100%
89%
91%
87%
N/A
86%
Graphed BMI
64%
100%
74%
74%
77%
N/A
73%
Aggregate Element Rate
90%
95%
89%
92%
91%
95%
92%
__ Denotes that the element score is below 75% which may impact the minimum level compliance score for the component.

2014 Annual Technical Report
Delmarva Foundation
62
Findings
All MCO aggregate scores exceeded the minimum compliance rate of 75% for the Comprehensive
Physical Exam component for CY 2013.
The CY 2013 HealthChoice Aggregate score for the Comprehensive Physical Exam component is 91%,
which represents a two percentage point decrease from 93% in CY 2012.

2014 Annual Technical Report
Delmarva Foundation
63
Laboratory Tests/At-Risk Screenings
Rationale: The Healthy Kids Program requires assessments of risk factors associated with heart disease,
tuberculosis, lead exposure, and sexually transmitted infection/human immunodeficiency virus (STI/HIV).
Components: Assessment of risk factors includes:
Tuberculosis risk assessment beginning at 1 month of age.
Heart disease/cholesterol risk assessment beginning at 2 years of age.
STI/HIV risk assessment beginning at 12 years of age.
Lead risk assessment for 6 months through– 6 years of age. (A positive lead risk assessment necessitates
blood lead testing at any age. In addition, blood lead levels must be obtained at 12** and 24*** months
of age.)
Blood testing of hematocrit or hemoglobin at 12** and 24*** months of age, at the same time as the
blood lead test. (On the initial visit for all children 2 through 5 years of age, unless previous test results
are available, a hematocrit or hemoglobin test is required.)
A second hereditary/metabolic screen (lab test) by 2 to 4 weeks* of age.
Notes: *accepted until 8 weeks of age; **accepted from 9-23 months of age; ***accepted from 24-35 months of age
Table 28. CY 2013 Laboratory Test/At-Risk Screenings Element Scores
Maryland Schedule of
Preventive Health Care
Laboratory Test/At-Risk
Screenings
ACC
CY 2013
JMS
CY 2013
MPC
CY 2013
MSFC
CY 2013
PPMCO
CY 2013
RHMD
CY 2013
UHC
CY 2013
Cholesterol Risk
Assessment per Schedule
70%
98%
72%
73%
71%
N/A
62%
STI/HIV Risk Assessment
per Schedule
83%
98%
79%
84%
84%
N/A
79%
Referred for Lead Test
79%
71%
78%
65%
75%
N/A
51%
12 Month Lead Test Result
per Schedule
62%
95%
67%
80%
70%
N/A
61%
24 Month Lead Test Result
per Schedule
56%
94%
57%
77%
45%
N/A
48%
Lead Risk Assessment
85%
98%
88%
94%
86%
81%
82%
Anemia Screening per
Schedule
79%
92%
78%
87%
80%
N/A
61%
Conducted Second
Hereditary/Metabolic
Screening by 2-4 weeks
83%
88%
87%
73%
81%
49%
82%
Baseline Lead Testing
Completed
80%
75%
75%
89%
71%
N/A
52%
Tb Risk Assessment (1
mth-20yrs)
76%
98%
76%
75%
71%
60%
69%
Aggregate Element Rate
76%
94%
76%
78%
75%
58%*
66%*
__ Denotes that the element score is below 75% which may impact the minimum level compliance score for the component.

2014 Annual Technical Report
Delmarva Foundation
64
Findings
This component score historically represents an area in need of improvement. MCO specific
recommendations for quality improvement focused at the element level are shared annually with each
MCO in the EPSDT Medical Record Review Report.
RHMD and UHC scored below the minimum compliance rate of 75%. CY 2013 was the baseline year of
review for RHMD. RHMD’s sample was limited (48 records) as the MCO had not participated in the
HealthChoice system for a full calendar year. Therefore, RHMD was not required to submit CAPs in the
baseline year of review.
UHC was required to submit a CAP.
The CY 2013 HealthChoice Aggregate score for the Laboratory Tests/At-Risk Screenings component is
77%, which represents a three percentage point decrease from 80% in CY 2012.

2014 Annual Technical Report
Delmarva Foundation
65
Immunizations
Rationale: Children on Medical Assistance must be immunized according to the current Maryland DHMH
Recommended Childhood Immunization Schedule. The immunization schedule is endorsed by The Maryland
State Medical Society and is based on the current recommendations of the U.S. Public Health Service’s
Advisory Committee of Immunization Practices (ACIP) and the American Academy of Pediatrics. Primary
care providers who see Medicaid recipients through 18 years of age must participate in the Department’s
Vaccines for Children (VFC) Program.
Documentation: The VFC Program requires completion of the VFC Patient Eligibility Screening Record for
each patient receiving free vaccines. Additionally, federal law requires documentation of date, dosage, site of
administration, manufacturer, lot number, publication date of Vaccine Information Statement (VIS), and
name/location of provider. Immunization components are listed in the table below.
Table 29. CY 2013 Immunizations Element Scores
Maryland Schedule of
Preventive Health Care
Immunizations
ACC
CY 2013
JMS
CY 2013
MPC
CY 2013
MSFC
CY 2013
PPMCO
CY 2013
RHMD
CY 2013
UHC
CY 2013
TD Vaccine(s) per Schedule
87%
96%
87%
87%
87%
N/A
82%
Hepatitis B Vaccine(s) per
Schedule
91%
96%
88%
94%
93%
96%
86%
MMR Vaccine(s) per Schedule
95%
98%
93%
99%
98%
N/A
93%
Polio Vaccine(s) per Schedule
95%
98%
92%
98%
93%
98%
88%
Hib Vaccine(s) per Schedule
76%
81%
77%
78%
79%
97%
73%
DTP/DTaP (DT) Vaccine(s) per
Schedule
92%
93%
91%
95%
95%
98%
88%
Hepatitis A Vaccine(s) per
Schedule (2 dose requirement)
82%
96%
78%
91%
57%
N/A
90%
Influenza Vaccine(s)
(Beginning at 6 months of age
per schedule)
59%
52%
55%
66%
84%
77%
54%
Meningococcal (MCV4)
Vaccine(s) per Schedule
89%
94%
81%
81%
88%
N/A
78%
Varicella Vaccine(s) per
Schedule (2 dose requirement)
90%
90%
87%
90%
92%
N/A
82%
Rotavirus Vaccine(s) per
Schedule
84%
81%
78%
95%
86%
89%
62%
Assessed if Immunizations are
Up to Date
76%
71%
77%
80%
78%
97%
77%
PCV-13 Vaccine(s) per
Schedule
92%
96%
88%
97%
94%
96%
87%
Human Papillomavirus
Vaccine(s)*
73%
77%
54%
64%
63%
N/A
61%
Aggregate Element Rate
84%
84%
81%
87%
85%
95%
79%
__ Denotes that the element score is below 75% which may impact the minimum level compliance score for the component.
* This immunization data was collected for informational purposes only and was not used in the calculation of the overall component
score.

2014 Annual Technical Report
Delmarva Foundation
66
Findings
All MCO aggregate scores exceeded the minimum compliance rate of 75% for the Immunization
component for CY 2013.
The HealthChoice Aggregate score for this component decreased two percentage points in CY 2013,
from 86% in CY 2012 to the current rate of 84%. MCOs were encouraged to continue efforts to improve
administration immunizations according to the DHMH Recommended Childhood and Adolescent
Immunization Schedule.

2014 Annual Technical Report
Delmarva Foundation
67
Health Education/Anticipatory Guidance
Rationale: Health education enables the patient and family to make informed health care decisions.
Anticipatory guidance provides the family with information on what to expect in terms of the child’s current
and next developmental stage. Information should be provided about the benefits of healthy lifestyles and
practices, as well as injury and disease prevention.
Components: A minimum of three topics must be discussed at each Healthy Kids Preventive Care visit.
These topics may include, but are not limited to, social interactions, parenting, nutrition, health, play,
communication, sexuality, and injury prevention. Beginning at 2 years of age, annual routine dental referrals
are required for the purpose of educating the parents about appropriate dental care, providing a cursory view
of the child’s dental health, and familiarizing the child with the dental equipment. Educating the family about
the preventative care schedule and scheduling the next preventive care visit increase the chances of having the
child or adolescent return for future preventive care visits. Additionally, follow-up for missed appointments
needs to occur as soon as possible when the well-child visit is missed to prevent the child or adolescent from
becoming “lost to care.”
Documentation: The primary care provider must specifically document whenever 2-year intervals for
preventive care are the usual and customary schedule of the practice instead of annual visits.
Table 30. CY 2013 Health Education/Anticipatory Guidance Element Scores
Maryland Schedule of
Preventive Health Care
Health Education/
Anticipatory Guidance
ACC
CY 2013
JMS
CY 2013
MPC
CY 2013
MSFC
CY 2013
PPMCO
CY 2013
RHMD
CY 2013
UHC
CY 2013
Provided Education and
Referral to Dentist
79%
95%
77%
80%
85%
N/A
77%
Provided Age Appropriate
Guidance
96%
99%
95%
97%
94%
100%
93%
Specified Requirements
for Return Visit
84%
82%
81%
74%
84%
88%
77%
Provided Ed/Referral for
Identified Problems/Tests
98%
100%
98%
99%
98%
100%
98%
Aggregate Element Rate
89%
94%
88%
87%
90%
96%
86%
__ Denotes that the element score is below 75% which may impact the minimum level compliance score for the component.
Findings
All MCO’s aggregate scores exceeded the minimum compliance rate for the Health
Education/Anticipatory Guidance component for CY 2013.
The CY 2013 HealthChoice Aggregate score for this component was 89%, which is a three percentage
point decrease from 92% in CY 2012.

2014 Annual Technical Report
Delmarva Foundation
68
Figure 1 compares the HealthChoice Aggregate Rates for three reporting periods: January 1 – December 31,
2011 (CY 2011), January 1 – December 31, 2012 (CY 2012), and January 1 – December 31, 2013 (CY 2013).
The HealthChoice Aggregate Total scores have shown very little variation from CY 2011 to CY 2013. Total
scores remained the same from CY 2011 to CY 2012 and decreased by two percentage points from CY 2012
to CY 2013.
The component scores from CY 2011 to CY 2013 have likewise shown little variation. The CY 2012 to CY
2013 component scores decreased in four areas (PE - Comprehensive Physical Exam, LAB – Laboratory
Tests/At-Risk Screenings, IMM – Immunizations, and HED - Health Education/Anticipatory Guidance),
and remained the same in one area (HX – Health and Developmental History).
All component scores remained above the 75% minimum threshold for compliance from CY 2011 to CY
2013.

2014 Annual Technical Report
Delmarva Foundation
69
Corrective Action Plan Process
DHMH sets high performance standards for the Healthy Kids/EPSDT Program. In the event the minimum
compliance score is not met, MCOs are required to submit a CAP. The CAPs are evaluated by Delmarva
Foundation to determine whether the plans are acceptable. In the event that a CAP is deemed unacceptable,
Delmarva Foundation provides recommendations to the MCOs until an acceptable CAP is submitted.
Required Contents of EPSDT CAPs
It is expected that each required CAP will include, at a minimum, the following components:
Methodology for assessing and addressing the problem
Threshold(s) or benchmark(s)
Planned interventions
Methodology for evaluating effectiveness of actions taken
Plans for re-measurement
Timeline for the entire process, including all action steps and plans for evaluation
EPSDT CAP Evaluation
The review team will evaluate the effectiveness of any CAPs initiated as a result of the prior year’s review. A
review of all required EPSDT components are completed annually for each MCO. Since CAPs related to the
review can be directly linked to specific components, the annual EPSDT review will determine whether the
CAPs were implemented and effective. In order to make this determination, Delmarva Foundation will
evaluate all data collected or trended by the MCO through the monitoring mechanism established in the
CAP. In the event that an MCO has not implemented or followed through with the tasks identified in the
CAP, DHMH will be notified for further action.
Conclusions
Five of the seven MCOs (ACC, JMS, MPC, MSFC, and PPMCO) scored above the 75% minimum
compliance score for all five components. RHMD and UHC scored below the 75% minimum compliance
score for the Laboratory Tests/At-Risk Screenings component.
RHMD was not required to submit a CAP during this baseline year of review because its sample size was only
48 records and included five of the twenty-one applicable age groups of children (0 to 1 months, 2 to 3
months, 4 to 5 months, 6 to 8 months, and 9 to 11 months).
UHC was required to submit a CAP. The CAP was evaluated by Delmarva Foundation to determine whether
the plan was acceptable. Delmarva Foundation reviewed the CAP and found it acceptable for the area where
deficiencies occurred for CY 2013.

2014 Annual Technical Report
Delmarva Foundation
70
The result of the EPSDT review demonstrated strong compliance with the timely screening and preventive
care requirements of the Healthy Kids/EPSDT Program. Aggregate scores for each of the five components
remain above the 75% minimum threshold for compliance. UHC submitted a CAP for Laboratory/At-Risk
Screenings.
The CY 2013 Total Composite Score of 87% was a slight two percentage point decrease from the CY 2012
Total Composit Score of 89%. Overall scores demonstrate that the MCOs, in collaboration with PCPs, are
committed to the Department’s goals to provide care that is patient focused and prevention oriented, and
follows the Maryland Schedule of Preventive Health Care.

2014 Annual Technical Report
Delmarva Foundation
71
Section VI
Healthcare Effectiveness Data and Information Set®
Introduction
In accordance with COMAR 10.09.65.03B(2)(a), the HealthChoice MCOs are required to collect HEDIS
®
measures each year based on relevancy to the HealthChoice population. HEDIS
®
is one of the most widely
used sets of healthcare performance measures in the United States. The program is developed and maintained
by NCQA. NCQA develops and publishes specifications for data collection and results calculation in order to
promote a high degree of standardization of HEDIS
®
results. NCQA requires that the reporting entity
register with NCQA and undergo a HEDIS
®
Compliance Audit
TM
.
To ensure a standardized audit methodology, only NCQA-licensed organizations using NCQA-certified
auditors may conduct a HEDIS
®
Compliance Audit. The audit conveys sufficient integrity to HEDIS
®
data,
such that it can be released to the public to provide consumers and purchasers with a means of comparing
healthcare organization performance. DHMH contracted with HealthcareData Company, LLC (HDC), a
NCQA-Licensed Organization, to conduct HEDIS
®
Compliance Audits of all HealthChoice organizations
and to summarize the final results.
In July 2006, DHMH combined two of its programs, Maryland Pharmacy Assistance and Maryland Primary
Care, to form a new Medical Assistance program called Primary Adult Care (PAC). PAC offers healthcare
services to low-income Maryland residents, 19 years of age and older, who are not eligible for full Medicaid
benefits. Four MCOs participated in PAC.
Within DHMH, the HACA is responsible for the quality oversight of the HealthChoice and PAC programs.
DHMH continues to measure HealthChoice program clinical quality performance and enrollee satisfaction
using initiatives including HEDIS
®
reporting. Performance is measured at both the organization level and on
a statewide basis. In 2007, DHMH announced its intention to collect HEDIS
®
results from each organization
offering PAC for a subset of the HEDIS
®
measures already being reported by HealthChoice MCOs. All
seven HealthChoice MCOs submitted CY 2013 data for HEDIS
®
2014. Four PAC MCOs reported CY 2013
data for HEDIS
®
2014.
MCO
HealthChoice
PAC
AMERIGROUP Community Care
X
X
Jai Medical Systems
X
X
Maryland Physicians Care
X
MedStar Family Choice, Inc.
X
Priority Partners
X
X
Riverside Health of Maryland
X
UnitedHealthcare
X
X

2014 Annual Technical Report
Delmarva Foundation
72
Measures Designated for Reporting
Annually, DHMH determines the set of measures required for HEDIS
®
reporting. DHMH selects these
measures because they provide meaningful MCO comparative information and they measure performance
pertinent to DHMH’s priorities and goals.
Measures Selected by DHMH for HealthChoice Performance Reporting
DHMH required HealthChoice MCOs to report 32 HEDIS
®
measures for services rendered in CY 2013.
This required set reflected five additional measures for reporting:
Asthma Medication Ration (AMR)
Use of Sprirometry Testing in the Assessment and Diagnosis of COPD (SPR)
Pharmacotherapy Management of COPD Exacerbation (PCE)
Persistence of Beta Blocker Treatment After a Heart Attack (PBH)
Weight Assessment and Counseling for Nutrition and Physical Activity for Children and Adolescents
(WCC)
The HEDIS
®
Performance Measures are:
Effectiveness of Care
Childhood Immunization Status (CIS)
Immunizations for Adolescents (IMA)
Breast Cancer Screening (BCS)
Cervical Cancer Screening (CCS)
Comprehensive Diabetes Care, all indicators except HbA1c <7.0% (CDC)
Use of Appropriate Medications for People with Asthma (ASM)
Appropriate Treatment for Children with Upper Respiratory Infection (URI)
Appropriate Testing for Children with Pharyngitis (CWP)
Avoidance of Antibiotic Treatment in Adults with Acute Bronchitis (AAB)
Chlamydia Screening in Women (CHL)
Use of Imaging Studies for Low Back Pain (LBP)
Annual Monitoring for Patients on Persistent Medications (MPM)
Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis (ART)
Medication Management for People with Asthma (MMA)
Controlling High Blood Pressure (CBP)
Adult BMI Assessment (ABA)
Asthma Medication Ration (AMR)
Use of Sprirometry Testing in the Assessment and Diagnosis of COPD (SPR)
Pharmacotherapy Management of COPD Exacerbation (PCE)

2014 Annual Technical Report
Delmarva Foundation
73
Persistence of Beta Blocker Treatment After a Heart Attack (PBH)
Weight Assessment and Counseling for Nutrition and Physical Activity for Children and Adolescents
(WCC)
Access/Availability of Care
Adults' Access to Preventive/Ambulatory Health Services (AAP)
Children and Adolescents' Access to Primary Care Practitioners (CAP)
Prenatal and Postpartum Care (PPC)
Call Answer Timeliness (CAT)
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET)
Utilization and Relative Resource Use
Frequency of Ongoing Prenatal Care (FPC)
Well-Child Visits in the First 15 Months of Life (W15)
Well-Child Visits in the Third, Fourth, Fifth and Sixth Years of Life (W34)
Adolescent Well-Care Visits (AWC)
Ambulatory Care (AMB)
Identification of Alcohol and Other Drug Services (IAD)
Measures Selected by DHMH for PAC Performance Reporting
DHMH required PAC MCOs to report 5 HEDIS
®
measures for services rendered in CY 2013:
Effectiveness of Care
Breast Cancer Screening (BCS)
Cervical Cancer Screening (CCS)
Comprehensive Diabetes Care, all indicators except HbA1c <7.0% (CDC)
Avoidance of Antibiotic Treatment in Adults with Acute Bronchitis (AAB)
Access/Availability of Care
Adults' Access to Preventive/Ambulatory Health Services (AAP)
HEDIS
®
Measures Reporting History
The following table shows the history of DHMH required reporting. A notation of < 2005 indicates that
DHMH chose to report the measure since at least 2005. The year refers to the HEDIS
®
-reporting year.

2014 Annual Technical Report
Delmarva Foundation
74
NCQA Domain
Measure Name
Indicators
(Indicators reported for HEDIS
®
but not included in this
report are italicized.)
HealthChoice
reporting history
PAC reporting
history
Prevention and Screening – Adult and Child
EOC
Adult BMI Assessment (ABA)
2013
EOC
Avoidance of Antibiotic Treatment in Adults
with Acute Bronchitis (AAB)
2012
2012
EOC
Childhood Immunization Status
(CIS)
DTaP; IPV; MMR; HiB; Hepatitis B;VZV
Combination 2
1
< 2005
Pneumococcal conjugate
Combination 3
1
2006
Hepatitis A; Rotavirus; Influenza
Combinations 4,5,6,7,8,9, and 10
1
2010
EOC
Immunizations for Adolescents
(IMA)
Meningococcal; Tdap/Td
Combination 1 (Meningococcal, Tdap/Td)
2010
URR
Well-Child Visits in the First 15
Months of Life (W15)
No visits; One visit; Two visits; Three visits; Four visits; Five
visits; Six or more visits
DHMH non-HEDIS measure: Five or six-or-more visits
(additive percentage of HEDIS five visits and six-or-more
visits rates)
< 2005
URR
Well-Child Visits in the Third, Fourth,
Fifth and Sixth Years of Life (W34)
< 2005
URR
Adolescent Well-Care Visits (AWC)
< 2005
EOC
Weight Assessment and Counseling for
Nutrition and Physical Activity for
Children/Adolescents (WCC)
2014
EOC
Appropriate Testing for Children with
Pharyngitis (CWP)
2007
*Domain abbreviations: EOC: Effectiveness of Care, AAC: Access/Availability of Care, URR: Utilization and Relative Resource Use.
1. Please refer to the table on page 12 for delineation of antigens included in each combination.
The table is continued on the next page

2014 Annual Technical Report
Delmarva Foundation
75
Respiratory Conditions
EOC
Use of Appropriate Medications
for People with Asthma (ASM)
<2009: 5-9 years of age; 10-17 years of age; 18-56 years of age;
Total (5-56 years of age)
2006
2010: 5-11 years of age; 12-50 years of age; Total (5-
50 years of age)
2012: 5-11 years of age; 12-18 years of age; 19-50 years of age; 51-64
years of age; Total (5-64 years of age); DHMH non-HEDIS measure: Total
(5-50 years of age) – additive percentage of HEDIS 5-11 yrs, 12-18 yrs,
19-50 yrs.
EOC
Medication Management for
People With Asthma (MMA)
Percentage of members who remained on an asthma controller
medication for at least 50% of their treatment period
Percentage of members who remained on an asthma controller
medication for at least 75% of their treatment period
2013
EOC
Appropriate Treatment for Children
with Upper Respiratory Infection
(URI)
2007
EOC
Asthma Medication Ration (AMR)
2014
EOC
Use of Sprirometry Testing in the
Assessment and Diagnosis of COPD
(SPR)
2014
EOC
Pharmacotherapy Management of
COPD Exacerbation (PCE)
2014
Member Access
AAC
Children and Adolescents' Access
to Primary Care Practitioners
(CAP)
12-24 months of age
25 months-6 years of age
7-11 years of age
12-19 years of age
2007
AAC
Adults' Access to Preventive/
Ambulatory Health Services
(AAP)
20-44 years of age
45-65 years of age
2007
2009
Women’s Health
EOC
Breast Cancer Screening (BCS)
2007
2009
EOC
Cervical Cancer Screening (CCS)
2007
2009
EOC
Chlamydia Screening in Women
(CHL)
16-20 years of age
2007
2009: 21-25 years of age
2007-2008: 21-24 years of age
2007
2009: Total (16-24 years of age)
2007-2008: Total (16-25 years of age)
2007
Prenatal & Postpartum Care
AAC
Prenatal and Postpartum Care
(PPC)
Timeliness of prenatal care
< 2005
Postpartum care
< 2005
URR
Frequency of Ongoing Prenatal
Care (FPC)
<21 percent of expected visits
21 percent of expected visits
41 percent of expected visits
61 percent of expected visits
>81 percent of expected visits
< 2005
*Domain abbreviations: EOC: Effectiveness of Care, AAC: Access/Availability of Care, URR: Utilization and Relative Resource Use
NCQA Domain
*
Measure Name
Indicators
(Indicators reported for HEDIS
®
but not included in this
report are italicized.)
HealthChoice
reporting history
PAC reporting
history
Prevention and Screening – Adult and Child

2014 Annual Technical Report
Delmarva Foundation
76
* Domain abbreviations: EOC: Effectiveness of Care, AAC: Access/Availability of Care, URR: Utilization and Relative Resource Use
NCQA Domain
*
Measure Name
Indicators
(Indicators reported for HEDIS
®
but not included in this
report are italicized.)
HealthChoice
reporting history
PAC reporting
history
Cardiovascular Conditions
EOC
Controlling High Blood Pressure (CBP)
2013
EOC
Persistence of Beta-Blocker Treatment
After a Heart Attack (PBH)
2014
Diabetes
EOC
Comprehensive Diabetes Care (CDC)
HbA1c testing
< 2005
2009
HbA1c poor control (>9.0%)
< 2005
2009
HbA1c control (<8.0%)
2009
2009
Eye exam (retinal) performed
< 2005
2009
LDL-C screening
2007
2009
LDL-C control (<100mg/dL)
Medical attention for nephropathy
Blood pressure control (<140/80 mm Hg)
2011
2011
Blood pressure control (<140/90 mm Hg)
2007
2009
Musculoskeletal Conditions
EOC
Use of Imaging Studies for Low
Back Pain (LBP)
2012
EOC
Disease-Modifying Anti- Rheumatic
Drug Therapy for Rheumatoid
Arthritis (ART)
2013
Medication Management
EOC
Annual Monitoring for Patients on
Persistent Medications(MPM)
Members on angiotensin converting enzyme (ACE) inhibitors
or angiotensin receptor blockers (ARB) Digoxin Diuretics
Anticonvulsants Total Rate
2013
Behavioral Health
AAC
Initiation and Engagement of
Alcohol and Other Drug Dependence
Treatment (IET)
Initiation:
13-17 years of age
18+ years of age
Total (ages 13-65)
Engagement:
13-17 years of age
18+ years of age
Total (ages 13-65)
2009
URR
Identification of Alcohol and
Other Drug Services (IAD)
Any services
Inpatient services
Intensive Outpatient/Partial Hospitalization
Outpatient/ED
2009
Ambulatory Care (Utilization)
URR
Ambulatory Care (AMB)
Outpatient visits
ED visits
Note: Ambulatory Surgery/Procedures and Observation
Room Stays categories were retired in 2011
2007
Call Services
AAC
Call Answer Timeliness (CAT
2006

2014 Annual Technical Report
Delmarva Foundation
77
HEDIS
®
Methodology
The HEDIS
®
-reporting organization follows guidelines for data collection and specifications for measure
calculation described in HEDIS
®
2014 Volume 2: Technical Specifications.
Data collection: The organization pulls together all data sources, typically into a data warehouse, against
which HEDIS
®
software programs are applied to calculate measures. Three approaches may be taken for data
collection:
Administrative data: Data from transaction systems (claims, encounters, enrollment, practitioner) provide
the majority of administrative data. Organizations may receive encounter files from pharmacy, laboratory,
vision, and behavioral health vendors.
Supplemental data: NCQA defines supplemental data as atypical administrative data, i.e., not claims or
encounters. Sources include immunization registry files, laboratory results files, case management databases,
and medical record-derived databases.
Medical record data: Data abstracted from paper or electronic medical records may be applied to certain
measures, using the NCQA-defined hybrid method. HEDIS
®
specifications describe statistically sound
methods of sampling, so that only a subset of the eligible population’s medical records needs to be chased.
NCQA specifies hybrid calculation methods, in addition to administrative methods, for several measures
selected by DHMH for HEDIS
®
reporting:
Childhood Immunization Status (CIS)
Immunizations for Adolescents (IMA)
Cervical Cancer Screening (CCS)
Comprehensive Diabetes Care (CDC)—HbA1c testing; poor control >9.0; control <8.0*
Comprehensive Diabetes Care (CDC)—Eye exam (retinal) performed
Comprehensive Diabetes Care (CDC)—LDL-C screening; LDL-C control <100mg/dL*
Comprehensive Diabetes Care (CDC)—Medical attention for nephropathy
Comprehensive Diabetes Care (CDC)—Blood pressure control <140/90 mm Hg;
Comprehensive Diabetes Care (CDC)—Blood pressure control <140/80 mm Hg*
Prenatal and Postpartum Care (PPC)
Frequency of Ongoing Prenatal Care (FPC)
Well-Child Visits in the First 15 Months of Life (W15)
Well-Child Visits in the Third, Fourth, Fifth and Sixth Years of Life (W34)
Adolescent Well-Care Visits (AWC)
Adult BMI Assessment (ABA)
Controlling High Blood Pressure (CBP)
Weight Assessment and Counseling for Nutrition and Physical Activity for Children/Adolescents (WCC)
Use of the hybrid method is optional. NCQA maintains that no one approach to measure
calculation or data collection is considered superior to another. From organization to
organization, the percentages of data obtained from one data source versus another are highly

2014 Annual Technical Report
Delmarva Foundation
78
variable, making it inappropriate to make across-the-board statements about the need for, or
positive impact of, one method versus another. In fact, an organization’s yield from the hybrid
method may impact the final rate by only a few percentage points, an impact that is also
achievable through improvement of administrative data systems.
* An organization must use the same method for the group of indicators.
The following table shows actual HEDIS
®
2014 use of the administrative or hybrid method. The choice of
using the administrative vs. hybrid method is usually one of available resources. The hybrid method takes
significant resources to perform.
Measure
ACC
HC
JMS
HC
MPC
HC
MSFC
HC
PP
HC
RH
HC
UHC
HC
ACC
PAC
JMS
PAC
MPC
PAC
PP
PAC
UHC
PAC
CIS
H
H
H
H
H
A
H
IMA
H
H
A
H
H
A
H
W15
H
H
H
H
H
A
H
W34
H
H
A
H
H
A
H
AWC
H
H
A
H
H
A
H
CCS
H
H
A
H
H
A
H
A
A
A
A
A
PPC Pre
H
H
H
H
H
A
H
PPC Post
H
H
H
H
H
A
H
FPC
H
H
A
A
H
A
H
CDC - HbA1c
testing
H
H
H
H
H
A
H
A
H
A
A
A
CDC - HbA1c
Poor Control
H
H
H
H
H
A
H
A
H
A
A
A
CDC HbA1c
Control
(<8.0%)
H
H
H
H
H
A
H
A
H
A
A
A
CDC - Eye
exam retinal)
performed
H
H
H
H
H
A
H
A
H
A
A
A
CDC - LDL-C
screening and
control
H
H
H
H
H
A
H
A
H
A
A
A
CDC - Medical
attention for
nephropathy
H
H
H
H
H
A
H
A
H
A
A
A
CDC - Blood
pressure control
140/80
H
H
H
H
H
A
H
A
H
A
A
A
CDC – Blood
pressure control
140/90
H
H
H
H
H
A
H
A
H
A
A
A
ABA
H
A
H
H
H
A
H
CBP
H
H
H
H
H
A
H
WCC
H
H
H
H
H
A
H
H – Hybrid A - Administrative
HC – HealthChoice PAC - Primary Adult Care

2014 Annual Technical Report
Delmarva Foundation
79
HEDIS
®
Audit Protocol
The HEDIS
®
auditor follows NCQA’s Volume 5: HEDIS
®
Compliance Audit
TM
: Standards, Policies, and
Procedures described briefly below:
Offsite preparation for the onsite audit: To prepare the MCOs for the upcoming audit, HDC takes the
following steps:
Conference call: A conference call is held to introduce key personnel, review the onsite agenda,
identify session participants, and determine a plan to audit data sources used for HEDIS
®
.
HEDIS
®
Roadmap review: Each MCO must complete the HEDIS
®
Record of Administration,
Data Management, and Processes (ROADMAP). The Roadmap includes detailed questions about all
audit standards and describes the operational and organizational structure of the organization. The
auditor reviews the HEDIS
®
Roadmap to make preliminary assessments regarding information
systems compliance and to identify areas requiring follow-up at the onsite audit.
Information Systems (IS) standards compliance: The onsite portion of the HEDIS
®
Audit that
expands upon information gleaned from the HEDIS
®
Roadmap to enable the auditor to make
conclusions about the organization’s compliance with IS standards. IS standards, describing the minimum
requirements for information systems and processes used in HEDIS
®
data collection, are the foundation
on which the auditor assesses the organization’s ability to report HEDIS
®
data accurately and reliably.
The auditor reviews data collection and management processes, including the monitoring of vendors, and
makes a determination regarding the soundness and completeness of data to be used for HEDIS
®
reporting.
HEDIS
®
Measure Determination (HD) standards compliance: The auditor uses both onsite and
offsite activities to determine compliance with HD standards and to assess the organization’s adherence
to HEDIS
®
Technical Specifications and report-production protocols. The auditor confirms the use of
NCQA certified software. (All Maryland Medicaid organizations continue to use certified software to
produce HEDIS
®
reports.) The auditor reviews the organization’s sampling protocols for the hybrid
method. Later in the audit season, the auditor reviews HEDIS
®
results for algorithmic compliance and
performs benchmarking against NCQA-published means and percentiles.
Medical record review validation (MRRV): The HEDIS
®
audit includes a protocol to validate the
integrity of data obtained from medical record review (MRR) for any measures calculated using the
hybrid method. The audit team compares its medical record findings to the organization’s abstraction
forms for a sample of positive numerator events. Part one of the validation may also include review of a
convenience sample of medical records for the purpose of finding procedural errors early in the medical
record abstraction process so that timely corrective action can be made. This is optional based on NCQA
standards and auditor opinion. MRRV is an important component of the HEDIS
®
Compliance Audit. It
ensures that medical records reviews performed by the organization, or by its contracted vendor, meet
audit standards for sound processes and that abstracted medical data are accurate. In part two of the
MRRV, the auditor selects hybrid measures from like-measure groupings for measure validation. MRRV
tests medical records and appropriate application of the HEDIS
®
hybrid specifications (i.e., the member

2014 Annual Technical Report
Delmarva Foundation
80
is a numerator negative, a numerator positive or an exclusion for the measure). NCQA uses an acceptable
quality level of 2.5 percent for the sampling process, which translates to a sample of 16 medical records
for each selected measure.
Audit designations: The auditor approves the rate/result of each measure included in the HEDIS
®
report, as shown in the table of audit results, excerpted from Volume 5: HEDIS
®
Compliance Audit
TM
:
Standards, Policies, and Procedures.
Rate/Result
Comment
O-XXX
Reportable rate or numeric result for HEDIS
®
measures
NR
Not Reported:
1. Plan chose not to report.*
2. Calculated rate was materially biased.
NA
Small Denominator: The organization followed the specifications but the denominator
was too small to report a valid rate.
* An organization may exercise this option only for those measures not included in the measurement set required by DHMH.
Bias Determination: If the auditor determines that a measure is biased, the organization cannot report a
rate for that measure and the auditor assigns the designation of NR. Bias is based on the degree of error
or data completeness for the data collection method used. NCQA defines four bias determination rules,
applied to specific measures. These are explained in Appendix 10 of Volume 5: HEDIS
®
Compliance
Audit
TM
: Standards, Policies and Procedures.
Final Audit Opinion: At the close of the audit, the auditor renders the Final Audit Opinion, containing
a Final Audit Statement along with measure-specific rates/results and comments housed in the Audit
Review Table.
Measure-specific Findings – Explanation
Two metrics are calculated to accompany the MCO-specific scores:
Maryland Average Reportable Rate (MARR): The MARR is an average of HealthChoice MCO’s rates
as reported to NCQA. In most cases, all seven MCOs contributed a rate to the average. Where one or
more organizations reported NA or NR instead of a rate, the average consisted of fewer than seven
component rates.
National HEDIS
®
Mean (NHM): The mean value is taken from NCQA’s HEDIS
®
Audit Means,
Percentiles and Ratios – Medicaid, released each year to HEDIS
®
auditors and reporting organizations. The
NCQA data set gives prior-year rates for each measure displayed as the mean rate and the rate at the 5th,
10th, 25th, 50th, 75th, 90th and 95th percentiles. HEDIS
®
2013 Means, Percentiles, and Ratios pertinent to
this report, as well as additional rates for measure components are reported to the Department. Any
questions regarding such rates can be directed to the Department. NCQA averages the rates of all
organizations submitting HEDIS
®
results, regardless of the method of calculation (administrative or
hybrid). NCQA’s method is the same as that used for the MARR, but on a larger scale.

2014 Annual Technical Report
Delmarva Foundation
81
Year-to-year trending: Year-to-year trending is possible when specifications remain consistent from
year to year. (Expected updates to industry-wide coding systems are not considered specification
changes.) For each measure, the tables display up to five-years of results, where available.
Prior year results are retained in the trending tables, regardless of specification changes. Text in italics
notes when prior-year results fall under different specifications. Performance trends at the organization
level are juxtaposed with the trends for the MARR and the NHM for the same measurement year.
Rounding of figures: Rates are rounded to one decimal point from the rate/ratio reported to NCQA. This
rounding corresponds to the rounding used by NCQA for the NHM. Where any two or more rates are
identical at this level of detail, an additional decimal place of detail is provided.
Audit designation other than a rate/ratio: According to NCQA reporting protocols, NA or NR may
replace a rate.
Organization of data: The following pages contain the comparative results for HEDIS
®
2014. This report
does group and sequence measures by like populations or functions.
Prevention and Screening-Adult: ABA, AAB
Prevention and Screening-Child: CIS, IMA, W15, W34, AWC, WCC, CWP
Respiratory Conditions: ASM, MMA, URI, AMR, SPR, PCE
Member Access: CAP, AAP
Women’s Health: BCS, CCS, CHL
Prenatal and Postpartum Care: PPC, FPC
Cardiovascular Conditions: CBP, PBH
Diabetes: CDC
Musculoskeletal Conditions: LBP, ART
Medication Management: MPM
Behavioral Health: IET, IAD
Ambulatory Care (utilization): AMB
Call Services: CAT
Sources of accompanying information:
Description – The source of the information is NCQA’s HEDIS
®
2014 Volume 2: Technical
Specifications.
Rationale – For all measures, except Call Answer Timeliness (CAT) the source of the information is the
Agency for Healthcare Research and Quality (AHRQ) citations of NCQA as of 2013. These citations
appear under the Brief Abstract on the Web site of the National Quality Measures Clearinghouse,
http://www.qualitymeasures.ahrq.gov/. For CAT the rationale was adapted from HEDIS
®
2004 Vol. 2:
Technical Specifications.

2014 Annual Technical Report
Delmarva Foundation
82
Summary of Changes for HEDIS
®
2014 – The source of the text, is the HEDIS
®
2014 Volume 2:
Technical Specifications, incorporating additional changes published in the HEDIS
®
2014 Volume 2:
“October” Technical Update.
Year-to-year Changes
Table 31 shows the numbers of organizations that experienced a lower or higher change in HEDIS
®
rates from 2013 to 2014. The change in the MARR (2014 rate minus 2013 rate) and the change in the
NHM (2013 rate minus 2012 rate) place Maryland HealthChoice organization trends in perspective.
For measures where a lower rate indicates better performance (single asterisk), the number of lower
performing organizations appears in the higher column and the number of higher performing
organizations appear in the lower column. New measures or indicators with no trendable history are
not included in this analysis of change. HEDIS
®
2014 results of NA are not included in tallies. Rates
that
stayed the same from last year and did not increase or decrease are not included in this table.
Table 31. Changes in HEDIS
®
Rates from 2013 to 2014
HEDIS
®
Measure
eeeeeee
e
Lower
Higher
MARR
change
NHM
change
Adult BMI Assessment (ABA)
1
5
11
14.9
Avoidance of Antibiotic Treatment in Adults with Acute Bronchitis (AAB)
1
5
3.1
-0.1
Childhood Immunization Status (CIS) – Combination 2
3
3
0.7
1.2
Childhood Immunization Status (CIS) – Combination 3
3
3
1.4
1.5
Childhood Immunization Status (CIS) – Combination 4
3
3
1.2
26.7
Childhood Immunization Status (CIS) – Combination 5
2
4
4.4
3.4
Childhood Immunization Status (CIS) – Combination 6
3
3
2.3
3.9
Childhood Immunization Status (CIS) – Combination 7
2
4
4.4
21.6
Childhood Immunization Status (CIS) – Combination 8
2
4
1.8
16.4
Childhood Immunization Status (CIS) – Combination 9
2
4
3.6
4.2
Childhood Immunization Status (CIS) – Combination 10
2
4
3.5
14.1
Immunizations for Adolescents (IMA) – Combination 1
0
6
5.6
6.7
Well-Child Visits in the First 15 months of Life (W15) – No well-child visits*
2
4
-0.1
-0.1
Well-Child Visits in the First 15 months of Life (W15) – DHMH Five or Six-or-more
visits rates**
3
3
1.8
1.3
Well-Child Visits in the Third, Fourth, Fifth and Sixth Years of Life (W34)
1
5
1.8
0
Adolescent Well-Care Visits (AWC)
4
2
1.9
0
Appropriate Testing for Children with Pharyngitis (CWP)
1
5
-0.2
1.3
Use of Appropriate Medications for People With Asthma (ASM) – Ages 5-11
4
2
0.3
-0.8
Use of Appropriate Medications for People With Asthma (ASM) – Ages 12-18
4
2
-2.5
-1.0
Use of Appropriate Medications for People With Asthma (ASM) – Ages 19-50
6
0
-6.8
-1.7
Use of Appropriate Medications for People With Asthma (ASM) – Ages 51-64
5
-7.4
-1.5
Use of Appropriate Medications for People With Asthma (ASM) – Ages 5-64
5
1
-2.7
-1.1
Medication Management for People With Asthma (MMA) – Total 50% of treatment
period
2
4
3.4
-1.2

2014 Annual Technical Report
Delmarva Foundation
83
HEDIS
®
Measure
eeeeeee
e
Lower
Higher
MARR
change
NHM
change
Medication Management for People With Asthma (MMA) – Total 75% of treatment
period
3
3
1.5
-1.4
Appropriate Treatment for Children with Upper Respiratory Infection (URI)
2
4
0.3
-0.2
Children and Adolescents’ Access to Primary Care Practitioners (CAP) – Age 12-24
months
4
2
1.0
-0.1
Children and Adolescents’ Access to Primary Care Practitioners (CAP) – Age 25
months-6 years
3
2
0.5
0.1
Children and Adolescents’ Access to Primary Care Practitioners (CAP) – Age 7-11
years
1
5
0.8
0.4
Children and Adolescents’ Access to Primary Care Practitioners (CAP) – Age 12-19
years
2
4
0.9
0.5
Adults’ Access to Preventive/Ambulatory Health Services (AAP) – Age 20-44
5
1
-0.7
0.4
Adults’ Access to Preventive/Ambulatory Health Services (AAP) – Age 45-64
2
4
1.1
0.5
Breast Cancer Screening (BCS)
0
6
7.3
1.5
Cervical Cancer Screening (CCS)
2
4
1.5
-2.2
Chlamydia Screening in Women (CHL) – Age 16-20 years
4
2
-0.6
-1.4
Chlamydia Screening in Women (CHL) – Age 21-24 years
3
3
0.0
0.2
Chlamydia Screening in Women (CHL) – Total, 16-24 years of age
4
2
-0.6
-0.9
Prenatal and Postpartum Care (PPC) – Timeliness of prenatal care
3
3
-11.8
0.1
Prenatal and Postpartum Care (PPC) – Postpartum care
2
4
-8.1
-1.0
Frequency of Ongoing Prenatal Care (FPC) – Less than 21% of expected visits*
2
3
3.4
2.3
Frequency of Ongoing Prenatal Care (FPC) – Greater than or equal to 81% of
expected visits
3
3
-5.5
-0.4
Controlling High Blood Pressures (CBP)
3
3
3
-0.5
Comprehensive Diabetes (CDC) – HbA1c testing
2
4
4.3
0.5
Comprehensive Diabetes (CDC) – HbA1c poor control (>9.0%)*
4
2
-2.8
1.7
Comprehensive Diabetes (CDC) – HbA1c control (< 8.0%)
2
4
2.4
-1.6
Comprehensive Diabetes (CDC) – Eye exam (retinal) performed
5
1
-0.3
-0.2
Comprehensive Diabetes (CDC) – LDL-C screening
2
4
1.5
0.5
Comprehensive Diabetes (CDC) – LDL-C control (<100 mg/dL)
2
4
2.6
-1.3
Comprehensive Diabetes (CDC) – Medical attention for nephropathy
2
4
1.7
0.6
Comprehensive Diabetes (CDC) – Blood pressure control (<140/80 mm Hg)
1
5
1.3
-1.6
Comprehensive Diabetes (CDC) – Blood pressure control (<140/90 mm Hg)
1
5
2.2
-2.1
Use of Imaging Studies for Low Back Pain (LBP)
2
4
0.5
-0.2
Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis (ART)
3
1
-1.8
1.0
Annual Monitoring for Patients on Persistent Medications (MPM) – members on
angiotensin coverting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARB)
4
2
0.2
0.4
Annual Monitoring for Patients on Persistent Medications (MPM) – members on
digoxin
3
1
-2.3
-0.1
Annual Monitoring for Patients on Persistent Medications (MPM) – members on
diuretics
4
2
0.3
0.6
Annual Monitoring for Patients on Persistent Medications (MPM) – members on
anticonvulsants
1
5
2.9
0.6
Annual Monitoring for Patients on Persistent Medications (MPM) – Total rate
2
3
0.8
0.6
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET) –
Initiation 13-17 years
2
3
4.6
-1.4

2014 Annual Technical Report
Delmarva Foundation
84
HEDIS
®
Measure
eeeeeee
e
Lower
Higher
MARR
change
NHM
change
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET) –
Initiation 18+ years
4
2
0.7
0.0
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET)
– Initiation overall ages
4
2
0.9
0.2
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET) –
Engagement 13-17 years
2
3
4.1
-0.9
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET) –
Engagement 18+ years
2
4
2.7
-1.3
Initiation and Engagement of Alcohol and Other Drug Dependence Treatment (IET) –
Engagement overall ages
2
4
2.6
-1.0
Identification of Alcohol and Other Drug Services (IAD) – Any
2
4
1.9
1.2
Identification of Alcohol and Other Drug Services (IAD) – Inpatient
4
2
0.1
0.4
Identification of Alcohol and Other Drug Services (IAD) – Intensive
outpatient/partial hospitalization
3
1
0.3
0.6
Identification of Alcohol and Other Drug Services (IAD) – Outpatient/ED
2
4
1.4
-0.5
Ambulatory Care (AMB) – Outpatient visits
5
1
-2.1
23.8
Ambulatory Care (AMB) – Emergency department*
0
6
-6.4
3.5
Call Answer Timeliness (CAT)
3
3
-0.2
0.6
* A lower rate indicates better performance.
** Not a HEDIS
®
sub-measure; HDC is calculating for DHMH trending purposes.
NA – NHM change cannot be calculated since these age groups first started in 2012
Three-year trends: The following table shows organizations that demonstrated incremental increases
in performance scores over the past three years. The analysis only shows a trend toward
improvement. It does not indicate superior performance. For a comparison of one organization
against another, please
refer
to the measure-specific tables in this report. For measures where a lower
rate indicates better performance (single asterisk), the table shows organizations having a decrease in
performance score over the past three years.
Table 32. HEDIS
®
Measures Incremental Increases in Performance
HEDIS
®
Measure
ACC
JMS
MPC
MSFC
PPMCO
RHMD
UHC
Avoidance of Antibiotic Treatment in Adults with Acute Bronchitis
(AAB)
X
Childhood Immunization Status (CIS) – Combination 2
X
Childhood Immunization Status (CIS) – Combination 3
X
Childhood Immunization Status (CIS) – Combination 4
X
X
X
Childhood Immunization Status (CIS) – Combination 5
X
X
Childhood Immunization Status (CIS) – Combination 6
X
Childhood Immunization Status (CIS) – Combination 7
X
X
X
X
Childhood Immunization Status (CIS) – Combination 8
X
X
X
X
Childhood Immunization Status (CIS) – Combination 9
X
X
Childhood Immunization Status (CIS) – Combination 10
X
X
X
X
Immunizations for Adolescents (IMA) – Combination 1
X
X
X
X

2014 Annual Technical Report
Delmarva Foundation
85
HEDIS
®
Measure
ACC
JMS
MPC
MSFC
PPMCO
RHMD
UHC
Well-Child Visits in the First 15 months of Life (W15) – No well-child
visits*
X
Well-Child Visits in the First 15 months of Life (W15) – DHMH Five or
Six-or-more visits rates (additive)**
Well-Child Visits in the Third, Fourth, Fifth and Sixth Years of Life
(W34)
Adolescent Well-Care Visits (AWC)
X
Appropriate Testing for Children with Pharyngitis (CWP)
X
X
X
X
Use of Appropriate Medications for People with Asthma (ASM) –
Ages 5-11
Use of Appropriate Medications for People with Asthma (ASM) –
Ages 12-18
Use of Appropriate Medications for People with Asthma (ASM) –
Ages 19-50
Use of Appropriate Medications for People with Asthma (ASM) –
Ages 51-64
Use of Appropriate Medications for People with Asthma (ASM) –
Ages 5-64
Use of Appropriate Medications for People with Asthma (ASM) –
Total combined ages 5-50**
Appropriate Treatment for Children with Upper Respiratory Infection
(URI)
Children and Adolescents’ Access to Primary Care Practitioners (CAP)
– Age 12-24 months
X
Children and Adolescents’ Access to Primary Care Practitioners (CAP)
– Age 25 months-6 years
Children and Adolescents’ Access to Primary Care Practitioners (CAP)
– Age 7-11 years
X
X
Children and Adolescents’ Access to Primary Care Practitioners (CAP)
– Age 12-19 years
X
X
X
Adults’ Access to Preventive/Ambulatory Health Services (AAP) –
Age 20-44
Adults’ Access to Preventive/Ambulatory Health Services (AAP) –
Age 45-64
X
X
Breast Cancer Screening (BCS)
X
X
X
X
X
Cervical Cancer Screening (CCS)
X
X
Chlamydia Screening in Women (CHL) – Age 16-20 years
Chlamydia Screening in Women (CHL) – Age 21-24 years
Chlamydia Screening in Women (CHL) – Total (16-24) years
Prenatal and Postpartum Care (PPC) – Timeliness of prenatal care
X
X
Prenatal and Postpartum Care (PPC) – Postpartum care
X
Frequency of Ongoing Prenatal Care (FPC) – Less than 21% of
expected visits*
X
Frequency of Ongoing Prenatal Care (FPC) – Greater than or equal to
81% of expected visits
Controlling High Blood Pressures (CBP)
X
X
Persistence of Beta-Blocker Treatment After a Heart Attack (PBH)
X
X
Comprehensive Diabetes (CDC) – Hemoglobin A1c testing
X
Comprehensive Diabetes (CDC) – Hemoglobin A1c poor control
(>9.0%)*
Comprehensive Diabetes (CDC) – Hemoglobin A1c control (<8.0%)
X
Comprehensive Diabetes (CDC) – Eye exam (retinal) performed
X
X

2014 Annual Technical Report
Delmarva Foundation
86
HEDIS
®
Measure
ACC
JMS
MPC
MSFC
PPMCO
RHMD
UHC
Comprehensive Diabetes (CDC) – LDL-C screening
X
Comprehensive Diabetes (CDC) – LDL-C control (<100 mg/dL)
X
X
X
Comprehensive Diabetes (CDC) – Medical attention for nephropathy
X
X
Comprehensive Diabetes (CDC) – Blood pressure control (<140/80
mm Hg)
X
Comprehensive Diabetes (CDC) – Blood pressure control (<140/90
mm Hg)
Use of Imaging Studies for Low Back Pain (LBP)
X
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Initiation 13-17 years
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Initiation 18+ years
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Initiation overall ages
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Engagement 13-17 years
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Engagement 18+ years
X
Initiation and Engagement of Alcohol and Other Drug Dependence
Treatment (IET) – Engagement overall ages
Identification of Alcohol and Other Drug Services (IAD) – Any
X
Identification of Alcohol and Other Drug Services (IAD) – Inpatient
X
Identification of Alcohol and Other Drug Services (IAD) – Intensive
Outpatient / Partial Hospitalization
Identification of Alcohol and Other Drug Services (IAD) – Outpatient /ED
X
X
Ambulatory Care (AMB) – Outpatient visits per 1,000 member months
Ambulatory Care (AMB) – Emergency department (ED) visits per 1,000
member months
Call Answer Timeliness (CAT)
X
X
TOTALS
17
12
9
10
10
0
16
* A lower rate indicates better performance. ** Not a HEDIS
®
sub-measure; HDC is calculating for DHMH trending purposes.
Findings
Implications
HEDIS
®
rates are widely used and respected standardized quality indicators. As with any measurement tool, it
is important to understand uses and limitations. HEDIS
®
results can be used as markers of care, but cannot
be used, on their own, to draw conclusions about the quality of care. A comparison among organizations on
the basis of HEDIS
®
rates alone would not take into account population differences, such as age, health
status, or catchment area (urban vs. rural). For example, Maryland Medicaid organizations are dissimilar in
location served: two organizations operate statewide (ACC and UHC), four are regional (MPC, MSFC,
PPMCO, and RHMD), and one operates in Baltimore City and parts of Baltimore County (JMS). The effect
of these geographic locations on HEDIS
®
rates is unknown.

2014 Annual Technical Report
Delmarva Foundation
87
Year-to-year trends: Trends in rates, as shown in the tables can indicate genuine improvement or can indicate
something else, e.g., familiarity with HEDIS
®
reporting or improved data systems. Significant changes (up or
down) from HEDIS
®
2013 to HEDIS
®
2014 include:
HealthChoice
The MARR for CIS Combo 10 (all immunizations) increased 3.5 percentage points
The MARR for Adult BMI Assessment increased 11 percentage points
The MARR for the ASM (5-50) measure decreased 2.5 percentage points
The MARR for Breast Cancer Screening increased 7.3 percentage points
The MARR for Timeliness of Prenatal Care (PPC measure) decreased 11.8 percentage points
The MARR for Post Partum Care (PPC measure) decreased 8.9 percentage points
The MARR for the IMA measure, Combo 1) increased 3.5 percentage points
Primary Adult Care
The MARR for the BCS measure increased 10.7 percentage points
HC MARR comparison to NHM: The HealthChoice MARR is above the NHM for all measures except in ten
areas. Differences of less than .5 percentage points are not listed.
CAT measure – the MARR is 3.5 percentage points above the NHM
PBH measure – the MARR is 3.5 percentage points above the NHM
CBP measure– the MARR is 3.5 percentage points below the NHM
BCS measure – the MARR is 6.4 percentage points above the NHM
CWP measure – the MARR is 11.7 percentage points above the NHM
PPC measure (Timeliness) – the MARR is 8.9 percentage points below the NHM
AWC measure – the MARR is 17.6 percentage points above the NHM
W34 measure – the MARR is 12 percentage points above the NHM
W15 (6+ visits) – the MARR is 6.5 percentage points above the NHM
HealthChoice Maryland Average Reportable Rate Highlights
Some changes in performance rates from HEDIS
®
2013 are highlighted below:
With the exception of JMS, all plans with a PAC product, used the administrative method for the
measures that could be done by the hybrid method. Rationale perhaps was that the PAC product
is being discontinued.
The Timeliness of Prenatal Care and Post-Partum Care numbers in the PPC measure decreased
significantly for HEDIS
®
2014. The TPC numerator dropped 11.8 percentage points and the Post-
Partum dropped 8.9 percentage points. The drop was due to RHP calculating the measure using
the administrative method. If you remove RHP from the calculations, the TPC actually increased
almost 1 percentage point and the PP indicator increased 2.2 percentage points.

2014 Annual Technical Report
Delmarva Foundation
88
HealthChoice plans still have difficulty achieving national benchmark performance scores for the
CBP measure. While the MARR did increase 3 percentage points, the MARR is still 3.5
percentage points below the NHM.
Customer Service, as reflected in the CAT measure, is still 3.5 percentage points above the NHM
but there was a slight decrease in the MARR.
The DHMH special performance score for the ASM measure remains problematic for the
HealthChoice plans. The overall performance score for the age group 5-50 in the ASM measure
again decreased 2.5 percentage points.
The HealthChoice plans had a significant increase in the performance score for the Adult BMI
Assessment measure, which increased 11 percentage points. This was due to increased emphasis
on supplemental data and a good medical record hybrid review program.
The specifications for the CCS measure changed this year. The NHM is based on prior
specifications. The MARR did increase 1.5 percentage points and the MARR is 10.7 percentage
points above the NHM.
A new measure this year was Weight Assessment and Counseling for Nutrition and Physical
Activity for Children/Adolescents (WCC). While no trend in the MARR is available, the MARR
is above the NHM in all three indicators.

2014 Annual Technical Report
Delmarva Foundation
89
Section VII
Consumer Assessment of Healthcare Providers and Systems®
Introduction
COMAR 10.09.65.03(C)(4) requires that all HealthChoice MCOs participate in the annual Consumer
Assessment of Healthcare Providers and Systems (CAHPS
®
) Survey. DHMH has contracted with WBA
Market Research (WBA), an NCQA-certified survey vendor, since 2008 to conduct its survey. WBA
administers this survey to a random sample of eligible adult and child members enrolled in HealthChoice via
mixed methodology (mail with telephone follow-up), per NCQA protocol. Seven MCOs participated in the
HealthChoice CAHPS
®
2014 survey based on services provided in CY 2013:
AMERIGROUP Community Care
Jai Medical Systems
Maryland Physicians Care
MedStar Family Choice, Inc.
Priority Partners
Riverside Health of Maryland
UnitedHealthcare
2014 CAHPS
®
5.0H Medicaid Survey Methodology
In 2014, the 5.0H version of the CAHPS
®
Adult and Child Medicaid Satisfaction Surveys was used to survey
the HealthChoice population about services provided in CY 2013. The survey measures those aspects of care
for which members are the best and/or the only source of information. From this survey, members’ ratings
of and experiences with the medical care they receive can be determined. Based on members’ health care
experiences, potential opportunities for improvement can be identified. Specifically, the results obtained from
this consumer survey will allow DHMH to:
Determine how well participating HealthChoice MCOs are meeting their members’ expectations
Provide feedback to the HealthChoice MCOs to improve quality of care
Encourage HealthChoice MCO accountability
Develop a HealthChoice MCO action plan to improve members’ quality of care
Results from the CAHPS
®
5.0H survey summarize member satisfaction with their health care through ratings,
composites, and question summary rates. In general, summary rates represent the percentage of respondents
who chose the most positive response categories as specified by NCQA. Ratings and composite measures in
the CAHPS
®
5.0H Adult and Child Medicaid Survey include:
Overall Ratings of Personal Doctor, Specialist, Health Care, and Health Plan

2014 Annual Technical Report
Delmarva Foundation
90
Getting Needed Care
Getting Care Quickly
How Well Doctors Communicate
Customer Service
Shared Decision-Making
Health Promotion and Education
Coordination of Care
Five additional composite measures are calculated for the Children with Chronic Conditions (CCC)
population:
Access to Prescription Medicine
Family Centered Care: Getting Needed Information
Family Centered Care: Personal Doctor Who Knows Child
Access to Specialized Services
Coordination of Care for CCC
Research Approach
Eligible adult and child members from each of the seven HealthChoice MCOs that provide Medicaid services
participated in this research. WBA administered a mixed methodology including mailing the CAHPS
®
survey
along with a telephonic survey follow-up. Two questionnaire packages and follow-up reminder postcards
were sent to random samples of eligible adult and child members from each of the seven HealthChoice
MCOs with “Return Service Requested” with WBA’s toll-free number included. The mailed materials also
included a toll-free number for Spanish-speaking members to complete the survey over the telephone. Those
who did not respond by mail were contacted by phone to complete the survey. During the telephone follow-
up, members had the option to complete the survey in either English or Spanish. The child surveys were
conducted by proxy, that is, with the parent/guardian who knows the most about the sampled child’s health
care.
Sampling Methodology
The NCQA required sample size is 1,350 for each of the adult Medicaid plans. In addition to the required
sample size, NCQA allows oversampling of up to 30%. DHMH elected to use this option. To qualify, adult
Medicaid members had to be 18 years of age or older, as well as continuously enrolled in the HealthChoice
MCO for five of the last six months as of the last day of the measurement year (December 31, 2013).
Following this sampling methodology, WBA mailed 1,755 surveys for each HealthChoice MCO, except for
RHMD as the MCO had fewer enrolled and eligible members than the required General Population sample
(1,350). Therefore, a total of 11,421 surveys were mailed for CAHPS
®
2014.

2014 Annual Technical Report
Delmarva Foundation
91
A total of 3,600 valid surveys were completed between February and May 2014 for the adult HealthChoice
population, 28 of which were completed in Spanish. Specifically, 2,145 were returned by mail and 1,455 were
conducted over the phone. The overall response rate from the eligible Medicaid adult population for
CAHPS
®
2014 was 32%.
The NCQA required sample size is 1,650 for child Medicaid plans (General Population/Sample A). In
addition to the required sample size, NCQA allows over-sampling up to 30%. DHMH elected to use this
option. To qualify, child Medicaid members had to be 17 years of age or younger. Furthermore, members had
to be continuously enrolled in the HealthChoice MCO for five of the last six months as of the last day of the
measurement year (December 31, 2013).
Among the child population, an additional over-sample of up to 1,840 child members with diagnoses
indicative of a probable chronic condition was also pulled (CCC Over-sample/Sample B). This is standard
procedure when the CAHPS
®
5.0H Child Medicaid Survey (with CCC Measurement Set) is administered, to
ensure the validity of the information collected.
The CCC population is identified based on child members’ responses to the CCC survey-based screening tool
(questions 60 to 73), which contains five questions representing five different health consequences; four are
three-part questions and one is a two-part question. A child member is identified as having a chronic
condition if all parts of the question for at least one of the specific health consequences are answered “Yes”.
It’s important to note that the General Population data set (Sample A) and CCC Over-sample data set
(Sample B) are not mutually exclusive groups. For example, if a child member is randomly selected for the
CAHPS
®
Child Survey sample (General Population/Sample A) and is identified as having a chronic condition
based on responses to the CCC survey-based screening tool, the member is included in both General and
CCC Population results.
Between February and May 2014, WBA collected 4,489 valid surveys, 251 of which were completed in
Spanish. Specifically, 2,727 were completed by mail and 1,762 were completed by phone. The overall
response rate from the eligible Medicaid child population was 34%. Of the responses, 1,971 child members
across all HealthChoice MCOs qualified as being children with chronic conditions based on the
parent’s/guardian’s responses to the CCC survey-based screening tool.
Ineligible adult and child members included those who were deceased, did not meet eligible population
criteria (indicated non-membership in the specified health plan), or had a language barrier (non-English or
Spanish). In addition, adult members who were mentally or physically incapacitated and unable to complete
the survey themselves were also considered ineligible. Non-respondents included those who had refused to
participate, could not be reached due to a bad address or telephone number, or were unable to be contacted

2014 Annual Technical Report
Delmarva Foundation
92
during the survey time period. Ineligible surveys are subtracted from the sample size when computing a
response rate.
Table 33 shows the total number of adult members in the sample that fell into each disposition category.
Table 33. Adult Dispositions
Disposition Group
Disposition Category
Number
Ineligible
Deceased
11
Does not meet eligibility criteria
122
Language barrier
42
Mentally/Physically incapacitated
40
Total Ineligible
215
Non-Response
Bad address/phone
944
Refusal
379
Maximum attempts made
6,283
Total Non-Response
7,606
Table 34 show the number of mail and phone completes as well as the response rate for each Health Choice
MCO.
Table 34. MCO Response Rate
HealthChoice MCO
Mail and Phone Completes*
Response Rate
AMERIGROUP Community Care
519
30%
Diamond Plan
588
34%
Jai Medical Systems
587
34%
Maryland Physicians Care
565
33%
MedStar Family Choice, Inc.
596
35%
Priority Partners
190
22%
UnitedHealthcare
555
32%
Total HealthChoice MCOs
3,600
32%
Findings
Key Findings from the 2014 CAHPS
®
5.0H Adult Medicaid Survey
There were four Overall Rating questions asked in the CAHPS
®
5.0H Adult Medicaid Survey that used a scale
of “0 to 10”, where a “0” represented the worst possible rating and a “10” represented the best possible
rating. Table 36 shows each of the four Overall Rating questions and the Summary Rate for these questions
from CAHPS
®
2013 and CAHPS
®
2014. The summary rate represents the percentage of members who rated
the question an 8, 9, or 10.

2014 Annual Technical Report
Delmarva Foundation
93
Table 35. CAHPS
®
2013 and CAHPS
®
2014 Adult Summary Rates for Overall Rating Questions
Overall Ratings
CAHPS 2013
(Summary Rate – 8,9,10)
CAHPS 2014
(Summary Rate – 8,9,10)
Health Care
69%
70%
Personal Doctor
76%
77%
Specialist Seen Most Often
77%↑
77%
Health Plan
69%
72%↑
Arrows (↓,↑) indicate that the particular measure is performing statistically better or worse than it did in the previous year.
Consistent with CAHPS
®
2013, HealthChoice adult members give their highest satisfaction ratings (a rating
of 8, 9, or 10) to their Specialist (77%) and/or their Personal Doctor (77%) in CAHPS
®
2014. HealthChoice
members continued to give slightly lower satisfaction ratings to their Health Plan (72%) and Health Care
(70%) overall.
Overall Ratings
In order to assess how the HealthChoice MCOs overall ratings compared with other Medicaid adult and child
plans nationwide, national benchmarks are provided. Specifically, the adult and child data are compared to the
Quality Compass
®
benchmarks (Reporting Year 2013). Quality Compass
®
is a national database created by
the NCQA to provide health plans with comparative information on the quality of the nation’s managed care
plans.
Table 36 shows a plan comparison of Adult Summary Ratings of the four Overall Rating questions for the
seven participating HealthChoice MCOs. Additionally, it indicates the Quality Compass
®
and the
HealthChoice Aggregate for each question.
Table 36. CAHPS
®
2014 MCO Adult Summary Rates for Overall Rating Questions
CAHPS 2014 Adult Overall Ratings
(Summary Rate – 8,9,10)
Health Care
Personal Doctor
Specialist Seen Most Often
Health Plan
Quality Compass
®1
71%
79%
80%
75%
HealthChoice Aggregate
70%
77%
77%
72%
AMERIGROUP Community Care
72%
74%
76%
71%
Jai Medical Systems
61%
78%
71%
64%
Maryland Physicians Care
70%
73%
79%
73%
MedStar Family Choice, Inc.
73%
79%
79%
76%*
Priority Partners
69%
78%
78%
76%*
Riverside Health of Maryland
2
74%*
77%
82%*
74%
UnitedHealthcare
74%*
81%*
78%
73%
*MCO with the highest Summary Rate.
1
Quality Compass® is a registered trademark of NCQA.
2
First-year HealthChoice MCO.
Composite measures assess results for main issues/areas of concern. The following composite measures were
derived by combining survey results of similar CAHPS
®
questions:

2014 Annual Technical Report
Delmarva Foundation
94
How Well Doctors Communicate – Measures how well personal doctor explains things, listens to
them, shows respect for what they have to say and spends enough time with them.
Customer Service – Measures members’ experiences with getting the information needed and treatment
by Customer Service staff.
Getting Care Quickly – Measures members’ experiences with receiving care and getting appointments
as soon as they needed.
Getting Needed Care – Measures members’ experiences in the last six months when trying to get care
from specialists and through health plan.
Coordination of Care – Measures members’ perception of whether their doctor is up-to-date about the
care he/she received from other doctors or health providers.
Health Promotion and Education – Measures members’ experience with their doctor discussing
specific things to do to prevent illness.
Shared Decision Making – Measures members’ experiences with doctors discussing the pros and cons
of starting or stopping a prescription medicine and asking the member what they thought was best for
them.
Table 37 shows the adult composite measure results from CAHPS
®
2013 and CAHPS
®
2014.
Table 37. CAHPS
®
2013 and CAHPS
®
2014 Adult Composite Measure Results
Composite Measure
CAHPS 2013
(Yes or A lot/
Some/Yes)
CAHPS 2014
(Yes or A lot/
Some/Yes)
How Well Doctors Communicate
89%
89%
Customer Service
81%
85%↑
Getting Care Quickly
80%
79%
Getting Needed Care
79%↑
80%
Coordination of Care
78%
79%
Health Promotion and Education
75%
74%
Shared Decision-Making
54%
52%
Arrows (↓,↑) indicate that the particular measure is performing statistically better or worse than it did in the previous year.
Consistent with CAHPS
®
2013, HealthChoice MCOs continued to receive the highest ratings among their
members on the “How Well Doctors Communicate” composite in CAHPS
®
2014 (89%).
Notably, the “Customer Service” composite score increased in 2014 (85%), up from 81% in 2013.
Research shows that HealthChoice MCOs receive the lowest ratings among their members on the following
composite measures:
Health Promotion and Education (74% Summary Rate – Yes); and
Shared Decision-Making (52% Summary Rate – A lot or Yes).

2014 Annual Technical Report
Delmarva Foundation
95
Key Findings from the 2014 CAHPS
®
5.0H Child Medicaid Survey
The results from the four Overall Ratings questions asked in the CAHPS
®
5.0H Child Medicaid Survey that
are represented in Table 38. The summary rate represents the percentage of members who rated the question
an 8, 9, or 10.
Table 38. CAHPS
®
2013 and CAHPS
®
2014 Child Summary Rates for Overall Rating Questions
Overall Ratings
CAHPS 2013
(Summary Rate – 8,9,10)
CAHPS 2014
(Summary Rate – 8,9,10)
General
CCC
General
CCC
Health Care
85%
82%
86%
83%
Personal Doctor
87%
86%
89%
87%
Specialist Seen Most Often
82%
82%
80%
82%
Health Plan
83%
80%
85%
83%
HealthChoice MCOs continued to receive high satisfaction ratings from both parents/guardians of the
general children’s population group and the parents/guardians of the children with chronic conditions
population group for each overall rating question.
Table 39 shows a plan comparison of Child Summary Ratings of the four Overall Rating questions for the
seven participating HealthChoice MCOs. Additionally, it indicates the Quality Compass
®
and HealthChoice
Aggregate for each question.
Table 39. CAHPS
®
2014 MCO Child Summary Rates for Overall Rating Questions
2014 Adult Overall Ratings
(Summary Rate – 8,9,10)
Health Care
Personal Doctor
Specialist Seen Most
Often
Health Plan
General
CCC
General
CCC
General
CCC
General
CCC
Quality Compass
®1
85%
83%
88%
87%
85%
85%
84%
81%
HC Aggregate
86%
83%
89%
87%
80%
82%
85%
83%
ACC
85%
82%
88%
87%
77%
76%
88%*
83%
JMS
87%
84%*
90%*
90%*
74%
68%
83%
80%
MPC
86%
84%*
88%
84%
75%
78%
84%
82%
MSFC
86%
83%
89%
86%
83%
83%
85%
83%
PPMCO
86%
83%
88%
89%
87%*
86%*
87%
84%*
RHMD
76%
76%
85%
88%
65%
80%*
77%
67%
UHC
89%*
84%*
89%
87%
84%
86%
85%
82%
*MCO with the highest Summary Rate.
1
Quality Compass® is a registered trademark of NCQA.
2
First-year HealthChoice MCO.
In CAHPS
®
2014, HealthChoice MCOs continue to receive the highest ratings among both the general child
population members and the child members with chronic conditions on the following composite measures:
How Well Doctors Communicate – Measures how well personal doctor explains things, listens to
them, shows respect for what they have to say and spends enough time with them.
Getting Care Quickly – Measures member’s experiences with receiving care and getting appointments
as soon as they needed.

2014 Annual Technical Report
Delmarva Foundation
96
In addition, HealthChoice MCOs also received high ratings among the general population members for the
following composite measure:
Customer Service – Measures member’s experiences with getting the information needed and treatment
by Customer Service staff.
Table 40 shows the child composite measure results from CAHPS
®
2013 and CAHPS
®
2014.
Table 40. CAHPS
®
2013 and CAHPS
®
2014 Child Composite Measure Results
Composite Measure
CAHPS 2013
(Summary Rate–
Always/Usually)
CAHPS 2014
(Yes or A lot/
Some/Yes)
General
CCC
General
CCC
How Well Doctors Communicate
94%
93%
94%
94%
Getting Care Quickly
91%↑
93%↑
90%
92%
Customer Service
87%↑
87%↑
87%
86%
Getting Needed Care
82%
84%↑
84%
85%
Coordination of Care
80%
79%
82%
81%
Shared Decision-Making
1
55%
61%
57%
62%
Health Promotion and Education
2
73%
78%
75%
80%
Arrows (↓,↑) indicate that the particular measure is performing statistically better or worse than it did in the previous year.
1
Shared Decision-Making composite revised in 2013. Added one question and significantly altered the existing questions and response choices. Trending impacted.
2
Health Promotion and Education composite revised in 2013. Question wording and response choices altered. Trending impacted.
Research shows that for both the general population and child members with chronic conditions,
HealthChoice MCOs received the lowest ratings on the “Health Promotion and Education” (75%) and
“Shared Decision-Making” composites (57%). HealthChoice MCOs also received a lower rating among the
CCC population for the “Coordination of Care” composite measure (81%). Of note, the “Getting Needed
Care” composite measure received higher ratings in CAHPS
®
2014 (84%, up from 82% the previous year).
Key Drivers of Satisfaction
In an effort to identify the underlying components of adult and child members’ ratings of their Health Plan
and Health Care, advanced statistical techniques were employed.
Regression analysis is a statistical technique used to determine which influences or “independent
variables” (composite measures) have the greatest impact on an overall attribute or “dependent variable”
(overall rating of Health Plan or Health Care).
In addition, correlation analyses were conducted between each composite measure attribute and overall
rating of Health Plan and Health Care in order to ascertain which attributes have the greatest impact.
Adult Medicaid Members – Key Drivers of Satisfaction with Health Plan
Based on the CAHPS
®
2014 findings, the “Customer Service” and “Getting Needed Care” composite
measures have the most significant impact on adult members’ overall rating of their Health Plan.
The attribute listed below is identified as an unmet need and should be considered a priority area for the
HealthChoice MCOs. If performance on this attribute is improved, it could have a positive impact on
adult members’ overall rating of their Health Plan.

2014 Annual Technical Report
Delmarva Foundation
97
Received information or help needed from health plan’s Customer Service
The following attributes are identified as driving strengths and performance in these areas should be
maintained. If performance on these attributes is decreased, it could have a negative impact on adult
members’ overall rating of their Health Plan.
Treated with courtesy and respect by health plan’s Customer Service
Doctor showed respect for what you had to say
Adult Medicaid Members – Key Drivers of Satisfaction with Health Care
Based on the CAHPS
®
2014 findings, the “Getting Needed Care” composite measure has the most significant
impact on adult members’ overall rating of their Health Care.
There were no attributes identified as unmet needs that should be considered priority areas for improving
adult members’ overall rating of their Health Care.
However, there are four attributes that are identified as key drivers that are of high importance to
members where they perceive HealthChoice MCOs to be performing moderately well. Improvement in
these areas could have a positive impact on members’ overall rating of their Health Care: “Got the care,
tests or treatment you needed”, “Doctor spent enough time with you”, “Doctor listened carefully to you”
and “Doctor explained things in way that was easy to understand”.
The attribute “Doctor showed respect for what you had to say” is identified as a driving strength and
performance in this area should be maintained. If performance on this attribute is decreased, it could
have a negative impact on adult members’ overall rating of their Health Care.
Child Medicaid Members – Key Drivers of Satisfaction with Health Plan
Based on the CAHPS
®
2014 findings, the “How Well Doctors Communicate” composite measure has the
most significant impact on child members’ overall rating of their Health Plan.
There were no attributes identified as unmet needs that should be considered priority areas for improving
child members’ overall rating of their Health Plan.
However, the attribute “Received information or help needed from child’s health plan’s Customer
Service” is an area that is of high importance to child members where HealthChoice MCOs perform
at a moderate level. Improvement in this area could have a positive impact on child members’ overall
rating of their Health Plan.
The attributes listed below are identified as driving strengths and performance in these areas should be
maintained. If performance on these attributes is decreased, it could have a negative impact on child
members’ overall rating of their Health Plan.
Treated with courtesy and respect by child’s health plan’s Customer Service
Got the care, tests or treatment your child needed

2014 Annual Technical Report
Delmarva Foundation
98
Child Medicaid Members – Key Drivers of Satisfaction with Health Care
Based on the CAHPS
®
2014 findings, the “Getting Needed Care” and “How Well Doctors Communicate”
composite measures are identified as having the most significant impact on child members’ overall rating of
their Health Care.
Given some of the high ratings received, there were no attributes identified as unmet needs that should
be considered priority areas for improving child members’ overall rating of their Health Care.
However, the attribute “Received an appointment for a check-up or routine care for your child as
soon as they needed” is an area that is of high importance to child members where HealthChoice
MCOs perform at a moderate level. Improvement in this area could have a positive impact on child
members’ overall rating of their Health Care.
Instead, the attributes listed below are identified as driving strengths and performance in these areas
should be maintained. If performance on these attributes is decreased, it could have a negative impact on
child members’ overall rating of their Health Care.
Got the care, tests or treatment your child needed
Child’s doctor listened carefully to you
Child’s doctor explained things about your child’s health in a way that was easy to understand
Child’s doctor showed respect for what you had to say
Child’s doctor spent enough time with your child

2014 Annual Technical Report
Delmarva Foundation
99
Section VIII
Consumer Report Card
Introduction
DHMH contracted with Delmarva Foundation to develop a Medicaid Consumer Report Card (Report Card).
Delmarva Foundation collaborated with the NCQA to assist in the Report Card development and
production.
The Report Card assists Medicaid beneficiaries in selecting one of the participating HealthChoice MCOs.
Information in the Report Card includes performance measures from HEDIS
, the CAHPS
survey, and
DHMH’s VBPI.
Information Report Strategy
The reporting strategy incorporates methods and recommendations based on experience and research about
presenting quality information to consumers. The most formidable challenge facing all consumer information
projects is how to communicate a large amount of complex information in an understandable and meaningful
manner while fairly and accurately representing the data.
To enhance comprehension and interpretation of quality measurement information provided for a Medicaid
audience, the NCQA and Delmarva Foundation team designed the Report Card to include six categories,
with one level of summary scores (measure roll-ups), per plan, for each reporting category. Research has
shown that people have difficulty comparing plan performance when information is presented in too many
topic areas. To include a comprehensive set of performance measures in an effective consumer information
product (one that does not present more information than is appropriate for the audience), measures must be
combined into a limited number of reporting categories that are meaningful to the target audience, Medicaid
participants.
Based on a review of the measures available for the Report Card (HEDIS
®
, CAHPS
®
and DHMH’s VBPI),
the team recommended the following reporting categories and their descriptions:
Access to Care
Appointments are scheduled without a long wait
The MCO has good customer service
Everyone sees a doctor at least once a year
HEDIS
®
is a registered trademark of the National Committee for Quality Assurance (NCQA).
CAHPS
®
is a registered trademark of the Agency for Healthcare Quality and Research (AHRQ).

2014 Annual Technical Report
Delmarva Foundation
100
Doctor Communication and Service
Doctors explain things clearly and answer questions
The doctor’s office staff is helpful
Doctors provide good care
Keeping Kids Healthy
Kids get shots to protect them from serious illness
Kids see a doctor and dentist regularly
Kids get tested for lead
Care for Kids With Chronic Illness
Doctors give personal attention
Kids get the medicine they need
A doctor or nurse knows the child’s needs
Doctors involve parents in decision making
Taking Care of Women
Women are tested for breast cancer and cervical cancer
Moms are taken care of when they are pregnant and after they have their baby
Care for Adults with Chronic Illness
Blood sugar levels are monitored and controlled
Cholesterol levels are tested and controlled
Eyes are examined for loss of vision
Kidneys are healthy and working properly
Appropriate use of antibiotics
Appropriate treatment for lower back pain
The first two categories are relevant to all beneficiaries. The remaining categories are focused on more
specific populations that are relevant to Maryland HealthChoice beneficiaries: children, children with chronic
illness, women, and adults with chronic illness.
In accordance with its research, NCQA did not recommend reporting specific measures individually, in
addition to the above reporting categories. Consumers comparing the performance of a category composed
of many measures to individual measures may give undue weight to the performance on the individual
measures.
Measure Selection
The measures that the project team considered for inclusion in the Report Card are derived from those that
DHMH requires MCOs to report, which include HEDIS
®
measures, the CAHPS
®
survey results from both
the Adult Questionnaire and the Child Questionnaire, and DHMH’s VBP measures.

2014 Annual Technical Report
Delmarva Foundation
101
NCQA created measure selection criteria that included a consistent and logical framework for determining
which quality of care measures are to be included in each composite each year.
Recent revisions to the CAHPS
®
survey and re-evaluations of HEDIS
®
measures influence NCQA’s
recommendations for the 2014 reporting strategy.
Reporting Category Changes:
Access to Care
Call Answer Timeliness measure will be added to this reporting category
CAHPS
®
Survey questions updated to 5.0H
Doctor Communication and Service
CAHPS
®
Survey questions updated to 5.0H
Keeping Kids Healthy
No changes
Care for Kids with Chronic Illness
CAHPS
®
Survey questions updated to 5.0H
Taking Care of Women
No changes
Care for Adults with Chronic Illness (formerly Diabetes Care)
Rename reporting category from ‘Diabetes Care’ to ‘Care for Adults with Chronic Illness’ to include
additional measures
Use of Imaging Studies for Low Back Pain and Avoidance of Antibiotic Treatment in Adults With Acute
Bronchitis measures will be added to this reporting category
Format
The following principles are important when designing report cards:
Space: Maximize the amount to display data and explanatory text
Message: Communicate MCO quality in positive terms to build trust in the information presented
Instructions: Be concrete about how consumers should use the information
Text: Relate the utility of the Report Card to the audience’s situation (e.g., new beneficiaries choosing a
plan for the first time, beneficiaries receiving the Annual Right to Change Notice and prioritizing their
current health care needs, current beneficiaries learning more about their plan) and reading level

2014 Annual Technical Report
Delmarva Foundation
102
Narrative: Emphasize why what is being measured in each reporting category is important, rather than
giving a detailed explanation of what is being measured. For example, “making sure that kids get all of
their shots protects them against serious childhood diseases” instead of “the percentage of children who
received the following antigens …”
Design: Use color and layouts to facilitate navigation and align the star ratings to be consistent with the
key.
The Report Card was printed as a 24 x 9.75 inch pamphlet folded in thirds, with English on one side and
Spanish on the opposite side. Pamphlets allow one-page presentation of all performance information.
Additionally, measure explanations can be integrated on the same page as the performance results, facilitating
a reader’s ability to match the explanation to actual data.
Pamphlet contents were drafted to present the information at a sixth-grade reading level, with short, direct
sentences intended to relate to the audience’s particular concerns. Terms and concepts unfamiliar to the
general public were avoided. Explanations of performance ratings, measure descriptions, and how to use the
Report Card were straightforward and action-oriented. Contents were translated into Spanish by an
experienced translation vendor.
Cognitive testing conducted for similar projects showed that Medicaid beneficiaries had difficulty associating
the data in charts with explanations if they were presented elsewhere in the Report Card. Consumers prefer a
format that groups related data on a single page. Given the number of MCOs whose information is being
presented in Maryland’s HealthChoice Report Card, a pamphlet format allows easy access to information.
Rating Scale
Performance is rated by comparing each MCO’s performance to the average of all MCOs potentially available
to the target audience; in this case, the average of all HealthChoice MCOs (a.k.a., the Maryland HealthChoice
MCO average). Stars are used to represent performance that is “above,” “the same as,” or “below” the
Maryland HealthChoice MCO average.
A tri-level rating scale in a matrix that displays performance across a select number of salient performance
categories provides beneficiaries with an easy-to-read “picture” of quality performance across plans and
presents data in a manner that emphasizes meaningful differences between plans that are available to them.
(The tri-level rating method is explained in Section III, Analytic Methods.) This methodology differs from
similar methodologies that compare plan performance to ideal targets or national percentiles. The team’s
recommended approach is more useful in an environment where consumers must choose from a group of
available plans.

2014 Annual Technical Report
Delmarva Foundation
103
At this time, the team does not recommend developing an overall rating for each MCO. The proposed
strategy allows the Report Card users to decide which performance areas are most important to them when
selecting a plan.
Analytic Methodology
NCQA and Delmarva Foundation recommend that the Report Card compare each MCO’s actual score to the
unweighted, statewide plan average for a particular reporting category. An icon or symbol would denote
whether a plan performed “above,” “the same as,” or “below” the statewide Medicaid plan average.
2
The goal of the analysis is to generate reliable and useful information that can be used by Medicaid consumers
to make relative comparisons of the quality of health care provided by Maryland’s HealthChoice MCOs. This
information should allow consumers to easily detect substantial differences in MCO performance. This
means that the index of difference should compare plan-to-plan quality performance directly and that
differences between MCOs should be statistically reliable.
Handling Missing Values
Three issues involve the replacement of missing values in this analysis. The first issue is deciding which pool
of observed (non-missing) plans should be used to derive replacement values for missing data.
The second issue concerns how imputed values will be chosen. Alternatives are fixed values (such as zero or
the 25th percentile for all plans in the nation), calculated values (such as means or regression estimates) or
probable selected values (such as multiplying imputed values).
The third issue is that the method used to replace missing values should not provide an incentive for plans
that perform poorly to purposefully fail to report data. For example, if missing values are replaced with the
mean of non-missing cases, scores for plans that perform below the mean would be increased if they fail to
report.
Replacing missing Medicaid plan data with commercial plan data is inappropriate because the characteristics
of Medicaid populations differ from those of commercial populations. This restricts the potential group to
2
For state performance reports directed at consumers, NCQA believes it is most appropriate to compare a plan’s
performance to the average of all plans serving the state. NCQA does not recommend comparing plans to a statewide
average that has been weighted proportionally to the enrollment size of each plan. A weighted average emphasizes
plans with higher enrollments and is used to measure the overall, statewide average. Report cards compare a plan’s
performance relative to other plans, rather than presenting how well the state’s Medicaid managed care plans serve
beneficiaries overall. In a Report Card, each plan represents an equally valid option to the reader, regardless of its
enrollment size.

2014 Annual Technical Report
Delmarva Foundation
104
national Medicaid plans, regional Medicaid plans, or Maryland HealthChoice plans. Analyses conducted by
NCQA for the annual State of Health Care Quality report have consistently shown substantial regional
differences in the performance of commercial managed care plans. Assuming that such regional differences
generalize to Medicaid plans, it would be inappropriate to use the entire group of national Medicaid plans to
replace missing values for Maryland HealthChoice plans.
Using a regional group of plans to derive missing values was also determined to be inappropriate because of
substantial differences in Medicaid program administration across states. In other words, reporting of
Medicaid data is skewed to a few large states with large Medicaid managed care enrollment.
For these reasons, Maryland HealthChoice plans should serve as the pool from which replacement values for
missing data are generated. A disadvantage to using only Maryland HealthChoice plans for missing data
replacement is that there are fewer than 20 plans available to derive replacement values. This makes it unlikely
that data-intensive imputation procedures such as regression or multiple imputations can be employed.
Plans are sometimes unable to provide suitable data (for example, if too few of their members meet the
eligibility criteria for a measure), despite their willingness to do so. These missing data are classified as “not
applicable” (N/A). If the NCQA HEDIS
®
Compliance Audit finds the measure to be materially biased, the
measure is assigned a “Not Reportable” designation (NR).
For Report Card purposes, missing values will be replaced where a plan has reported data for at least 50
percent of the indicators in a reporting category. A plan that is missing more than 50 percent of the indicators
that compose a reporting category will be given a designation of “insufficient data” for that measurement
category. If fewer than 50 percent of the plans report a measure, the measure is dropped from the report card
category. Therefore, the calculations in that category are based upon the remaining reportable measures.
“N/A” and “NR” designations will be treated differently where values are missing. “N/A” values will be
replaced with the mean of “non-missing observations” and “NR” values will be replaced with the minimum
value of the “non-missing observations.” This procedure minimizes any disadvantage to plans that are willing
but unable to report data.
Case-Mix Adjustment of CAHPS
®
Data
Several field tests indicate that there is a tendency for CAHPS
®
survey respondents who are in poor health to
have lower satisfaction scores. It is not clear whether this is because members in poor health experience lower
quality health care or because they are generally predisposed to give more negative responses (halo effect).
It is believed that respondents in poor health receive more intensive health care services, and their CAHPS
®
’
survey responses do contain meaningful information about the quality of care delivered in this more intensive
environment. Therefore, case-mix adjusting is not planned for the CAHPS
®
survey data used in this analysis.

2014 Annual Technical Report
Delmarva Foundation
105
Statistical Methodology
The statistical methodology includes the following steps:
1. Create standardized versions of all measures for each plan so that all component measures that
contribute to the summary scores for each reporting category are on the same scale. Measures are
standardized by subtracting the mean of all plans from the value for individual plans and dividing by the
standard deviation of all plans.
2. Combine the standard measures into summary scores in each reporting category for each plan.
3. Calculate standard errors for individual plan summary scores and for the mean summary scores for all
plans.
4. Calculate difference scores for each reporting category by subtracting the mean summary score for all
plans from individual plan summary score values.
5. Use the standard errors to calculate 95 percent confidence intervals for the difference scores.
6. Categorize plans into three categories on the basis of these confidence intervals (CI). If the entire 95
percent CI is in the positive range, the plan is categorized as “above average.” If a plan’s 95 percent CI
includes zero, the plan is categorized as “average.” If the entire 95 percent CI is in the negative range, the
individual plan is categorized as “below average.”
This procedure generates classification categories so differences from the group mean for individual plans in
the “above average” and “below average” categories are statistically significant at α = .05. Scores of plans in
the “average” category are not significantly different from the group mean.
CY 2014 Report Card Results
HealthChoice
MCOs
Performance Area
Access to
Care
Doctor
Communication
and Service
Keeping
Kids
Healthy
Care for
Kids with
Chronic
Illness
Taking
Care of
Women
Care for
Adults with
Chronic
Illness
ACC
JMS
Not Rated by
Researchers*
MPC
MSFC
PPMCO
RHMD
N/A
N/A
N/A
N/A
N/A
N/A
UHC
Below HealthChoice Average
HealthChoice Average
Above HealthChoice Average
N/A - RHMD became a HealthChoice MCO in 2013 and ratings are not applicable.
*“Not Rated By Researchers” does not describe the performance or quality of care provided by the health plan.

2014 Annual Technical Report
Delmarva Foundation
106
Section IX
Review of Compliance with Quality Strategy
Table 41 below describes HACA’s progress against the Quality Strategy’s goal.
Table 41. Quality Strategy Evaluation
Department’s
Quality Strategy Goal
Performance Against Goal
Met
Ensure compliance with
changes in Federal/State law
and regulation
The Department consistently reviews all new Federal and State laws and
regulations. Any new laws and regulations are immediately put into the
standards and guidelines for review and communicated to the MCOs.
√
Improve performance over time
The Department continually strives to improve performance, which is evident
through the high standards it sets for the MCOs in the Annual Systems
Performance Review, Value Based Purchasing Initiative, Performance
Improvement Projects, and other review activities. It continually monitors the
progress of MCO performance in multiple areas as demonstrated throughout
this report.
√
Allow comparisons to national
and state benchmarks
In almost every area of review, comparisons to national and state
benchmarks can be found to mark progress and delineate performance
against goals.
√
Reduce unnecessary
administrative burden on MCOs
The Department has attempted to reduce unnecessary administrative burden
to the MCOs in any way possible. Delmarva Foundation has assisted with this
goal in streamlining the Annual Systems Review Process so that
documentation can be submitted electronically.
√
Assist the Department with
setting priorities and
responding to identified areas
of concern such as children,
pregnant women, children with
special healthcare needs,
adults with a disability, and
adults with chronic conditions.
The HealthChoice and Acute Care Administration has assisted the
Department by:
Selecting performance measures to monitor compliance with quality of
care and access standards for participants.
Selecting the initial Adult and Child CORE health care quality measures
for Medicaid and CHIP. Maryland Volunteered to collect Medicaid Adult
and Child CORE Measures which will assist CMS to better understand
the quality of health care that adults enrolled in Medicaid receive.
Designing supplemental CAHPS
®
survey questions to address pregnant
women and children to provide data input for the Deputy Secretary of
Health Care Financing –Medical Care Programs Administration’s annual
Managing for Results report that includes key goals, objectives, and
performance measures’ results for calendar year.
√
√ - Goal Met

2014 Annual Technical Report
Delmarva Foundation
107
EQRO Recommendations for MCOs
Each MCO is committed to delivering high quality care and services to its participants. However,
opportunities exist for continued performance improvement. Based upon the evaluation of CY 2013
activities, Delmarva Foundation has developed several recommendations for all MCOs which are identified
within each section of the Annual Technical Report.
EQRO Recommendations for HACA
Considering the results for measures of quality, access, and timeliness of care for the contracted MCOs,
Delmarva Foundation developed the following recommendations for HACA:
Considering Health Care Reform activities began in 2014 and Maryland Medicaid enrollment increased
significantly, the Department should consider revising the layout of the MD Consumer Report Card. The
Information Reporting Strategy may continue to be relevant, but the format of the report card may need
to be revised, including different information displayed in a different manner. This update would include
funding for consumer focus groups to test the understanding/ease of language and layout.
Maryland MCOs are now required by DHMH to be NCQA accredited, and all but the new MCOs have
obtained their full accreditation. The Department should look at alternative ways to review the MCOs for
quality, access, and timeliness of care. Many of the MCOs have achieved the maximum compliance
threshold of 100% in all standards of the systems performance review. The Department may want to
concentrate their quality efforts in other areas such as focused quality studies or collaborative
performance improvement projects to reduce the burden of the annual reviews on the MCOs.
Conclusion
This report is a representation of all EQRO, HEDIS
®
, and CAHPS
®
activities that took place in calendar
years 2013-2014 for the Maryland HealthChoice program. Opportunities for improvement and best practices
of the MCOs are noted in the executive summary and within each individual review activity.
The Department sets high standards for MCO QA systems. As a result, the HealthChoice MCOs have quality
systems and procedures in place to promote high quality care with well-organized approaches to quality
improvement. The CY 2014 review activities provided evidence of the MCOs’ continuing progression and
demonstration of their abilities to ensure the delivery of quality health care for Maryland managed care
participants.

CY 2014 Annual Technical Report
Appendix A1
Acronym List
Delmarva Foundation
A1-1
ACC
AMERIGROUP Community Care
ADA
Americans with Disabilities Act of 1990
AHRQ
Agency for Healthcare Research and Quality
ASAM
American Society of Addictions Medicine
AVP
Associate Vice President
AWC
Adolescent Well Care
BBA
Balanced Budget Act of 1997
BH
Behavioral Health
BMI
Body Mass Index
CAHPS
Consumer Assessment of Healthcare Providers and Systems
CAP
Corrective Action Plan
CBP
Controlling High Blood Pressure
CC
Credentialing Committee
CCC
Children with Chronic Conditions
CDS
Controlled Dangerous Substance
CEO
Chief Executive Officer
CFR
Code of Federal Regulations
CI
Confidence Interval
CMS
Centers for Medicare and Medicaid Services
COMAR
Code of Maryland Regulations
CPT
Current Procedural Terminology
CRISP
Chesapeake Regional Information System for our Patients
CY
Calendar Year
DHMH
Department of Health and Mental Hygiene
DHQA
Division of HealthChoice Quality Assurance
DIA
Diamond Plan from Coventry Health Care, Inc.
DM
Disease Management
DOB
Date of Birth
EDV
Encounter Data Validation
EPSDT
Early and Periodic Screening, Diagnosis, and Treatment
EQR
External Quality Review
EQRO
External Quality Review Organization
ER
Emergency Room
FC
Fully Compliant
HACA
HealthChoice and Acute Care Administration
HD
HEDIS
®
Measure Determination
HDC
HealthcareData Company, LLC

CY 2014 Annual Technical Report
Appendix A1
Acronym List
Delmarva Foundation
A1-2
HED
Health Education/Anticipatory Guidance
HEDIS
Healthcare Effectiveness Data and Information Set
HIV
Human Immunodeficiency Virus
HRA
Health Risk Assessment
HX
Health and Developmental History
IDSS
Interactive Data Submission System
IMM
Immunizations
IS
Information Systems
JHHC
Johns Hopkins Health Care
JMS
Jai Medical Systems
LAB
Laboratory Tests/At-Risk Screenings
MARR
Maryland Average Reportable Rate
MCO
Managed Care Organization
MD
Maryland
MPC
Maryland Physicians Care
MRR
Medical Record Review
MRRV
Medical Record Review Validation
MSFC
MedStar Family Choice, Inc.
NA
Not Applicable
NCQA
National Committee for Quality Assurance
NHM
National HEDIS
®
Mean
NV
Not Valid
OB/GYN
Obstetrician/Gynecology
PAC
Primary Adult Care
PCP
Primary Care Physician
PE
Comprehensive Physical Exam
PIP
Performance Improvement Project
PPMCO
Priority Partners
QA
Quality Assurance
QAP
Quality Assurance Program
QIC
Quality Improvement Committee
QIO
Quality Improvement Organization
QIWG
Quality Improvement Work Group
QOC
Quality of Care
RHMD
Riverside Health of Maryland
SA
Substance Abuse
SC
Substantially Compliant

CY 2014 Annual Technical Report
Appendix A1
Acronym List
Delmarva Foundation
A1-3
SPR
Systems Performance Review
SSI
Supplemental Security Income
STI/HIV
Sexually Transmitted Infection/Human Immunodeficiency Virus
TAT
Turn Around Time
UHC
UnitedHealthcare
UM
Utilization Management
UMP
Utilization Management Program
UR
Utilization Review
VBP
Value Based Purchasing
VBPI
Value Based Purchasing Initiative
VFC
Vaccine for Children
WBA
WBA Market Research

Adolescent Well-Care Visits Appendix A2
A2-1
Adolescent Well-Care Visits (AWC)
SUMMARY OF CHANGES TO HEDIS 2014
Removed coding tables and replaced all coding table references with value set references.
Description
The percentage of enrolled members 12–21 years of age who had at least one comprehensive well-care visit
with a PCP or an OB/GYN practitioner during the measurement year.
Note: This measure has the same structure as measures in the Effectiveness of Care domain. Organizations
should follow the Guidelines for Effectiveness of Care Measures when calculating this measure. Only the
Administrative Method of data collection may be used when reporting this measure for the commercial
population.
Eligible Population
Product lines
Commercial, Medicaid (report each product line separately).
Ages
12–21 years as of December 31 of the measurement year.
Continuous
enrollment
The measurement year.
Allowable gap
Members who have had no more than one gap in enrollment of up to 45 days
during the measurement year. To determine continuous enrollment for a Medicaid
member for whom enrollment is verified monthly, the member may not have more
than a 1-month gap in coverage (i.e., a member whose coverage lapses for 2
months [60 days] is not considered continuously enrolled).
Anchor date
December 31 of the measurement year.
Benefit
Medical.
Event/diagnosis
None.
Administrative Specification
Denominator
The eligible population.
Numerator
At least one comprehensive well-care visit (Well-Care Value Set) with a PCP or an
OB/GYN practitioner during the measurement year. The practitioner does not have
to be the practitioner assigned to the member.

Adolescent Well-Care Visits Appendix A2
A2-2
Hybrid Specification
Denominator
A systematic sample drawn from the eligible population for the Medicaid product line.
Organizations may reduce the sample size using the current year’s administrative rate
or the prior year’s audited rate.
Refer to Guidelines for Calculations and Sampling for information on reducing sample
size.
Numerator
At least one comprehensive well-care visit with a PCP or an OB/GYN practitioner
during the measurement year, as documented through either administrative data or
medical record review. The PCP does not have to be assigned to the member.
Administrative
Refer to Administrative Specification to identify positive numerator hits from the
administrative data.
Medical record
Documentation in the medical record must include a note indicating a visit to a PCP or
OB/GYN practitioner, the date when the well-care visit occurred and evidence of all of
the following:
A health and developmental history (physical and mental).
A physical exam.
Health education/anticipatory guidance.
Do not include services rendered during an inpatient or ED visit.
Preventive services may be rendered on visits other than well-child visits. Well-child
preventive services count toward the measure, regardless of the primary intent of the
visit, but services that are specific to an acute or chronic condition do not count toward
the measure.
Visits to school-based clinics with practitioners whom the organization would consider
PCPs may be counted if documentation that a well-care exam occurred is available in
the medical record or administrative system in the time frame specified by the
measure. The PCP does not have to be assigned to the member.
The organization may count services that occur over multiple visits, as long as all
services occur in the time frame specified by the measure.
Note
Refer to Appendix 3 for the definition of PCP and OB/GYN and other prenatal care practitioners.
This measure is based on the CMS and American Academy of Pediatrics guidelines for EPSDT visits.
Refer to the American Academy of Pediatrics Guidelines for Health Supervision at www.aap.org and Bright
Futures: Guidelines for Health Supervision of Infants, Children and Adolescents (published by the National
Center for Education in Maternal and Child Health) at www.Brightfutures.org for more information about
well-care visits.

Adolescent Well-Care Visits Appendix A2
A2-3
Data Elements for Reporting
Organizations that submit HEDIS data to NCQA must provide the following data elements.
Table AWC-1/2: Data Elements for Adolescent Well-Care Visits
Administrative
Hybrid
Measurement year
Data collection methodology (Administrative or Hybrid)
Eligible population
Number of numerator events by administrative data in eligible population (before
exclusions)
Current year’s administrative rate (before exclusions)
Minimum required sample size (MRSS) or other sample size
Oversampling rate
Final sample size (FSS)
Number of numerator events by administrative data in FSS
Administrative rate on FSS
Number of original sample records excluded because of valid data errors
Number of employee/dependent medical records excluded
Records added from the oversample list
Denominator
Numerator events by administrative data
Numerator events by medical records
Reported rate
Lower 95% confidence interval
Upper 95% confidence interval

Controlling High Blood Pressure Appendix A3
A3-1
Controlling High Blood Pressure (CBP)
SUMMARY OF CHANGES TO HEDIS 2014
Removed coding tables and replaced all coding table references with value set references.
Removed “Telephone call record” as an acceptable method for confirming the hypertension diagnosis.
Clarified step 2 of the numerator to state when a BP reading is not compliant.
Revised the Optional Exclusion criteria to allow exclusion of all members who had a nonacute inpatient
encounter during the measurement year (previously the exclusion was limited to nonacute inpatient
admissions).
Description
The percentage of members 18–85 years of age who had a diagnosis of hypertension (HTN) and whose BP
was adequately controlled (<140/90) during the measurement year. Use the Hybrid Method for this measure.
Definitions
Adequate control
Both a representative systolic BP <140 mm Hg and a representative diastolic BP
<90 mm Hg (BP in the normal or high-normal range).
Representative
BP
The most recent BP reading during the measurement year (as long as it occurred
after the diagnosis of hypertension was made). If multiple BP measurements occur
on the same date, or are noted in the chart on the same date, the lowest systolic and
lowest diastolic BP reading should be used. If no BP is recorded during the
measurement year, assume that the member is “not controlled.”
Eligible Population
Product lines
Commercial, Medicaid, Medicare (report each product line separately).
Ages
18–85 years as of December 31 of the measurement year.
Continuous
enrollment
The measurement year.
Allowable gap
No more than one gap in continuous enrollment of up to 45 days during the
measurement year. To determine continuous enrollment for a Medicaid beneficiary
for whom enrollment is verified monthly, the member may not have more than a
1-month gap in coverage (i.e., a member whose coverage lapses for 2 months [60
days] is not considered continuously enrolled).
Anchor date
December 31 of the measurement year.
Benefit
Medical.

Controlling High Blood Pressure Appendix A3
A3-2
Event/
diagnosis
Members are identified as hypertensive if there is at least one outpatient visit
(Outpatient CPT Value Set) with a diagnosis of hypertension (Hypertension Value Set)
during the first six months of the measurement year.
Note: In order to increase the specificity of the eligible population, only CPT codes are
used to identify outpatient visits.
Hybrid Specification
Denominator
A systematic sample drawn from the eligible population for each product line whose
diagnosis of hypertension is confirmed by chart review. The organization may reduce
the sample size using the prior years audited, product line-specific rate. Refer to the
Guidelines for Calculations and Sampling for information on reducing the sample size.
To confirm the diagnosis of hypertension, the organization must find notation of one of
the following in the medical record on or before June 30 of the measurement year:
HTN.
High BP (HBP).
Elevated BP (BP).
Borderline HTN.
Intermittent HTN.
History of HTN.
Hypertensive vascular disease (HVD).
Hyperpiesia.
Hyperpiesis.
The notation of hypertension may appear on or before June 30 of the measurement
year, including prior to the measurement year. It does not matter if hypertension was
treated or is currently being treated. The notation indicating a diagnosis of hypertension
may be recorded in any of the following documents:
Problem list (this may include a diagnosis prior to June 30 of the measurement year
or an undated diagnosis; see Note at the end of this section).
Office note.
Subjective, Objective, Assessment, Plan (SOAP) note.
Encounter form.
Diagnostic report.
Hospital discharge summary.
Statements such as “rule out HTN,” “possible HTN,” “white-coat HTN,” “questionable
HTN” and “consistent with HTN” are not sufficient to confirm the diagnosis if such
statements are the only notations of hypertension in the medical record.
Identifying
the medical
record
Use one medical record for both the confirmation of the diagnosis of hypertension and
the representative BP. All eligible BP measurements recorded in the record must be
considered. If an organization cannot find the medical record, the member remains in
the measure denominator and is considered noncompliant for the numerator.
Use the following steps to find the appropriate medical record to review.
_____________
Current Procedural Terminology © 2013 American Medical Association. All rights reserved.

Controlling High Blood Pressure Appendix A3
A3-3
Step 1
Identify the member’s PCP.
If the member had more than one PCP for the time period, identify the PCP who most
recently provided care to the member.
If the member did not visit a PCP for the time period or does not have a PCP, identify
the practitioner who most recently provided care to the member.
If a practitioner other than the member’s PCP manages the hypertension, the
organization may use the medical record of that practitioner.
Step 2
Use one medical record to both confirm the diagnosis for the denominator and identify
the representative BP level for the numerator. There are circumstances in which the
organization may need to go to a second medical record to either confirm the
diagnosis or obtain the BP reading, as in the following two examples.
If a member sees one PCP during the denominator confirmation period (on or before
June 30 of the measurement year) and another PCP after June 30, the diagnosis of
hypertension and the BP reading may be identified through two different medical
records.
If a member has the same PCP for the entire measurement year, but it is clear from
claims or medical record data that a specialist (e.g., cardiologist) manages the
member’s hypertension after June 30, the organization may use the PCP’s chart to
confirm the diagnosis and use the specialist’s chart to obtain the BP reading. For
example, if all recent claims coded with 401 came from the specialist, the organization
may use this chart for the most recent BP reading. If the member did not have any visit
with the specialist prior to June 30 of the measurement year, the organization must go
to another medical record to confirm the diagnosis.
Numerator
The number of members in the denominator whose most recent BP is adequately
controlled during the measurement year. For a member’s BP to be controlled, both the
systolic and diastolic BP must be <140/90 (adequate control). To determine if a
member’s BP is adequately controlled, the representative BP must be identified.
Administrative
None.
Medical record
Follow the steps below to determine representative BP.
Step 1
Identify the most recent BP reading noted during the measurement year. The reading
must occur after the date when the diagnosis of hypertension was confirmed. Do not
include BP readings:
Taken during an acute inpatient stay or an ED visit.
Taken during an outpatient visit which was for the sole purpose of having a
diagnostic test or surgical procedure performed (e.g., sigmoidoscopy, removal of
a mole).
Obtained the same day as a major diagnostic or surgical procedure (e.g., stress
test, administration of IV contrast for a radiology procedure, endoscopy).
Reported by or taken by the member.

Controlling High Blood Pressure Appendix A3
A3-4
Step 2
Identify the lowest systolic and lowest diastolic BP reading from the most recent BP notation
in the medical record. If multiple readings were recorded for a single date, use the lowest
systolic and lowest diastolic BP on that date as the representative BP. The systolic and
diastolic results do not need to be from the same reading.
The member is not compliant if the BP reading is ≥140/90 or is missing, or if there is no BP
reading during the measurement year or if the reading is incomplete (e.g., the systolic or
diastolic level is missing).
Exclusions (optional)
Exclude from the eligible population all members with evidence of end-stage renal disease (ESRD) (ESRD
Value Set; ESRD Obsolete Value Set) or kidney transplant (Kidney Transplant Value Set) on or prior to
December 31 of the measurement year. Documentation in the medical record must include a dated note
indicating evidence of ESRD, kidney transplant or dialysis.
Exclude from the eligible population all members with a diagnosis of pregnancy (Pregnancy Value Set) during
the measurement year.
Exclude from the eligible population all members who had a nonacute inpatient encounter (Nonacute Care
Value Set) during the measurement year.
Note
Organizations may use an undated notation of hypertension on problem lists. Problem lists generally indicate
established conditions; to discount undated entries might hinder confirmation of the denominator.
Organizations generally require an oversample of 10 percent–15 percent to meet the MRSS for confirmed
cases of hypertension.

Controlling High Blood Pressure Appendix A3
A3-5
Data Elements for Reporting
Organizations that submit HEDIS data to NCQA must provide the following data elements.
Table CBP-1/2/3: Data Elements for Controlling High Blood Pressure
Hybrid
Measurement year
Data collection methodology (Hybrid)
Eligible population
Number of numerator events by administrative data in eligible population (before exclusions)
Current year’s administrative rate (before exclusions)
Minimum required sample size (MRSS) or other sample size
Oversampling rate
Final sample size (FSS)
Number of numerator events by administrative data in FSS
Administrative rate on FSS
Number of original sample records excluded because of valid data errors
Number of records excluded because of false-positive diagnoses
Number of administrative data records excluded
Number of medical record data records excluded
Number of employee/dependent medical records excluded
Records added from the oversample list
Denominator
Numerator events by administrative data
Numerator events by medical records
Reported rate
Lower 95% confidence interval
Upper 95% confidence interval

A4-1
Appendix A4
Table A – HealthChoice Organizations
HEDIS 2014 Results, page one of four
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2014
ACC
JMS
MPC
MSFC
PP
RHP
UHC
MARR
Adult BMI Assessment (ABA)
1
61.3%
72.0%
1
90.7%
80.2%
1
48.7%
70.2%
1
76.4%
82.6%
1
59.9%
82.9%
1
NA
2
1
49.1%
68.9%
76.1%
Avoidance of Antibiotic Treatment in Adults with
Acute Bronchitis (AAB)
23.7%
20.6%
23.88%
21.9%
35.5%
35.2%
19.7%
19.9%
22.0%
16.1%
14.1%
15.2%
21.1%
18.9%
23.94%
NA
2
19.6%
16.0%
20.8%
23.5%
Childhood Immunization Status (CIS) – Combination
2 (DTaP, IPV, MMR, HiB, Hep B, VZV)
85.6%
84.7%
81.3%
80.6%
86.1%
86.5%
81.8%
76.9%
73.7%
89.5%
85.4%
88.1%
86.0%
86.8%
83.1%
NA
2
82.7%
70.3%
73.0%
80.9%
Childhood Immunization Status (CIS) – Combination
3 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV)
81.9%
83.5%
78.2%
78.7%
83.7%
86.1%
80.8%
74.3%
72.09%
87.6%
83.7%
85.9%
83.7%
83.8%
80.8%
NA
2
78.8%
66.7%
71.3%
79.1%
Childhood Immunization Status (CIS) – Combination
4 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, Hep A)
39.1%
75.9%
73.6%
33.3%
80.9%
84.8%
32.8%
67.4%
62.8%
41.6%
80.3%
81.3%
38.8%
73.8%
69.4%
NA
2
37.2%
58.9%
66.2%
73.0%
Childhood Immunization Status (CIS) – Combination
5 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, RV)
59.7%
61.3%
63.9%
57.9%
59.4%
71.7%
53.5%
55.3%
47.0%
63.3%
56.0%
70.1%
55.1%
59.6%
54.6%
NA
2
57.2%
52.0%
56.9%
60.7%
Childhood Immunization Status (CIS) – Combination
6 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV,
Influenza)
48.6%
49.7%
49.3%
33.3%
39.0%
47.8%
39.2%
42.4%
37.7%
57.4%
55.2%
59.4%
51.4%
51.5%
49.5%
NA
2
41.8%
38.2%
44.3%
48.0%
Childhood Immunization Status (CIS) – Combination
7 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, Hep A,
RV)
30.1%
57.8%
60.7%
25.5%
59.0%
71.3%
20.2%
51.4%
44.0%
31.1%
54.3%
66.7%
25.3%
56.2%
50.7%
NA
2
28.2%
47.2%
54.7%
58.0%
Childhood Immunization Status (CIS) – Combination
8 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, Hep A,
Influenza)
25.7%
47.3%
47.9%
21.3%
39.0%
47.4%
17.0%
38.7%
34.9%
28.2%
53.5%
56.2%
24.2%
48.3%
44.4%
NA
2
21.7%
35.3%
41.4%
45.4%
Childhood Immunization Status (CIS) – Combination
9 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, RV,
Influenza)
38.2%
38.5%
42.4%
25.0%
29.5%
40.9%
29.2%
33.8%
28.4%
43.8%
38.7%
49.9%
38.8%
41.1%
36.3%
NA
2
32.8%
31.6%
37.0%
39.1%
Childhood Immunization Status (CIS) – Combination
10 (DTaP, IPV, MMR, HiB, Hep B, VZV, PCV, Hep A,
RV, Influenza)
20.6%
37.1%
41.2%
18.1%
29.5%
40.9%
12.2%
31.0%
27.7%
22.1%
37.7%
47.0%
17.9%
39.7%
34.3%
NA
2
17.5%
29.2%
35.3%
37.7%
Immunizations for Adolescents (IMA) Combination 1
(Meningococcal, Tdap/Td)
56.7%
65.0%
69.4%
73.2%
70.66%
75.5%
51.1%
57.6%
62.7%
70.7%
70.69%
70.7%
52.0%
67.4%
74.5%
NA
2
48.4%
56.4%
63.4%
69.4%
Well-Child Visits in the First 15 months of Life (W15)
– No well-child visits
3
1.6%
1.0%
1.012%
0.87%
2.7%
3.1%
1.4%
1.11%
0.5%
1.3%
1.013%
1.2%
1.1%
1.14%
1.1%
NA
2
0.9%
2.2%
1.9%
1.5%
Well-Child Visits in the First 15 months of Life (W15)
– DHMH Five or Six-or-more visits (rate constructed
by adding together HEDIS five visits and six-or-more
visits rates)
87.3%
86.1%
88.9%
84.0%
85.9%
84.4%
89.9%
77.8%
83.6%
88.2%
89.2%
86.0%
84.3%
84.3%
83.7%
NA
2
86.8%
82.1%
87.4%
85.7%
Well-Child Visits in the Third, Fourth, Fifth and Sixth
Years of Life (W34)
86.4%
83.6%
83.9%
88.9%
87.7%
88.9%
89.1%
87.5%
88.8%
82.3%
79.6%
83.5%
82.4%
80.7%
83.8%
NA
2
83.1%
83.8%
75.0%
84.0%
Adolescent Well-Care Visits (AWC)
61.9%
68.1%
67.9%
79.9%
76.9%
76.7%
75.8%
60.2%
68.8%
67.7%
69.4%
67.8%
66.1%
67.6%
61.6%
NA
2
55.7%
59.7%
60.8%
67.3%
Weight Assessment and Counseling for Nutrition and
Physical Activity for Children/Adolescents (WCC) -
BMI Percentile- Total Rate
5
5
49.5%
5
5
92.2%
5
5
46.5%
5
5
59.8%
5
5
52.1%
5
5
NA
2
5
5
45.5%
57.6%
Weight Assessment and Counseling for Nutrition and
Physical Activity for Children/Adolescents (WCC) –
Counseling for Nutrition – Total Rate
5
5
59.0%
5
5
94.4%
5
5
54.4%
5
5
74.1%
5
5
54.2%
5
5
NA
2
5
5
67.6%
67.3%
Weight Assessment and Counseling for Nutrition and
Physical Activity for Children/Adolescents (WCC) –
Counseling for Physical Activity – Total Rate
5
5
51.4%
5
5
89.8%
5
5
58.8%
5
5
72.9%
5
5
44.7%
5
5
NA
2
5
5
60.6%
63.0%
Appropriate Testing for Children with Pharyngitis
(CWP)
68.8%
75.9%
78.36%
74.5%
75.3%
70.8%
76.9%
77.4%
78.42%
85.9%
85.2%
86.9%
74.5%
78.2%
80.5%
NA
2
76.4%
79.8%
83.1%
79.7%
1
New measure for HEDIS 2013.
2
When denominator is less than 30 eligible members, NA is automatically assigned as the performance score.
3
A lower rate indicates better performance.
5
New measure for HEDIS 2014.
MARR = Maryland Average Reportable Rate
ACC = AMERIGROUP Community Care JMS = Jai Medical Systems MPC = Maryland Physicians Care MSFC = MedStar Family Choice PP = Priority Partners RHP = Riverside Health Plan UHC = UnitedHealthcare

A4-2
Table A – HealthChoice Organizations
HEDIS 2014 Results, page two of four
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2014
ACC
JMS
MPC
MSFC
PP
RHP
UHC
MARR
Use of Appropriate Medications for People with
Asthma (ASM) – Ages 5–11
91.4%
88.7%
90.3%
94.2%
91.4%
93.59%
93.0%
92.3%
91.4%
96.7%
93.7%
93.62%
91.7%
92.3%
91.6%
NA
2
95.7%
96.1%
91.9%
92.1%
Use of Appropriate Medications for People with
Asthma (ASM) – Total Ages 12–18
88.2%
86.2%
87.8%
100.0%
92.9%
86.0%
91.1%
92.3%
90.4%
93.3%
90.2%
94.2%
90.8%
89.6%
88.5%
NA
2
96.6%
93.4%
88.0%
89.1%
Use of Appropriate Medications for People with
Asthma (ASM) – Total Ages 19–50
78.0%
79.5%
73.7%
91.3%
93.3%
81.3%
82.8%
81.8%
80.1%
85.2%
76.8%
75.2%
77.9%
80.7%
76.8%
NA
2
95.1%
88.0%
72.9%
76.7%
Use of Appropriate Medications for People with
Asthma (ASM) – Total Ages 51–64
71.2%
77.7%
68.6%
83.7%
82.0%
71.43%
81.7%
78.5%
76.3%
NA
77.1%
NA
69.2%
77.0%
73.0%
NA
2
95.0%
94.1%
79.0%
73.7%
Use of Appropriate Medications for People with
Asthma (ASM) – Total Ages 5–64
89.1%
86.5%
86.29%
95.7%
90.7%
83.6%
90.7%
88.7%
86.97%
95.5%
88.8%
90.1%
89.3%
88.9%
87.02%
NA
2
96.7%
94.0%
86.28%
86.7%
Use of Appropriate Medications for People with
Asthma (ASM) – Total Ages 5–50
4
88.5%
86.7%
86.8%
93.9%
92.5%
86.4%
89.8%
89.2%
87.53%
93.6%
89.4%
90.1%
88.9%
89.3%
87.6%
NA
2
95.9%
94.0%
86.6%
87.51%
Medication Management for People With Asthma
(MMA) – Total 50% of treatment period
1
44.8%
45.8%
1
53.2%
49.4%
1
49.4%
57.9%
1
52.4%
51.9%
1
40.3%
43.3%
1
NA
2
1
47.3%
49.9%
49.7%
Medication Management for People With Asthma
(MMA) – Total 75% of treatment period
1
24.1%
22.9%
1
28.9%
24.5%
1
26.6%
32.9%
1
28.7%
26.6%
1
19.7%
20.0%
1
NA
2
1
26.7%
27.8%
25.8%
Appropriate Treatment for Children with Upper
Respiratory Infection (URI)
86.13%
85.1%
86.5%
89.8%
85.2%
83.0%
86.08%
86.06%
86.6%
89.0%
86.13%
84.3%
86.01%
85.0%
86.0%
NA
2
80.2%
80.1%
82.0%
84.7%
Asthma Medication Ratio (AMR)
5
68.59%
5
60.5%
5
69.1%
5
73.7%
5
69.6%
5
NA
2
5
69.8%
68.56%
Use of Spirometry Testing in the Assessment and
Diagnosis of COPD (SPR)
5
25.8%
5
26.3%
5
21.1%
5
34.5%
5
23.7%
5
NA
2
5
25.6%
26.2%
Pharmacotherapy Management of COPD
Exacerbation (PCE) – Systemic Corticosteroid Rate
5
73.6%
5
69.2%
5
72.6%
5
76.3%
5
69.7%
5
NA
2
5
78.2%
73.3%
Pharmacotherapy Management of COPD
Exacerbation (PCE) – Bronchodilator Rate
5
87.5%
5
82.5%
5
84.93%
5
90.3%
5
84.0%
5
NA
2
5
84.88%
85.7%
Children and Adolescents’ Access to Primary Care
Practitioners (CAP) - Age 12–24 months
97.45%
97.5%
97.8%
92.9%
91.1%
94.7%
96.8%
97.1%
96.5%
96.6%
96.6%
96.4%
91.4%
90.3%
89.8%
NA
2
97.41%
96.7%
96.3%
96.6%
Children and Adolescents’ Access to Primary Care
Practitioners (CAP) - Age 25 months–6 years
92.8%
92.6%
92.8%
89.3%
90.4%
88.7%
90.7%
89.0%
90.0%
91.4%
90.3%
89.8%
92.9%
92.5%
93.5%
NA
2
92.1%
91.1%
91.1%
90.8%
Children and Adolescents’ Access to Primary Care
Practitioners (CAP) - Age 7–11 years
93.6%
93.9%
94.3%
94.0%
93.3%
93.8%
92.0%
91.5%
92.1%
92.9%
92.5%
93.5%
90.9%
92.5%
92.7%
NA
2
93.0%
93.3%
93.1%
93.5%
Children and Adolescents’ Access to Primary Care
Practitioners (CAP) - Age 12–19 years
89.3%
89.5%
90.5%
92.4%
91.7%
90.8%
88.4%
87.7%
88.5%
90.9%
92.5%
92.7%
91.6%
92.0%
91.9%
NA
2
88.5%
89.2%
90.1%
90.7%
Adults’ Access to Preventive/Ambulatory Health
Services (AAP) – Age 20–44 years
80.4%
79.7%
79.4%
75.5%
74.8%
72.9%
81.2%
81.4%
81.1%
79.6%
79.9%
79.7%
83.7%
83.5%
81.7%
NA
2
80.3%
80.2%
80.36%
79.2%
Adults’ Access to Preventive/Ambulatory Health
Services (AAP) – Age 45–64 years
87.0%
86.4%
87.2%
88.8%
87.8%
86.58%
87.28%
86.8%
87.8%
85.9%
86.2%
86.9%
0.0%
0.0%
0.0%
NA
2
87.31%
87.5%
87.8%
87.5%
Breast Cancer Screening (BCS)
48.5%
49.1%
58.1%
63.9%
60.8%
69.4%
43.6%
43.9%
48.5%
54.5%
56.8%
64.4%
49.9%
51.5%
57.0%
NA
2
46.6%
48.4%
52.7%
58.3%
Cervical Cancer Screening (CCS)
75.71%
73.6%
79.64%
78.5%
80.9%
79.5%
73.6%
74.0%
79.58%
75.74%
70.9%
74.0%
73.9%
75.0%
75.9%
NA
2
69.5%
69.8%
62.8%
75.2%
Chlamydia Screening in Women (CHL) – Age 16–20
years
61.1%
62.6%
62.4%
84.0%
81.1%
86.7%
58.5%
58.1%
58.2%
57.4%
59.6%
54.8%
62.6%
61.8%
61.5%
NA
2
57.1%
56.9%
55.4%
63.2%
1
New measure for HEDIS 2013.
2
When denominator is less than 30 eligible members, NA is automatically assigned as the performance score.
3
A lower rate indicates better performance.
4
HEDIS specifications changed in 2012, and this age range is no longer reported. For 2013 and 2014, this rate is being calculated by HDC.
5
New measure for HEDIS 2014.
MARR = Maryland Average Reportable Rate NHM = National HEDIS Mean
ACC = AMERIGROUP Community Care JMS = Jai Medical Systems MPC = Maryland Physicians Care MSFC = MedStar Family Choice PP = Priority Partners RHP = Riverside Health Plan UHC = UnitedHealthcare

A4-3
Table A – HealthChoice Organizations
HEDIS 2014 Results page three of four
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2014
ACC
JMS
MPC
MSFC
PP
RHP
UHC
MARR
Chlamydia Screening in Women (CHL) – Age 21–24
years
70.6%
72.5%
71.9%
77.4%
63.9%
72.3%
66.6%
67.6%
67.1%
70.5%
74.0%
68.4%
69.8%
68.9%
69.9%
NA
2
64.8%
63.7%
64.8%
69.1%
Chlamydia Screening in Women (CHL) – Total (16–
24) years
64.8%
66.4%
66.0%
81.3%
74.2%
81.2%
62.0%
62.3%
62.0%
62.5%
65.0%
60.1%
65.4%
64.6%
64.8%
NA
2
60.0%
59.5%
59.0%
65.5%
Prenatal and Postpartum Care (PPC) – Timeliness of
Prenatal Care
90.4%
87.8%
84.2%
86.2%
82.9%
85.8%
82.1%
86.279
%
84.9%
87.7%
86.280
%
85.4%
87.1%
89.3%
90.9%
52.2%
83.8%
84.7%
87.1%
81.5%
Prenatal and Postpartum Care (PPC) – Postpartum
Care
70.7%
71.5%
71.6%
78.1%
83.7%
78.5%
71.3%
68.4%
71.9%
74.0%
74.4%
72.0%
73.0%
72.5%
75.6%
43.5%
64.7%
60.3%
63.8%
68.1%
Frequency of Ongoing Prenatal Care (FPC) – Less
than 21% of expected visits
3
3.4%
4.2%
8.2%
2.8%
3.6%
2.2%
5.7%
10.6%
5.6%
2.9%
2.7%
4.4%
7.7%
4.4%
4.4%
37.0%
5.4%
12.1%
5.8%
9.7%
Frequency of Ongoing Prenatal Care (FPC) – Greater
than or equal to 81% of expected visits
80.3%
72.2%
75.5%
76.9%
75.8%
70.8%
69.6%
60.1%
70.6%
82.7%
79.3%
71.3%
64.7%
78.8%
78.8%
21.7%
72.2%
70.8%
73.2%
66.0%
Controlling High Blood Pressures (CBP)
1
47.0%
49.0%
1
52.3%
56.2%
1
23.9%
46.8%
1
70.5%
65.5%
1
59.1%
57.0%
1
NA
2
1
43.1%
42.3%
52.8%
Persistence of Beta-Blocker Treatment After a Heart
Attack (PBH)
5
NA
5
NA
5
87.5%
5
NA
5
86.1%
5
NA
2
5
82.9%
85.5%
Comprehensive Diabetes (CDC) – Hemoglobin A1c
(HbA1c) Testing
78.8%
81.1%
83.4%
90.5%
89.8%
89.1%
77.1%
76.0%
79.5%
88.1%
83.5%
84.7%
81.9%
82.4%
78.1%
NA
2
75.9%
78.1%
79.1%
82.3%
Comprehensive Diabetes (CDC) – HbA1c Poor
Control (>9.0%)
3
43.3%
44.0%
38.8%
33.6%
35.4%
31.0%
56.7%
52.6%
48.6%
27.5%
35.3%
37.2%
38.3%
41.7%
48.1%
NA
2
51.1%
54.3%
45.5%
41.5%
Comprehensive Diabetes (CDC) – HbA1c Control (<
8.0%)
48.4%
47.1%
51.4%
56.2%
54.7%
61.5%
37.0%
39.9%
43.3%
57.7%
58.9%
54.0%
50.8%
49.1%
44.3%
NA
2
42.1%
38.9%
46.47%
50.2%
Comprehensive Diabetes (CDC) – Eye Exam
(Retinal) Performed
62.2%
69.3%
65.4%
80.8%
80.1%
79.6%
76.2%
64.6%
72.0%
75.7%
72.8%
71.1%
71.6%
78.1%
71.0%
NA
2
60.8%
57.7%
56.9%
69.3%
Comprehensive Diabetes (CDC) – LDL-C Screening
77.4%
76.0%
76.9%
89.4%
88.5%
87.8%
71.3%
69.2%
72.9%
81.7%
77.4%
78.4%
74.9%
73.1%
70.1%
NA
2
72.3%
74.2%
77.4%
77.2%
Comprehensive Diabetes (CDC) – LDL-C Control
(<100 mg/dL)
35.9%
36.2%
36.0%
48.7%
44.2%
45.26%
27.0%
28.0%
30.5%
44.6%
41.1%
39.9%
36.1%
44.5%
45.28%
NA
2
35.0%
30.7%
35.0%
38.7%
Comprehensive Diabetes (CDC) – Medical Attention
for Nephropathy
79.7%
73.6%
75.7%
94.7%
93.6%
93.1%
75.2%
74.4%
75.3%
89.6%
78.8%
82.7%
79.0%
77.6%
73.8%
NA
2
72.7%
74.2%
75.9%
79.4%
Comprehensive Diabetes (CDC) – Blood Pressure
Control (<140/80 mm Hg)
31.1%
29.1%
34.4%
34.1%
38.0%
39.2%
24.1%
30.3%
32.0%
46.3%
55.7%
44.3%
42.2%
42.6%
44.1%
NA
2
33.8%
25.3%
32.4%
37.7%
Comprehensive Diabetes (CDC) – Blood Pressure
Control (<140/90 mm Hg)
54.6%
48.4%
55.6%
54.74%
59.1%
60.4%
45.7%
47.1%
55.4%
73.3%
73.7%
70.1%
65.1%
63.3%
64.2%
NA
2
54.74%
47.0%
51.6%
59.5%
Use of Imaging Studies for Low Back Pain (LBP)
78.5%
77.8%
76.7%
81.6%
70.9%
77.2%
76.8%
75.2%
76.6%
74.5%
73.1%
73.3%
74.7%
75.0%
75.2%
NA
2
75.5%
74.8%
73.4%
75.4%
Disease-Modifying Anti-Rheumatic Drug Therapy for
Rheumatoid Arthritis (ART)
1
61.8%
60.0%
1
NA
NA
1
71.9%
73.8%
1
NA
NA
1
69.5%
67.6%
1
NA
2
1
73.3%
67.7%
67.3%
Annual Monitoring for Patients on Persistent
Medications (MPM) - members on angiotensin
converting enzyme (ACE) inhibitors or angiotensin
receptor blockers (ARB).
1
90.1%
89.0%
1
95.8%
95.1%
1
88.9%
87.0%
1
87.6%
90.2%
1
88.224
%
88.1%
1
NA
2
1
88.222
%
88.6%
89.7%
1
New measure for HEDIS 2013.
2
When denominator is less than 30 eligible members, NA is automatically assigned as the performance score.
3
A lower rate indicates better performance.
4
HEDIS specifications changed in 2012, and this age range is no longer reported. For 2013 and 2014, this rate is being calculated by HDC.
5
New measure for HEDIS 2014.
MARR = Maryland Average Reportable Rate NHM = National HEDIS Mean
ACC = AMERIGROUP Community Care JMS = Jai Medical Systems MPC = Maryland Physicians Care MSFC = MedStar Family Choice PP = Priority Partners RHP = Riverside Health Plan UHC = UnitedHealthcare

A4-4
Table A – HealthChoice Organizations
HEDIS 2014 Results – page four of four
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2014
ACC
JMS
MPC
MSFC
PP
RHP
UHC
MARR
Annual Monitoring for Patients on Persistent
Medications (MPM) - members on digoxin
1
95.8%
95.7%
1
NA
2
NA
2
1
91.4%
92.2%
1
NA
2
NA
2
1
91.5%
88.9%
1
NA
2
1
93.4%
86.4%
90.8%
Annual Monitoring for Patients on Persistent
Medications (MPM) - members on diuretics.
1
88.2%
86.9%
1
94.3%
94.1%
1
88.04%
86.2%
1
88.02%
88.5%
1
87.2%
87.4%
1
NA
2
1
87.8%
87.5%
88.4%
Annual Monitoring for Patients on Persistent
Medications (MPM) - members on anticonvulsants
1
66.0%
66.3%
1
64.8%
75.6%
1
69.9%
70.42%
1
58.1%
67.1%
1
73.3%
68.3%
1
NA
2
1
72.4%
75.0%
70.44%
Annual Monitoring for Patients on Persistent
Medications (MPM) - Total rate
1
86.2%
85.4%
1
93.1%
94.1%
1
88.0%
86.3%
1
84.1%
86.6%
1
87.3%
87.3%
1
NA
2
1
87.5%
87.7%
87.9%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Initiation 13–17 Years
41.0%
42.0%
37.7%
NA
2
NA
2
NA
2
49.7%
42.3%
38.9%
19.5%
5.0%
30.9%
47.4%
38.4%
41.8%
NA
2
49.8%
42.9%
44.3%
38.7%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Initiation 18+ Years
47.4%
41.9%
38.8%
46.7%
37.1%
45.4%
47.7%
43.1%
37.3%
36.6%
29.2%
43.2%
42.8%
38.5%
37.0%
NA
2
47.3%
47.9%
45.7%
41.2%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Initiation Overall Ages
46.4%
41.9%
38.6%
46.5%
36.8%
45.2%
47.9%
43.0%
37.45%
35.5%
27.4%
41.7%
43.4%
38.5%
37.49%
NA
2
47.6%
47.3%
45.5%
41.0%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Engagement 13–17
Years
26.5%
27.7%
24.1%
NA
2
NA
2
NA
2
33.2%
26.5%
22.1%
9.8%
2.5%
19.8%
29.2%
22.6%
27.6%
NA
2
31.5%
24.0%
30.3%
24.8%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Engagement 18+
Years
20.7%
18.2%
17.9%
19.5%
15.4%
17.0%
24.0%
20.5%
19.8%
8.3%
5.5%
21.6%
18.7%
17.0%
17.2%
NA
2
17.0%
17.8%
20.8%
19.1%
Initiation and Engagement of Alcohol and Other Drug
Dependence Treatment (IET) – Engagement Overall
Ages
21.6%
19.7%
18.8%
19.4%
15.4%
16.9%
24.9%
21.0%
20.0%
8.4%
5.3%
21.4%
19.9%
17.6%
18.4%
NA
2
18.8%
18.5%
21.6%
19.5%
Identification of Alcohol and Other Drug Services
(IAD) – Any
2.5%
2.6%
2.7%
16.7%
15.8%
16.9%
6.2%
6.3%
6.0%
3.3%
3.1%
4.3%
5.2%
5.2%
5.0%
14.9%
4.0%
3.6%
4.7%
7.9%
Identification of Alcohol and Other Drug Services
(IAD) – Inpatient
0.6%
0.6%
0.5%
4.1%
3.8%
4.0%
1.3%
1.3%
0.95%
2.2%
0.90%
0.8%
1.1%
0.943%
0.9%
1.6%
0.9%
0.941%
1.03%
1.4%
Identification of Alcohol and Other Drug Services
(IAD) - Intensive Outpatient/Partial Hospitalization
0.33%
0.3%
0.3%
2.9%
2.5%
2.5%
0.94%
0.82%
0.7%
0.34%
0.18%
0.5%
0.8%
0.7%
0.6%
1.3%
0.43%
0.22%
0.0%
1.0%
Identification of Alcohol and Other Drug Services
(IAD) - Outpatient/ED
2.2%
2.4%
2.5%
15.2%
14.5%
15.6%
5.7%
5.8%
5.6%
2.5%
2.5%
3.9%
4.8%
4.9%
4.6%
11.9%
3.5%
3.0%
4.2%
6.9%
Ambulatory Care (AMB) – Outpatient visits per 1,000
member months
370.88
363.6
365.1
347.4
373.9
340.8
386.8
385.3
365.3
370
361.6
344.5
415.9
407.8
386.6
269.8
381
374.2
373.3
349.3
Ambulatory Care (AMB) – Emergency department
(ED) visits per 1,000 member months
3
60.7
59.8
56.2
91.3
93.4
90.1
78.8
79.3
74.6
72.3
70.8
62.66
65.7
66
62.7
66
65.8
65.2
62.1
67.8
Call Answer Timeliness (CAT)
78.9%
81.9%
89.7%
93.1%
95.0%
93.4%
91.1%
87.7%
89.2%
89.2%
89.4%
91.3%
73.1%
84.9%
71.0%
NA
2
85.5%
92.4%
89.4%
87.3%
1
New measure for HEDIS 2013.
2
When denominator is less than 30 eligible members, NA is automatically assigned as the performance score.
3
A lower rate indicates better performance.
MARR = Maryland Average Reportable Rate NHM = National HEDIS Mean
ACC = AMERIGROUP Community Care JMS = Jai Medical Systems MPC = Maryland Physicians Care MSFC = MedStar Family Choice PP = Priority Partners RHP = Riverside Health Plan UHC = UnitedHealthcare

A4-5
Appendix 4
Table A1 – HealthChoice Organizations Reporting PAC
HEDIS 2014 Results – page one of one
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
2012
2013
2014
ACC
PAC
JMS
PAC
PP
PAC
UHC
PAC
MARR PAC
Adult BMI Assessment (ABA)
32.6%
23.2%
25.0%
15.2%
NA
2
NA
2
30.7%
39.7%
23.9%
19.9%
22.8%
27.6%
24.4%
27.3%
25.5%
Adults’ Access to Preventive/Ambulatory Health Services (AAP) – Age 20–44 years
70.6%
71.5%
65.1%
72.8%
71.8%
65.3%
65.2%
64.0%
63.6%
69.8%
71.4%
67.2%
68.1%
66.7%
65.3%
Adults’ Access to Preventive/Ambulatory Health Services (AAP) – Age 45–64 years
80.5%
81.1%
79.6%
82.1%
82.6%
77.6%
76.8%
78.2%
79.3%
81.4%
82.5%
81.5%
78.7%
76.9%
79.5%
Breast Cancer Screening (BCS)
41.2%
42.5%
49.5%
52.6%
52.5%
63.2%
34.4%
37.5%
43.3%
38.0%
41.1%
48.0%
40.8%
40.3%
51.0%
Cervical Cancer Screening (CCS)
37.8%
39.8%
32.8%
66.1%
61.7%
52.6%
40.3%
40.2%
43.5%
38.9%
39.0%
34.3%
44.5%
42.8%
40.8%
Comprehensive Diabetes (CDC) – Hemoglobin A1c (HbA1c) Testing
80.9%
82.0%
81.9%
91.5%
86.6%
84.9%
78.5%
78.6%
79.2%
77.4%
78.8%
79.8%
81.6%
79.9%
81.5%
Comprehensive Diabetes (CDC) – HbA1c Poor Control (>9.0%)
3
49.8%
50.3%
53.0%
32.1%
38.1%
40.8%
52.2%
58.2%
57.6%
44.0%
57.5%
61.9%
45.5%
51.8%
53.4%
Comprehensive Diabetes (CDC) – HbA1c Control (< 8.0%)
44.0%
42.5%
37.8%
58.6%
52.2%
49.8%
40.3%
35.8%
34.6%
47.4%
36.6%
31.1%
46.7%
41.0%
53.4%
Comprehensive Diabetes (CDC) – Eye Exam (Retinal) Performed
34.9%
31.7%
37.7%
66.2%
62.1%
49.1%
31.0%
33.4%
33.2%
42.3%
35.1%
35.6%
40.7%
37.6%
38.9%
Comprehensive Diabetes (CDC) – LDL-C Screening
74.6%
74.5%
76.4%
90.5%
87.3%
82.1%
68.1%
70.2%
71.1%
73.2%
75.0%
72.4%
76.2%
74.5%
75.5%
Comprehensive Diabetes (CDC) – LDL-C Control (<100 mg/dL)
29.7%
30.4%
29.7%
45.7%
44.9%
41.0%
26.3%
45.9%
46.0%
40.1%
28.1%
22.2%
34.5%
35.1%
34.7%
Comprehensive Diabetes (CDC) – Medical Attention for Nephropathy
80.4%
76.1%
80.9%
94.4%
90.7%
89.3%
73.5%
77.3%
79.0%
79.5%
79.1%
77.3%
81.5%
79.4%
81.7%
Comprehensive Diabetes (CDC) – Blood Pressure Control (<140/80 mm Hg)
0.0%
0.0%
13.7%
33.8%
34.2%
30.5%
2.4%
0.0%
1.6%
24.8%
0.2%
0.1%
17.5%
8.6%
11.5%
Comprehensive Diabetes (CDC) – Blood Pressure Control (<140/90 mm Hg)
0.0%
0.0%
21.1%
56.4%
53.5%
52.9%
4.4%
0.0%
2.4%
42.8%
0.2%
0.1%
29.6%
17.0%
19.1%
2
When denominator is less than 30 eligible members, NA is automatically assigned as the performance score.
3
A lower rate indicates better performance.
MARR = Maryland Average Reportable Rate NHM = National HEDIS Mean
ACC = AMERIGROUP Community Care JMS = Jai Medical Systems PP = Priority Partners UHC = UnitedHealthcare

HealthChoice
MARYLAND’S MEDICAID HEALTH PLAN PROGRAM

